Physicochemical Properties
| Molecular Formula | C60H92N12O10 |
| Molecular Weight | 1141.44688 |
| Exact Mass | 1140.705 |
| Elemental Analysis | C, 56.13; H, 6.92; F, 8.32; N, 12.27; O, 16.36 |
| CAS # | 113-73-5 |
| Related CAS # | 113-73-5; |
| PubChem CID | 73357 |
| Appearance | Solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 1394.8±65.0 °C at 760 mmHg |
| Flash Point | 797.4±34.3 °C |
| Vapour Pressure | 0.0±0.3 mmHg at 25°C |
| Index of Refraction | 1.596 |
| LogP | 0.1 |
| Hydrogen Bond Donor Count | 10 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 16 |
| Heavy Atom Count | 82 |
| Complexity | 2020 |
| Defined Atom Stereocenter Count | 10 |
| SMILES | NCCC[C@@H]1NC(=O)[C@H](C(C)C)NC(=O)[C@@H]2CCCN2C(=O)[C@@H](CC2C=CC=CC=2)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN)NC(=O)[C@H](C(C)C)NC(=O)[C@@H]2CCCN2C(=O)[C@@H](CC2C=CC=CC=2)NC(=O)[C@H](CC(C)C)NC1=O |
| InChi Key | IUAYMJGZBVDSGL-XNNAEKOYSA-N |
| InChi Code | InChI=1S/C60H92N12O10/c1-35(2)31-43-53(75)67-45(33-39-19-11-9-12-20-39)59(81)71-29-17-25-47(71)55(77)70-50(38(7)8)58(80)64-42(24-16-28-62)52(74)66-44(32-36(3)4)54(76)68-46(34-40-21-13-10-14-22-40)60(82)72-30-18-26-48(72)56(78)69-49(37(5)6)57(79)63-41(23-15-27-61)51(73)65-43/h9-14,19-22,35-38,41-50H,15-18,23-34,61-62H2,1-8H3,(H,63,79)(H,64,80)(H,65,73)(H,66,74)(H,67,75)(H,68,76)(H,69,78)(H,70,77)/t41-,42-,43-,44-,45+,46+,47-,48-,49-,50-/m0/s1 |
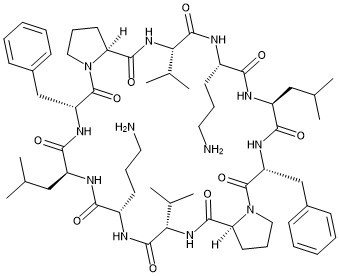
| Chemical Name | (3R,6S,9S,12S,15S,21R,24S,27S,30S,33S)-9,27-bis(3-aminopropyl)-3,21-dibenzyl-6,24-bis(2-methylpropyl)-12,30-di(propan-2-yl)-1,4,7,10,13,19,22,25,28,31-decazatricyclo[31.3.0.015,19]hexatriacontane-2,5,8,11,14,20,23,26,29,32-decone |
| Synonyms | Gramicidin S; Gramicidin S TFA salt; Gramicidin S 1; Gramicidin S-1; Gramicidin S1; Gramicidin S-A; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | cyclic peptide antibiotic; bacterial membranes |
| ln Vitro | The COVID19 pandemic has led to multipronged approaches for treatment of the disease. Since de novo discovery of drugs is time consuming, repurposing of molecules is now considered as one of the alternative strategies to treat COVID19. Antibacterial peptides are being recognized as attractive candidates for repurposing to treat viral infections. In this study, we describe the anti-SARS-CoV-2 activity of the well-studied antibacterial peptides gramicidin S and melittin obtained from Bacillus brevis and bee venom respectively. The EC50 values for gramicidin S and melittin were 1.571 µg and 0.656 µg respectively based on in vitro antiviral assay. Significant decrease in the viral load as compared to the untreated group with no/very less cytotoxicity was observed. Both the peptides treated to the SARS-CoV-2 infected Vero cells showed viral clearance from 12 h onwards with a maximal viral clearance after 24 h post infection. Proteomics analysis indicated that more than 250 proteins were differentially regulated in the gramicidin S and melittin treated SARS-CoV-2 infected Vero cells against control SARS-CoV-2 infected Vero cells after 24 and 48 h post infection. The identified proteins were found to be associated in the metabolic and mRNA processing of the Vero cells post-treatment and infection. Both these peptides could be attractive candidates for repurposing to treat SARS-CoV-2 infection. [1] |
| Cell Assay |
Cell viability using MTT assay [1] The Vero cells were plated in 96 well culture plate and incubated at 37 °C with 5% CO2. After attaining 90–95% cell confluency, different concentrations of gramicidin S and melittin (0.5, 0.7, 3, 5 µg for both) were added to the cells to check the effect of the peptides on the cells for 24 h. After 24 h, 100 µl (50 µg) of MTT substrate was added to the cells and the plate was continued to incubate for 3 h at 37 °C with 5% CO2. Later the formazan crystals formed were dissolved in 100 µl of DMSO and the absorbance was measured at 570 nm in Multimode Micro plate reader. RT-qPCR assay [1] The effect of gramicidin S and melittin was tested against the SARS-CoV-2 with different concentrations. Remdesivir was run as an assay control. The titers for the virus were adjusted such that there was only viral replication and no cytolysis. Briefly, the virus (MOI 0.1) was pre-incubated with different concentrations of gramicidin S and melittin (0.1–10 µg) for an hour at 37 °C. After the incubation, virus inoculum containing gramicidin S and melittin was added to the Vero cells in duplicates (50 µl/well). Remdesivir (1 µM) was added to the Vero cells without pre-incubation as in the case of peptides. All the experimental groups were left for infection for 3 h while maintaining at 37 °C with 5% CO2. Post-infection (PI), media containing viral inoculum and the gramicidin S and melittin was removed and replaced with 200 µl of fresh DMEM media containing 10% FBS and the experimental groups were maintained for varying time points in an incubator maintained at 37 °C with 5% CO2. Post-incubation, cell supernatants from the experimental groups were collected and spun for 10 min at 6000 g to remove debris and the supernatant was transferred to fresh collection tubes and later were processed to isolate viral RNA. RNA was extracted from 200 μL aliquots of sample supernatant using the MagMAX™ Viral/Pathogen Extraction Kit. Briefly, the viral supernatants from the test groups were added into the deep well plate along with a lysis buffer containing the following components—260 μL, MagMAX™ Viral/Pathogen Binding Solution; 10 μL, MVP-II Binding Beads; 5 μL, MagMAXTMViral/Pathogen Proteinase-K, for 200 μL of sample. (Extraction was performed using KingFisher Flex according to manufactures instructions). The eluted RNA was immediately stored in – 80 °C until further use. |
| Toxicity/Toxicokinetics |
73357 rat LD50 intraperitoneal 17 mg/kg American Review of Soviet Medicine., 2(134), 1944 73357 rat LD50 intravenous 17 mg/kg CRC Handbook of Antibiotic Compounds, Vols.1- , Berdy, J., Boca Raton, FL, CRC Press, 1980, 4(1)(271), 1980 73357 mouse LD50 intravenous 40 mg/kg Antibiotics: Origin, Nature, and Properties, Korzyoski, T., et al., eds., Washington, DC, American Soc. for Microbiology, 1978, 3(1529), 1978 |
| References | [1]. Gramicidin S and melittin: potential anti-viral therapeutic peptides to treat SARS-CoV-2 infection. Sci Rep. 2022 Mar 2;12(1):3446. |
| Additional Infomation |
Gramicidin S is a homodetic cyclic peptide. A group of peptide antibiotics from BACILLUS brevis. Gramicidin C or S is a cyclic, ten-amino acid polypeptide and gramicidins A, B, D are linear. Gramicidin is one of the two principal components of TYROTHRICIN. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.8761 mL | 4.3804 mL | 8.7608 mL | |
| 5 mM | 0.1752 mL | 0.8761 mL | 1.7522 mL | |
| 10 mM | 0.0876 mL | 0.4380 mL | 0.8761 mL |