Physicochemical Properties
| Molecular Formula | C32H34F3N5O2 |
| Molecular Weight | 577.65 |
| Exact Mass | 577.266 |
| CAS # | 1315329-43-1 |
| PubChem CID | 53302361 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Index of Refraction | 1.651 |
| LogP | 6.24 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 42 |
| Complexity | 911 |
| Defined Atom Stereocenter Count | 0 |
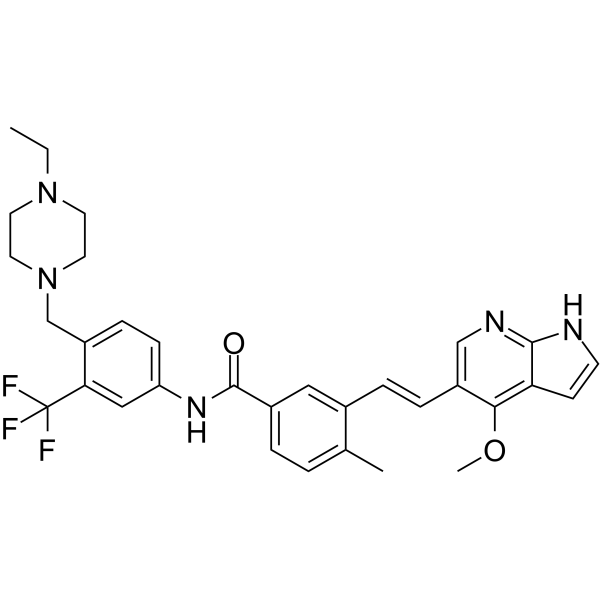
| SMILES | CCN1CCN(CC1)CC2=C(C=C(C=C2)NC(=O)C3=CC(=C(C=C3)C)/C=C/C4=CN=C5C(=C4OC)C=CN5)C(F)(F)F |
| InChi Key | UMBFDGUGXMOMHX-BQYQJAHWSA-N |
| InChi Code | InChI=1S/C32H34F3N5O2/c1-4-39-13-15-40(16-14-39)20-25-9-10-26(18-28(25)32(33,34)35)38-31(41)23-6-5-21(2)22(17-23)7-8-24-19-37-30-27(11-12-36-30)29(24)42-3/h5-12,17-19H,4,13-16,20H2,1-3H3,(H,36,37)(H,38,41)/b8-7+ |
| Chemical Name | N-[4-[(4-ethylpiperazin-1-yl)methyl]-3-(trifluoromethyl)phenyl]-3-[(E)-2-(4-methoxy-1H-pyrrolo[2,3-b]pyridin-5-yl)ethenyl]-4-methylbenzamide |
| Synonyms | HG6641; HG 6 64 1; HG-6-64-1 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References |
[1]. WO 2011090738 A2. |
| Additional Infomation | N-[4-[(4-ethyl-1-piperazinyl)methyl]-3-(trifluoromethyl)phenyl]-3-[2-(4-methoxy-1H-pyrrolo[2,3-b]pyridin-5-yl)ethenyl]-4-methylbenzamide is a member of benzamides. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 100 mg/mL (~173.12 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 3 mg/mL (5.19 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 30.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 3 mg/mL (5.19 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 30.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7312 mL | 8.6558 mL | 17.3115 mL | |
| 5 mM | 0.3462 mL | 1.7312 mL | 3.4623 mL | |
| 10 mM | 0.1731 mL | 0.8656 mL | 1.7312 mL |