Physicochemical Properties
| Molecular Formula | C26H30NOF3CL2 |
| Molecular Weight | 500.4237 |
| Exact Mass | 499.166 |
| CAS # | 69756-53-2 |
| Related CAS # | Halofantrine hydrochloride;36167-63-2 |
| PubChem CID | 37393 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.244 g/cm3 |
| Boiling Point | 596.2ºC at 760 mmHg |
| Melting Point | 136-138ºC |
| Flash Point | 314.4ºC |
| Index of Refraction | 1.577 |
| LogP | 8.644 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 33 |
| Complexity | 584 |
| Defined Atom Stereocenter Count | 0 |
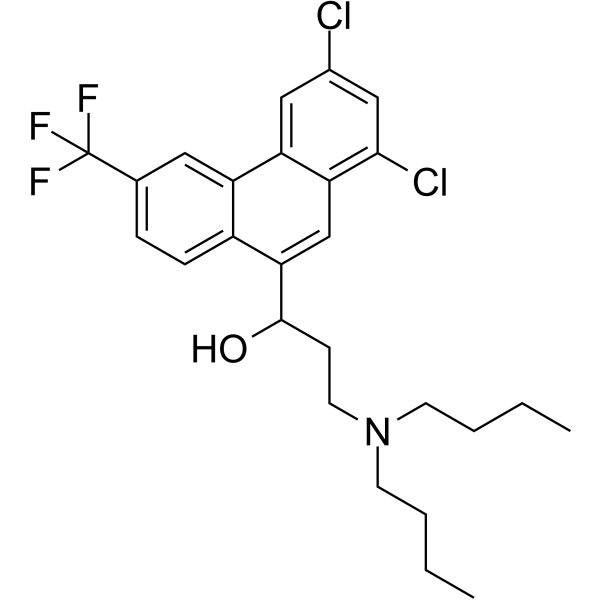
| SMILES | CCCCN(CCCC)CCC(C1=CC2=C(C=C(C=C2Cl)Cl)C3=C1C=CC(=C3)C(F)(F)F)O |
| InChi Key | FOHHNHSLJDZUGQ-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C26H30Cl2F3NO/c1-3-5-10-32(11-6-4-2)12-9-25(33)23-16-22-21(14-18(27)15-24(22)28)20-13-17(26(29,30)31)7-8-19(20)23/h7-8,13-16,25,33H,3-6,9-12H2,1-2H3 |
| Chemical Name | 3-(dibutylamino)-1-[1,3-dichloro-6-(trifluoromethyl)phenanthren-9-yl]propan-1-ol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | At an IC50 of 196.9 nM, halofantrine inhibits the HERG tail current generated after repolarization from +30 mV to 760 mV [2]. At an IC50 of 7.73±0.23 μM, halofantrine suppresses the growth of MDA-MB-231 triple-negative breast cancer (TNBC) cells[3]. With IC50s of 0.0011, 0.0012, and 6.70 μM, respectively, halofantrine shows action against adult gametocytes IV-V, asexual forms (3D7A), and asexual forms (3D7HT-GFP) [4]. |
| Cell Assay |
Cell viability assay [3] Cell Types: MDA-MB-231 TNBC Tested Concentrations: 0, 2.50, 5.00, 10.00, 20.00, 40.00 and 80.00 μM Incubation Duration: 3 days Experimental Results: IC50 is 7.73±0.23 μM. |
| ADME/Pharmacokinetics |
Metabolism / Metabolites Hepatic Biological Half-Life 6-10 days |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Because no information is available on the use of halofantrine during breastfeeding, an alternate drug may be preferred, especially while nursing a newborn or preterm infant. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding 60-70%; |
| References |
[1]. Effect of altered serum lipid concentrations on the IC50 of halofantrine against Plasmodium falciparum. J Pharm Sci. 1998 Feb;87(2):256-8. [2]. Inhibition of HERG potassium channels by the antimalarial agent halofantrine. Br J Pharmacol. 2000 Aug;130(8):1967-75. [3]. CDA: combinatorial drug discovery using transcriptional response modules. PLoS One. 2012;7(8):e42573. [4]. Activity of clinically relevant antimalarial drugs on Plasmodium falciparum mature gametocytes in an ATP bioluminescence "transmission blocking" assay. PLoS One. 2012;7(4):e35019. |
| Additional Infomation |
3-(dibutylamino)-1-[1,3-dichloro-6-(trifluoromethyl)-9-phenanthrenyl]-1-propanol is a member of phenanthrenes. Halofantrine is an antimalarial. It belongs to the phenanthrene class of compounds that includes quinine and lumefantrine. It appears to inhibit polymerisation of heme molecules (by the parasite enzyme "heme polymerase"), resulting in the parasite being poisoned by its own waste. Halofantrine has been shown to preferentially block open and inactivated HERG channels leading to some degree of cardiotoxicity. Halofantrine is an Antimalarial. Drug Indication For treatment of Severe malaria FDA Label Mechanism of Action The mechanism of action of Halofantrine may be similar to that of chloroquine, quinine, and mefloquine; by forming toxic complexes with ferritoporphyrin IX that damage the membrane of the parasite. Pharmacodynamics Halofantrine is a synthetic antimalarial which acts as a blood schizonticide. It is effective against multi drug resistant (including mefloquine resistant) P. falciparum malaria. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9983 mL | 9.9916 mL | 19.9832 mL | |
| 5 mM | 0.3997 mL | 1.9983 mL | 3.9966 mL | |
| 10 mM | 0.1998 mL | 0.9992 mL | 1.9983 mL |