Physicochemical Properties
| Molecular Formula | C6H10BRNO2 |
| Molecular Weight | 208.05300 |
| Exact Mass | 206.989 |
| CAS # | 6027-92-5 |
| Related CAS # | Guvacine hydrochloride;6027-91-4;Guvacine;498-96-4 |
| PubChem CID | 15560295 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 1.277 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 10 |
| Complexity | 151 |
| Defined Atom Stereocenter Count | 0 |
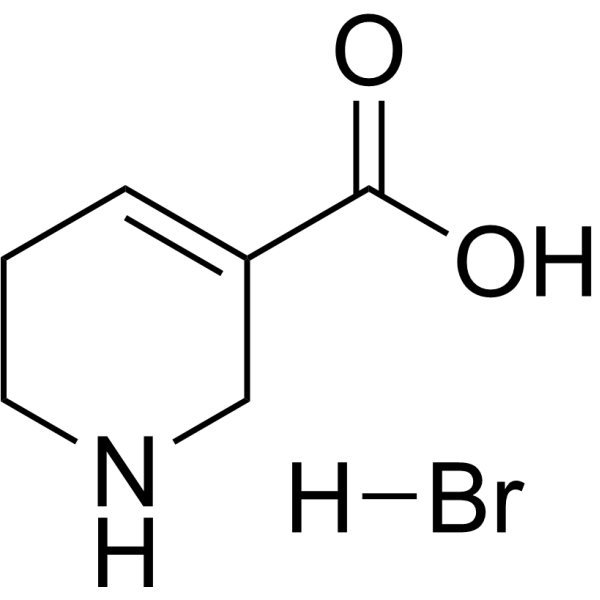
| SMILES | OC(C1CNCCC=1)=O.Br |
| InChi Key | CAQYXXITPFGYGT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H9NO2.BrH/c8-6(9)5-2-1-3-7-4-5;/h2,7H,1,3-4H2,(H,8,9);1H |
| Chemical Name | 1,2,3,6-tetrahydropyridine-5-carboxylic acid;hydrobromide |
| Synonyms | guvacine hydrobromide; Guvacine (hydrobromide); 6027-92-5; SCHEMBL9569008; 1,2,3,6-tetrahydropyridine-5-carboxylic acid;hydrobromide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | human GAT-1 (IC50 = 14 μM); rat GAT-1 (IC50 = 39 μM); rat GAT-2 (IC50 = 58 μM); human GAT-3 (IC50 = 119 μM); rat GAT-3 (IC50 = 378 μM) |
| ln Vitro | Guvacine HCl has IC50s of 14 μM (human GAT-1), 39 μM (rat GAT-1), and 58 μM (rat GAT-2); 119 μM (human GAT-3); 378 μM (rat GAT-3) and 1870 μM (human BGT-3). It is a strong inhibitor of GABA transporters with moderate selectivity for cloned GABA transporters. Guvacine's affinity (IC50 > 1 mM) for hBGT-1 is low [1]. Guvacine hydrochloride is a strong inhibitor of GABA uptake, although it has little or no effect as a GABA receptor agonist and does not prevent sodium-dependent GABA binding [2]. Guvacine inhibits the absorption of GABA and β-alanine, with IC50s in the cat spinal cord of 23 ± 2 μM and 66 ± 11 μM, respectively, and in the rat cerebral cortex of 8 ± 1 μM and 123 ± 28 μM [3]. |
| ln Vivo |
Arecaidine and Guvacine, constituents of the nut of Areca catechu, inhibited the uptake of GABA and beta-alanine, but not that of glycine, by slices of cat spinal cord. In cats anesthetised with pentobarbitone, electrophoretic arecaidine enhanced the inhibitory actions of GABA and beta-alanine, but not those of glycine or taurine, on the firing of spinal neurones. Similarly, electrophoretic guvacine enhanced the inhibition of spinal neurones by GABA but not that by glycine. The uptake of GABA by slices of cat cerebellum was inhibited by arecaidine, and the effect of electrophoretic GABA on the firing of cerebellar Purkinje cells was enhanced by electrophoretic arecaidine. When administered intravenously arecaidine failed to affect synaptic inhibitions considered to be mediated by GABA. Intravenous arecaidine had no effect on either spinal prolonged (presynaptic) inhibition (20mg/kg), dorsal root potentials (20mg/kg) or basket cell inhibition of Purkinje cells (250 mg/kg), although topical arecaidine (6.6-10 x 10(-3) M) blocked this latter inhibition. Large doses of arecaidine (1 g/kg subcutaneous) marginally reduced the lethal effects of bicuculline in mice but appeared to have little or no anticonvulsant activity [3].
Gamma-aminobutyric acid A receptor (GABA(A)R)-mediated postsynaptic currents (IPSCs) were recorded from dopaminergic neurons of the ventral tegmental area of young rats in acute brain slices and from mechanically dissociated neurons. Low concentrations (0.1-0.3 microm) of muscimol, a selective GABA(A)R agonist, increased the amplitude, and reduced the paired pulse ratio of evoked IPSCs. Moreover, muscimol increased the frequency but not the amplitude of spontaneous IPSCs (sIPSCs). These data point to a presynaptic locus of muscimol action. It is interesting that 1 microm muscimol caused an inhibition of sIPSCs, which was reversed to potentiation by the GABA(B) receptor antagonist CGP52432. Isoguvacine, a selective GABA(A)R agonist that belongs to a different class, mimicked the effects of muscimol on sIPSCs: it increased them at low ( |
| Animal Protocol | Most of the chemicals including d(–)-2-amino-5-phospho-pentanoate (AP5), 6,7-dinitroquinoxaline-2,3-dione (DNQX), bicuculline (BIC), cadmium chloride, isoguvacine, furosemide, 4,5,6,7-tetrahydroisoxazolo [5,4-c]pyridine-3-ol (gaboxadol, or THIP), Guvacine hydrochloride, Tyr-D-Ala-Gly-N-Me-Phe-Gly-ol enkephalin (DAMGO) and tetrodotoxin (TTX), Muscimol and CGP52432 were obtained commercially. The solutions, prepared before the experiment, were applied to a dissociated neuron with a Y-tube. This exchanged the external solution surrounding the neurons within 40 ms (Zhou et al. 2006). In experiments on brain slices, chemicals were added in known concentrations to the superfusate. The fact that 10 μm bicuculline blocked most IPSCs (in slices) within 90 s is an indication of the effective bath exchange time. [4] |
| References |
[1]. Tiagabine, SK&F 89976-A, CI-966, and NNC-711 are selective for the cloned GABA transporter GAT-1. Eur J Pharmacol. 1994 Oct 14;269(2):219-24. [2]. Structure-activity studies on the inhibition of GABA binding to rat brain membranes by muscimol and related compounds. J Neurochem. 1978 Jun;30(6):1377-82. [3]. Effects of the Areca nut constituents arecaidine and guvacine on the action of GABA in the cat central nervous system. Brain Res. 1977 Nov 18;136(3):513-22. [4]. Presynaptic GABAA receptors facilitate GABAergic transmission to dopaminergic neurons in the ventral tegmental area of young rats. J Physiol. 2007 May 1;580(Pt.3):731-43. |
| Additional Infomation | The application of Guvacine (20 μm), a GABA transporter blocker, induced a robust and reversible increase in the frequency of sIPSCs recorded in slices (Fig. 7A and C) without inducing a significant postsynaptic current (5 ± 1 pA). Guvacine (20 μm) induced a large leftward shift in the cumulative probability plot of inter-event intervals between successive sIPSCs (K-S test, P < 0.001; upper panel in Fig. 7B) and an increase of 72 ± 14% (n = 7, P = 0.001, upper panel inset in Fig. 7B) in sIPSC frequency, while it did not alter sIPSC amplitude (K-S test, P > 0.5, lower panel in Fig. 7B; 0 ± 3%, n = 7, P = 0.46, lower panel inset in Fig. 7B). These results suggest that an increase in ambient GABA level enhances GABAergic sIPSCs via a presynaptic mechanism in the developing brain.[4] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.8065 mL | 24.0327 mL | 48.0654 mL | |
| 5 mM | 0.9613 mL | 4.8065 mL | 9.6131 mL | |
| 10 mM | 0.4807 mL | 2.4033 mL | 4.8065 mL |