Physicochemical Properties
| Molecular Formula | C13H18O5 |
| Molecular Weight | 254.27900 |
| Exact Mass | 254.115 |
| CAS # | 1087-26-9 |
| PubChem CID | 9831030 |
| Appearance | Off-white to light yellow solid powder |
| LogP | 2.54 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 18 |
| Complexity | 244 |
| Defined Atom Stereocenter Count | 0 |
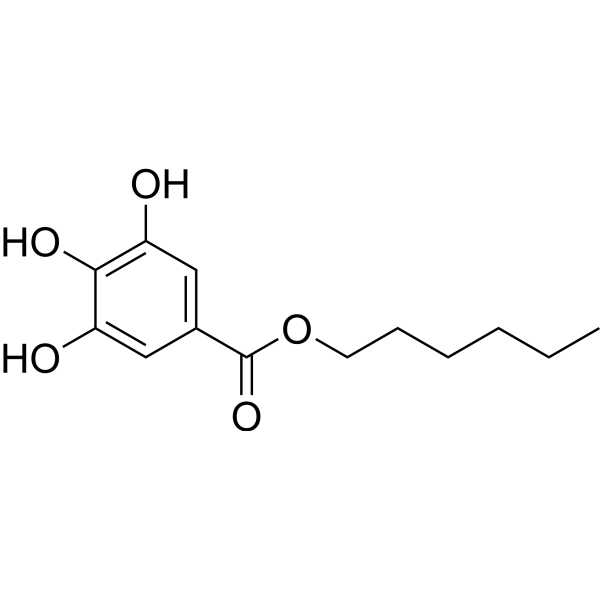
| SMILES | CCCCCCOC(C1=CC(O)=C(O)C(O)=C1)=O |
| InChi Key | DQHJNOHLEKVUHU-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C13H18O5/c1-2-3-4-5-6-18-13(17)9-7-10(14)12(16)11(15)8-9/h7-8,14-16H,2-6H2,1H3 |
| Chemical Name | hexyl 3,4,5-trihydroxybenzoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Hexyl gallate (3,4,5-trihydroxybenzoate) inhibits RhlR, but it has no effect on the Las system-regulated elastase production; instead, it only suppresses Rhl-dependent rhamnolipid and pyocyanin production. and the development of biofilms. At 10–30 μM, hexyl gallate has antimicrobial activity in addition to inhibiting the synthesis of pigment. In CV026 cells, hexyl gallate exhibited antibacterial action as well. Hexyl gallate does not impact the formation of N-(3-oxododecanoyl)-l-homoserine lactone (OdDHL) or 2-hydroxy-4(1H)quinolone (PQS); instead, it solely inhibits the production of N-butyrylhomoserine lactone (BHL) at 100 and 300 μM [1]. Hexyl gallate is an antibacterial substitute for copper compounds that targets the bacterial membrane of Xanthomonas citrus and suppresses the growth of the organism in a dose-response manner, with more substantial activity in the range of 30-50 μg/ml [3]. |
| References |
[1]. Kim B, et al. Differential effects of alkyl gallates on quorum sensing in Pseudomonas aeruginosa. Sci Rep. 2019;9(1):7741. Published 2019 May 23. [2]. Ade Arsianti, et al. Synthesis and in vitro antimalarial activity of alkyl esters of gallate as a growth inhibitor of plasmodium falciparum. Oriental Journal of Chemistry, 34(2), 655-662. [3]. Cavalca LB, et al. Hexyl gallate for the control of citrus canker caused by Xanthomonas citri subsp citri [published online ahead of print, 2020 Aug 6]. Microbiologyopen. 2020;e1104. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~41.67 mg/mL (~163.87 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (8.18 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (8.18 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (8.18 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.9327 mL | 19.6634 mL | 39.3267 mL | |
| 5 mM | 0.7865 mL | 3.9327 mL | 7.8653 mL | |
| 10 mM | 0.3933 mL | 1.9663 mL | 3.9327 mL |