Physicochemical Properties
| Molecular Formula | C22H26N2O3 |
| Molecular Weight | 366.4534 |
| Exact Mass | 366.194 |
| CAS # | 35467-43-7 |
| PubChem CID | 3037151 |
| Appearance | Off-white to yellow solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 533.2±50.0 °C at 760 mmHg |
| Flash Point | 276.3±30.1 °C |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.623 |
| LogP | 3.75 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 27 |
| Complexity | 604 |
| Defined Atom Stereocenter Count | 3 |
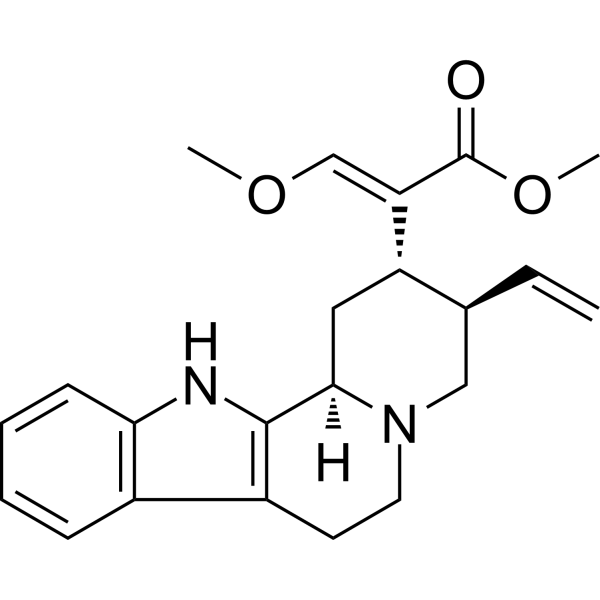
| SMILES | CO/C=C(\[C@H]1C[C@@H]2C3=C(CCN2C[C@@H]1C=C)C4=CC=CC=C4N3)/C(=O)OC |
| InChi Key | TZUGIFAYWNNSAO-AZQGJTAVSA-N |
| InChi Code | InChI=1S/C22H26N2O3/c1-4-14-12-24-10-9-16-15-7-5-6-8-19(15)23-21(16)20(24)11-17(14)18(13-26-2)22(25)27-3/h4-8,13-14,17,20,23H,1,9-12H2,2-3H3/b18-13+/t14-,17-,20+/m0/s1 |
| Chemical Name | methyl (E)-2-[(2S,3R,12bR)-3-ethenyl-1,2,3,4,6,7,12,12b-octahydroindolo[2,3-a]quinolizin-2-yl]-3-methoxyprop-2-enoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Nicotinic acetylcholine receptor (nAChR) (Ki: 1.2 ± 0.3 μM[1] |
| ln Vitro |
1. Inhibition of nicotine-induced dopamine release: Hirsuteine dose-dependently inhibited dopamine release from rat pheochromocytoma cells (PC12 cells) induced by nicotine. At concentrations of 0.1 μM, 1 μM, 5 μM, and 10 μM, Hirsuteine reduced the nicotine (100 μM)-induced dopamine release by 8.2 ± 1.5%, 30.5 ± 3.2%, 58.7 ± 4.1%, and 75.3 ± 5.4%, respectively. The maximum inhibition was reached at 10 μM, with no further increase at higher concentrations (20 μM) [1] 2. Non-competitive antagonism of nAChR: Hirsuteine exhibited non-competitive antagonism against nAChR-mediated dopamine release. Concentration-response curves for nicotine showed that the maximum response (Emax) of dopamine release was reduced from 100% (control) to 24.7 ± 3.6% in the presence of 10 μM Hirsuteine, while the EC50 of nicotine (≈95 μM) remained unchanged. Lineweaver-Burk plot analysis further confirmed non-competitive binding—plots of 1/dopamine release vs. 1/nicotine concentration for different Hirsuteine concentrations intersected on the x-axis [1] 3. No effect on basal dopamine release: Treatment with Hirsuteine alone (0.1–20 μM) for 30 minutes did not alter the basal level of dopamine release from PC12 cells (variation <5% compared to control), indicating it only targets nAChR-activated dopamine release [1] |
| Enzyme Assay |
- Dopamine release detection assay (via high-performance liquid chromatography, HPLC): Rat pheochromocytoma cells were seeded in 24-well plates and cultured until 80% confluence. The cells were washed twice with Krebs-Ringer bicarbonate buffer (KRB buffer, pH 7.4) and pre-incubated in KRB buffer for 30 minutes at 37°C. Hirsuteine solutions (0.1–20 μM) were added to the wells and incubated for 10 minutes, followed by the addition of nicotine (100 μM) to induce dopamine release. After 20 minutes of incubation at 37°C, the culture supernatant was collected and mixed with 0.1 M perchloric acid (1:1, v/v) to stop the reaction and precipitate proteins. The mixture was centrifuged at 12,000 × g for 15 minutes at 4°C, and the supernatant was passed through a 0.22 μm filter. Dopamine concentration was determined using an HPLC system equipped with a reverse-phase C18 column and an electrochemical detector (detection potential: +0.7 V). The mobile phase consisted of 0.1 M sodium acetate buffer (pH 4.5) containing 0.1 mM EDTA and 5% methanol, with a flow rate of 1 mL/min. Dopamine levels were quantified by comparing the peak area of samples with that of standard solutions [1] |
| Cell Assay |
- Rat pheochromocytoma cell (PC12 cell) culture and functional assay: PC12 cells were maintained in Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 10% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin, under a humidified atmosphere of 5% CO₂ at 37°C. For the dopamine release assay, cells were trypsinized and seeded in 24-well plates at a density of 2 × 10⁵ cells per well. After 48 hours of culture, the medium was aspirated, and cells were washed with KRB buffer (containing 118 mM NaCl, 4.7 mM KCl, 2.5 mM CaCl₂, 1.2 mM MgSO₄, 1.2 mM KH₂PO₄, 25 mM NaHCO₃, and 10 mM glucose, pH 7.4) to remove serum components. Cells were pre-incubated in KRB buffer for 30 minutes to stabilize basal dopamine levels, then treated with Hirsuteine (0.1–20 μM) for 10 minutes before adding nicotine (100 μM). After 20 minutes of nicotine stimulation, the supernatant was collected for dopamine detection (as described in Enzyme Assay), and the remaining cells were lysed to determine protein concentration (using BCA assay) for normalization of dopamine release data [1] |
| References |
[1]. Non-competitive antagonism by hirsuteine of nicotinic receptor-mediated dopamine release from rat pheochromocytoma cells. Jpn J Pharmacol. 1993 Apr;61(4):351-6. |
| Additional Infomation |
Hirsuteine is an alkaloid. Hirsuteine has been reported in Uncaria sinensis, Uncaria tomentosa, and other organisms with data available. See also: Cat's Claw (part of). - Hirsuteine is an alkaloid compound that exerts its biological effect by non-competitively antagonizing nicotinic acetylcholine receptors (nAChRs) on rat pheochromocytoma cells. This non-competitive mechanism suggests it binds to an allosteric site on nAChR rather than the orthosteric (nicotine-binding) site, thereby inhibiting receptor activation without competing for nicotine binding [1] - The inhibition of nAChR-mediated dopamine release by Hirsuteine indicates its potential role in regulating cholinergic signaling pathways associated with dopamine neurotransmission, which may be relevant to studies on neurological disorders involving abnormal nAChR activity [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~272.89 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7289 mL | 13.6444 mL | 27.2889 mL | |
| 5 mM | 0.5458 mL | 2.7289 mL | 5.4578 mL | |
| 10 mM | 0.2729 mL | 1.3644 mL | 2.7289 mL |