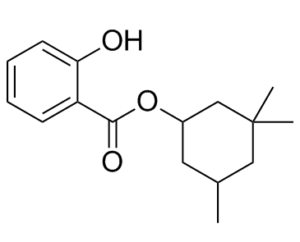
Homosalate is an organic chemical substance used as a UV filter in cosmetics and sunscreens to absorb UVB rays, shielding the skin from sun damage.
Physicochemical Properties
| Molecular Formula | C16H22O3 |
| Molecular Weight | 262.35 |
| Exact Mass | 262.156 |
| Elemental Analysis | C, 73.25; H, 8.45; O, 18.30 |
| CAS # | 118-56-9 |
| Related CAS # | Homosalate-d4 |
| PubChem CID | 8362 |
| Appearance | Colorless to light yellow liquid |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 341.1±15.0 °C at 760 mmHg |
| Flash Point | 132.3±13.2 °C |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.539 |
| LogP | 5.82 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 19 |
| Complexity | 324 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C(OC1CC(C)(C)CC(C)C1)C2=CC=CC=C2O |
| InChi Key | WSSJONWNBBTCMG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H22O3/c1-11-8-12(10-16(2,3)9-11)19-15(18)13-6-4-5-7-14(13)17/h4-7,11-12,17H,8-10H2,1-3H3 |
| Chemical Name | (3,3,5-trimethylcyclohexyl) 2-hydroxybenzoate |
| Synonyms | Homomenthyl |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
Extracellular vesicles (EVs) released by Homosalate-treated cells increase a recipient epithelial tumor cell line's resistance to anchorage loss.[2] Homosalate (10 μM) robustly increased the release of extracellular vesicles (EVs) from MDA-MB-231 triple-negative breast cancer cells, as quantified by Nanoluciferase activity and nanoparticle tracking analysis (NTA). This effect was also observed in other tumor cell lines (HeLa, MCF7, Jurkat) and mouse cell lines (Pfal, E0771). EVs released from Homosalate-treated MDA-MB-231 cells were enriched in surface markers SLC3A2/CD98, CD40, and CD9, suggesting the induction of a specific EV subpopulation, likely of plasma membrane origin (ectosomes). Treatment of recipient MCF7 luminal breast cancer cells with EVs from Homosalate-treated MDA-MB-231 cells (5 x 10^8 particles) enhanced cell migration in a wound healing assay and conferred resistance to anchorage-loss (anoikis) in an ultralow-attachment plate assay, compared to EVs from control or Bafilomycin A1-treated cells. This effect was not due to protection against staurosporine-induced apoptosis. Homosalate at 10 μM did not significantly affect the viability of MDA-MB-231, HeLa, or MCF7 cells over 24 hours, but showed significant toxicity in Jurkat cells. The effect of Homosalate on EV release was distinct from that of Bafilomycin A1, which increased EVs originating from multivesicular bodies (exosomes). [2] |
| Cell Assay |
For EV isolation, 6 × 106 MDA-MB-231 parental cells were seeded per 15 cm culture dish in an ideal number of dishes to produce 70 × 106, 27 × 106 or 50 × 106 secreting cells. After 24 hours, cells were washed once with PBS and then 15 ml of serum- and phenol-red-free Leibovitz's L-15 were added. In phenol-red-free, serum-free Leibovitz's L-15 medium, DMSO (0.1%), Homosalate (10 μM), or Bafilomycin A1 (100 nM) were added before the cells were incubated for 24 or 16 hours, respectively. The following day, cells were counted, and Trypan Blue was used to calculate the percentage of cell viability. High-Content Screening (HCS) for EV Secretion: Stable MDA-MB-231 clones expressing Nanoluciferase (Nluc)-tagged CD63 or CD9 were used. Cells were seeded in 384-well plates (2,500-4,000 cells/well). After 24 hours, cells were washed and incubated in serum-free, phenol-red-free Leibovitz's L-15 medium containing 10 μM Homosalate (0.1% DMSO as vehicle) for 72 hours. Extracellular Nluc activity (indicative of EV secretion) was measured in the conditioned medium using the Nano-Glo Luciferase Assay System. Cell viability was simultaneously assessed by Hoechst 33342 nuclei staining and automated image analysis to exclude cytotoxic effects. [2] EV Isolation and Characterization: For detailed analysis, parental MDA-MB-231 cells (e.g., 27 x 10^6 to 50 x 10^6 cells) were treated with 10 μM Homosalate in serum-free medium for 24 hours. Conditioned media were collected, subjected to sequential centrifugation (300 g, 10 min; 2,000 g, 20 min), concentrated using 10 kDa molecular weight cut-off filters, and EVs were isolated by size-exclusion chromatography (SEC, qEV columns). EV-rich fractions (F7-11) were pooled and analyzed. Particle concentration and size were determined by Nanoparticle Tracking Analysis (NTA). EV markers (SLC3A2/CD98, CD63, CD9, Syntenin, CD81, Lamp-1) were analyzed by Western blot, loading equal amounts of either particles or material from an equal number of secreting cells. EV surface protein profiles were analyzed using a multiplexed bead-based flow cytometry assay (MACSPlex Exosome Kit) with capture beads coated with 37 different antibodies, followed by detection with a mix of anti-tetraspanin (CD9/CD63/CD81) or anti-SLC3A2/CD98 antibodies. [2] EV Functional Assays - Anoikis Resistance: MCF7 recipient cells (150,000 per well) were pre-treated for 1 hour with 5 x 10^8 SEC-purified EVs from Homosalate-, Bafilomycin A1-, or DMSO-treated MDA-MB-231 cells. Cells were then trypsinized and subjected to anoikis by culturing in serum-free DMEM with 0.1% BSA on ultra-low attachment plates for 24 hours. Subsequently, cells were collected, washed, trypsinized to disrupt aggregates, and transferred to xCELLigence plates. Cell adhesion and proliferation were monitored in real-time for 50 hours via impedance measurement. [2] EV Functional Assays - Migration: MCF7 cells were seeded in 24-well plates. After 24 hours, cells were incubated with 2 x 10^9 EVs from different treatments for 1 hour. A scratch wound was created with a pipette tip. Cells were washed and incubated with EV-depleted, serum-containing medium. Wound closure was monitored by time-lapse microscopy over 21 hours, and wound area was quantified using image analysis software. [2] EV Uptake Assay: Parental MDA-MB-231 cells were pre-treated with DMSO, 10 μM Homosalate (24h), or 100 nM Bafilomycin A1 (16h). They were then fed with 3 x 10^7 Nluc-CD9 EVs from MDA-MB-231 cells for 2 or 16 hours. After washing, intracellular Nluc activity was measured to assess EV uptake efficiency. [2] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion For local use only, no systemic absorption. For local use only, no systemic absorption. For local use only, no systemic absorption. For local use only, no systemic absorption. Metabolism / Metabolites For local use only, no systemic absorption. Biological Half-Life For local use only, no systemic absorption. |
| Toxicity/Toxicokinetics |
Protein Binding For local use only, no systemic absorption. In vitro, treatment with 10 μM Homosalate for 24 hours did not significantly reduce the viability of MDA-MB-231, HeLa, or MCF7 cells as assessed by Trypan Blue exclusion or Resazurin assay. However, under the same conditions, it significantly reduced the viability of Jurkat cells, and this toxicity may have contributed to the observed increase in EV release in this cell line. [2] The screening assay established a cell viability threshold (>85% live cells) to avoid false-positive hits due to compound-induced cell death, which can increase extracellular Nluc activity independently of specific EV secretion modulation. [2] |
| References |
[1]. Environ Pollut. 2018 Dec;243(Pt B):1263-1273. [2]. J Extracell Vesicles. 2022 Jul; 11(7): e12242. |
| Additional Infomation |
3,3,5-trimethylcyclohexylsalicylate appears as viscous or light yellow to slightly tan liquid or oil. (NTP, 1992) 2-hydroxybenzoic acid (3,3,5-trimethylcyclohexyl) ester is a benzoate ester and a member of phenols. It is functionally related to a salicylic acid. Homosalate is an organic compound that belongs to salicylates. It is an ester formed from salicylic acid and 3,3,5-trimethylcyclohexanol, a derivative of cyclohexanol. Salicylates prevent direct skin exposure to the sun’s harmful rays by absorbing ultraviolet (UV) light. Homosalate specifically absorbs short-wave UVB rays, which are associated with DNA damage and increased risk of skin cancer. It is a common ingredient in many commercially available sunscreens. There are no reported adverse effects from homosalate. Homosalate has been reported in Camellia sinensis with data available. See also: Homosalate; OCTINOXATE (component of); Avobenzone; homosalate (component of); Homosalate; Zinc oxide (component of) ... View More ... Drug Indication As ingredient in many sunscreen for protection against sunburn, skin aging and skin cancer. Mechanism of Action Homosalate has the ability to convert incident ultraviolet radiation into less damaging infrared radiation (heat). Homosalate is an anti-inflammatory drug commonly used as a chemical UV filter in sunscreens. [2] It displays estrogen-like properties and is suspected to be a potential endocrine disruptor. [2] This study identified a novel, previously unsuspected biological activity of Homosalate: boosting the release of specific subpopulations of tumor-derived extracellular vesicles (EVs) that can promote pro-migratory and anchorage-independent survival phenotypes in recipient cancer cells. [2] |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~100 mg/mL (~381.2 mM; Need ultrasonic) H2O: <0.1 mg/mL (insoluble) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (9.53 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (9.53 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (9.53 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.8117 mL | 19.0585 mL | 38.1170 mL | |
| 5 mM | 0.7623 mL | 3.8117 mL | 7.6234 mL | |
| 10 mM | 0.3812 mL | 1.9059 mL | 3.8117 mL |