Physicochemical Properties
| Molecular Formula | C26H42NNAO5 |
| Molecular Weight | 471.6052 |
| Exact Mass | 449.314 |
| CAS # | 16409-34-0 |
| Related CAS # | Glycodeoxycholic Acid;360-65-6;Glycodeoxycholic acid monohydrate;1079043-81-4 |
| PubChem CID | 3035026 |
| Appearance | White to off-white solid powder |
| Density | 0.9330g/cm3 |
| Boiling Point | 655.6ºC at 760 mmHg |
| Melting Point | 207 °C |
| Flash Point | 350.3ºC |
| Vapour Pressure | 5.74E-20mmHg at 25°C |
| LogP | 2.65 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 32 |
| Complexity | 727 |
| Defined Atom Stereocenter Count | 10 |
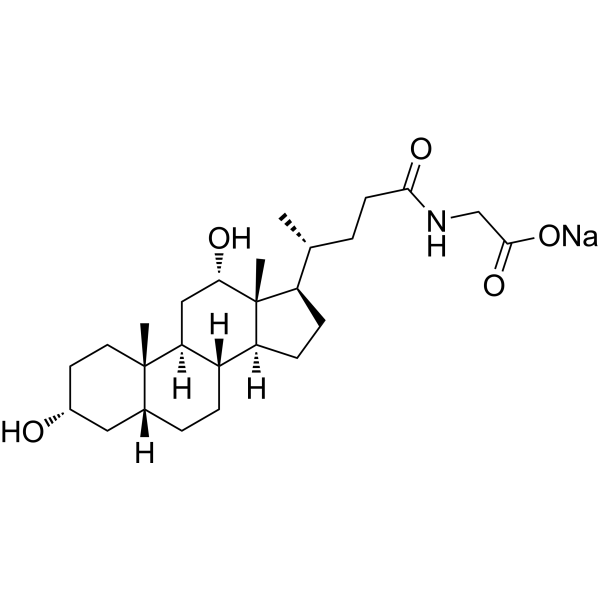
| SMILES | C[C@H](CCC(=O)NCC(=O)O)[C@H]1CC[C@@H]2[C@@]1([C@H](C[C@H]3[C@H]2CC[C@H]4[C@@]3(CC[C@H](C4)O)C)O)C |
| InChi Key | WVULKSPCQVQLCU-BUXLTGKBSA-N |
| InChi Code | InChI=1S/C26H43NO5/c1-15(4-9-23(30)27-14-24(31)32)19-7-8-20-18-6-5-16-12-17(28)10-11-25(16,2)21(18)13-22(29)26(19,20)3/h15-22,28-29H,4-14H2,1-3H3,(H,27,30)(H,31,32)/t15-,16-,17-,18+,19-,20+,21+,22+,25+,26-/m1/s1 |
| Chemical Name | 2-[[(4R)-4-[(3R,5R,8R,9S,10S,12S,13R,14S,17R)-3,12-dihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl]pentanoyl]amino]acetic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro |
In Caco-2 intestinal epithelial cells, Glycodeoxycholate Sodium exhibited an IC50 of 2.0 mM in the MTS cytotoxicity assay after 120 min exposure. It reduced transepithelial electrical resistance (TEER) and increased the apparent permeability coefficient (Papp) of [¹⁴C]-mannitol across Caco-2 monolayers at 0.5 mM. At 10 mM, it increased the Papp of FITC-dextran 4000 (FD4) and FITC-LKP across isolated rat colonic mucosae. In the buccal epithelial cell line TR146, the IC50 was 1.9 mM in the MTS assay. It also increased the Papp of FD4 across TR146 multilayers at 1.5 mM and induced actin rearrangement. High-content analysis (HCA) in Caco-2 and TR146 cells showed that GDC altered nuclear area, nuclear intensity, mitochondrial membrane potential, and plasma membrane potential at concentrations ≥1.5 mM. [1] |
| Cell Assay |
Cytotoxicity Assays: Caco-2 and TR146 cells were seeded in 96-well plates and exposed to GDC for 120 min. Viability was assessed using MTS, CellTox Green (membrane integrity), Caspase-Glo 3/7 (apoptosis), ROS-Glo H₂O₂ (reactive oxygen species), JC-1 (mitochondrial membrane potential), Laurdan (membrane fluidity), and RealTime-Glo Annexin V (apoptosis/necrosis) assays. High-content analysis (HCA) was performed using dyes for nuclei (Hoechst 33342), intracellular calcium (Fluo-4 AM), mitochondria (TMRM), and dead cells (TOTO-3 iodide). [1] Permeability Assay: Caco-2 monolayers or TR146 multilayers grown on Transwell filters were used. TEER was measured before and after apical addition of GDC. The Papp of markers ([¹⁴C]-mannitol, FD4, FITC-LKP, [³H]-octreotide) was calculated from basolateral samples collected over 120 min. [1] Immunocytochemistry: TR146 cells grown on chamber slides were exposed to GDC, fixed, permeabilized, stained with Alexa Fluor 488 Phalloidin (F-actin) and Hoechst (nuclei), and imaged by confocal microscopy. [1] |
| Animal Protocol |
Rat Colonic Mucosae: Male Wistar rats (250–300 g) were euthanized, and colonic tissue was dissected. The smooth muscle layer was removed, and mucosae were mounted in Ussing chambers. Tissues were bathed in Krebs-Henseleit buffer at 37°C. GDC was added apically, and TEER and Papp of markers were monitored over 120 min. [1] Porcine Buccal Mucosae: Buccal tissue from pigs was collected post-mortem, trimmed, and the epithelial layer was prepared using a dermatome. Tissues were mounted in Ussing chambers, equilibrated, and treated apically with GDC. TEER and Papp of markers were measured over 3 h. [1] |
| Toxicity/Toxicokinetics |
In Caco-2 cells, GDC showed low cytotoxicity at concentrations ≤2 mM. In isolated rat colonic mucosae, 10 mM GDC did not cause macroscopic damage in histology. In porcine buccal mucosae, concentrations up to 10 mM did not induce histological changes. GDC primarily induced necrotic rather than apoptotic cell death in TR146 cells at concentrations >0.5 mM. [1] |
| References |
[1]. Sodium glycodeoxycholate and sodium deoxycholate as epithelial permeation enhancers: in vitro and ex vivo intestinal and buccal bioassays. Eur J Pharm Sci. 2021 Apr 1;159:105737. [2]. Effects of bile salts on permeability and morphology of main pancreatic duct in cats. Dig Dis Sci. 1984 Aug;29(8):740-51. [3]. Bioadhesive starch microspheres and absorption enhancing agents act synergistically to enhance the nasal absorption of polypeptides. Int J Pharm. 2001 Jul 3;222(1):109-19. |
| Additional Infomation |
Glycodeoxycholic acid is a bile acid glycine conjugate of deoxycholic acid. It has a role as a human metabolite. It is functionally related to a deoxycholic acid. It is a conjugate acid of a glycodeoxycholate. Glycodeoxycholic acid has been reported in Streptomyces nigra, Trypanosoma brucei, and Caenorhabditis elegans with data available. A bile salt formed in the liver by conjugation of deoxycholate with glycine, usually as the sodium salt. It acts as a detergent to solubilize fats for absorption and is itself absorbed. It is used as a cholagogue and choleretic. Glycodeoxycholate Sodium is a secondary bile salt with amphipathic surfactant properties. It functions as a permeation enhancer by increasing plasma membrane fluidity, transiently opening tight junctions, and altering actin cytoskeleton organization. It is being explored for oral and buccal delivery of macromolecules (e.g., peptides) and is a component in some clinical-stage oral peptide formulations. Its critical micelle concentration (CMC) ranges from 2–6 mM. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 150 mg/mL (~318.06 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.30 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.30 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.30 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1204 mL | 10.6020 mL | 21.2040 mL | |
| 5 mM | 0.4241 mL | 2.1204 mL | 4.2408 mL | |
| 10 mM | 0.2120 mL | 1.0602 mL | 2.1204 mL |