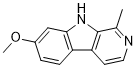
Harmine (telepathine) is a naturally occuring beta-carboline and fluorescent harmala alkaloid found in a number of different plants, most notably the Middle Eastern plant harmal or Syrian rue (Peganum harmala) and the South American vine Banisteriopsis caapi (formerly known as 'yage' or 'ayahuasca'). Harmine is a RIMA because it reversibly inhibits the enzyme monoamine oxidase A (MAO-A), which breaks down monoamines. While it does not inhibit the MAO-B variant, harmine binds to MAO-A specifically. In addition, it is an inhibitor of tyrosine phosphorylation-regulated kinase (DYRK) with anti-inflammatory and anti-cancer properties. With a Ki of 397 nM, harmine exhibits a high affinity for the 5-HT2A serotonin receptor.
Physicochemical Properties
| Molecular Formula | C13H12N2O |
| Molecular Weight | 212.2472 |
| Exact Mass | 212.094 |
| Elemental Analysis | C, 73.56; H, 5.70; N, 13.20; O, 7.54 |
| CAS # | 442-51-3 |
| Related CAS # | Harmine hydrochloride;343-27-1 |
| PubChem CID | 5280953 |
| Appearance | White to yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 421.4±40.0 °C at 760 mmHg |
| Melting Point | 262-264 °C(lit.) |
| Flash Point | 139.8±17.0 °C |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.706 |
| LogP | 3.17 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 16 |
| Complexity | 258 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O(C([H])([H])[H])C1C([H])=C([H])C2=C(C=1[H])N([H])C1C(C([H])([H])[H])=NC([H])=C([H])C2=1 |
| InChi Key | BXNJHAXVSOCGBA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C13H12N2O/c1-8-13-11(5-6-14-8)10-4-3-9(16-2)7-12(10)15-13/h3-7,15H,1-2H3 |
| Chemical Name | 7-methoxy-1-methyl-9H-pyrido[3,4-b]indole |
| Synonyms | Harmine; telepathine; Yageine; 7-Methoxy-1-methyl-9H-pyrido[3,4-b]indole; Banisterine; Telepathine; Leucoharmine; Yajeine; Banisterine; Leucoharmine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | 5-HT2A Receptor (Ki = 397 nM); DYRK1A |
| ln Vitro | Harmine inhibits tau phosphorylation by DYRK1A by specific DANDYs, with an IC50 of 190 nM[2]. Harmine causes extreme cytotoxicity in hepatoma cells by disrupting Rad51 recruitment, which is how it negatively regulates homologous recombination (HR). Moreover, Hep3B cells are significantly more susceptible to the anti-proliferative effects of harmine when exposed to the NHEJ inhibitor Nu7441[3]. |
| ln Vivo | It has been demonstrated that the TBI group has a noticeably higher brain water content. In comparison to the TBI group, treatment with harmine dramatically lowers the tissue water content at 1, 3, and 5 days. When comparing the escape latency at 3 and 5 days to the TBI group, the use of ammine treatment significantly lowers it. When compared to the TBI group that did not receive Harmine treatment, the motor function recovery of the rats is significantly improved by the administration of Harmine post-TBI at 1, 3, and 5 days post-TBI. In comparison with the TBI group, the neuronal survival rate is markedly higher in the group treated with harmine. When Harmine is administered, compared to the TBI group, there is a noticeable increase in GLT-1 expression. In contrast to the TBI group, the administration of harmine dramatically lowers caspase 3 expression[4]. |
| Cell Assay | Experiments on DNA damage, cell death, and proliferation are carried out using the High Content Screening (HCS) format. A multiwell 384 µClear plate coated with 100 µg/mL Poly-L-ornithine and 10 µg/mL laminin is used to plate the hNPCs (1,500 cells/per well). Following a 24-hour period, cells are treated with harmine, INDY, and pargyline in quintuplicate (five wells per condition) for four days in N2B27 medium supplemented with bFGF and EGF. Day 4: 30 minutes before fixation or image acquisition, cells are labeled with 10 µM EdU for cell proliferation or BOBO™ -3 for cell death. |
| Animal Protocol | Rats: The study uses 150 male Sprague-Dawley rats, weighing between 280 and 320 g and aged between 10 and 12 weeks. Three groups of rats are randomly assigned: the TBI group (n=35); the TBI + Harmine-treated group (n=35); and the Sham-operated group (n= 15). Immediately after traumatic brain injury, heroine (i.p., 30 mg/kg daily) is given for a maximum of five days. Equal volumes of 0.9% saline solution are given to the TBI and sham groups (i.p.). For the purpose of examining behavioral recovery, the rats are divided into three groups: Sham (n = 3), TBI (n = 7), and Harmine (n = 7). The NSS is assessed 1, 3, and 5 days after a traumatic brain injury. An observer who is blind to the animal treatment evaluates each rat individually[4]. |
| ADME/Pharmacokinetics |
Metabolism / Metabolites Harmine has known human metabolites that include 6-Hydroxy-harmaline and Harmol. |
| References |
[1]. Binding of beta-carbolines and related agents at serotonin (5-HT(2) and 5-HT(1A)), dopamine (D(2)) and benzodiazepine receptors. Drug Alcohol Depend. 2000 Aug 1;60(2):121-32. [2]. DYRK1A inhibition and cognitive rescue in a Down syndrome mouse model are induced by new fluoro-DANDY derivatives. Sci Rep. 2018 Feb 12;8(1):2859. [3]. Harmine suppresses homologous recombination repair and inhibits proliferation of hepatoma cells. Cancer Biol Ther. 2015;16(11):1585-92. [4]. Treatment with harmine ameliorates functional impairment and neuronal death following traumatic brain injury. Mol Med Rep. 2015 Dec;12(6):7985-91. |
| Additional Infomation |
Harmine is a harmala alkaloid in which the harman skeleton is methoxy-substituted at C-7. It has a role as a metabolite, an anti-HIV agent and an EC 1.4.3.4 (monoamine oxidase) inhibitor. It derives from a hydride of a harman. Harmine has been reported in Passiflora phoenicia, Symplocos setchuensis, and other organisms with data available. Alkaloid isolated from seeds of PEGANUM HARMALA; ZYGOPHYLLACEAE. It is identical to banisterine, or telepathine, from Banisteria caapi and is one of the active ingredients of hallucinogenic drinks made in the western Amazon region from related plants. It has no therapeutic use, but (as banisterine) was hailed as a cure for postencephalitic PARKINSON DISEASE in the 1920's. |
Solubility Data
| Solubility (In Vitro) | DMSO: ≥ 30 mg/mL (~141.3 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (11.78 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (11.78 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.7114 mL | 23.5571 mL | 47.1143 mL | |
| 5 mM | 0.9423 mL | 4.7114 mL | 9.4229 mL | |
| 10 mM | 0.4711 mL | 2.3557 mL | 4.7114 mL |