Physicochemical Properties
| Molecular Formula | C20H20N2O2F4 |
| Molecular Weight | 396.3786 |
| Exact Mass | 396.146 |
| CAS # | 1245526-82-2 |
| PubChem CID | 46937290 |
| Appearance | White to yellow solid powder |
| LogP | 4.611 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 28 |
| Complexity | 545 |
| Defined Atom Stereocenter Count | 1 |
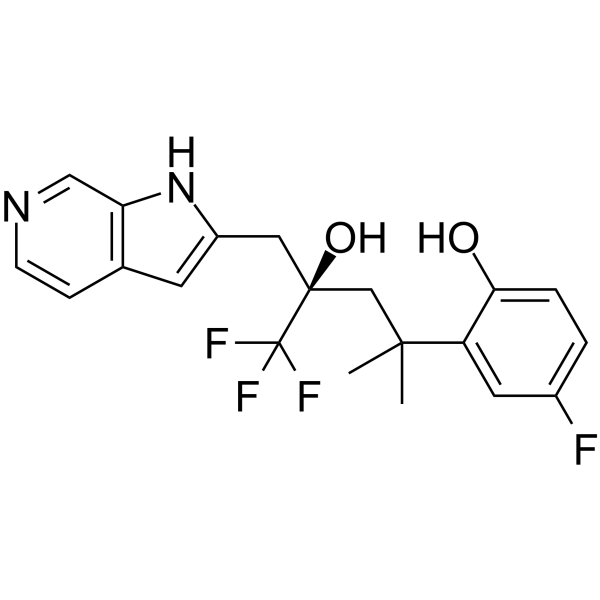
| SMILES | CC(C)(C[C@@](CC1=CC2=C(N1)C=NC=C2)(C(F)(F)F)O)C3=C(C=CC(=C3)F)O |
| InChi Key | JFUAWXPBHXKZGA-IBGZPJMESA-N |
| InChi Code | InChI=1S/C20H20F4N2O2/c1-18(2,15-8-13(21)3-4-17(15)27)11-19(28,20(22,23)24)9-14-7-12-5-6-25-10-16(12)26-14/h3-8,10,26-28H,9,11H2,1-2H3/t19-/m0/s1 |
| Chemical Name | 4-fluoro-2-[(4R)-5,5,5-trifluoro-4-hydroxy-2-methyl-4-(1H-pyrrolo[2,3-c]pyridin-2-ylmethyl)pentan-2-yl]phenol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | The glucocorticoid receptor agonist ((R)-16) affects aromatase with an EC50 value of 11 nM as well as MMTV and IL-6 with IC50 values of 3.3 and 80 nM, respectively [1]. |
| ln Vivo | In Sprague-Dawley rats, the glucocorticoid receptor agonist ((R)-16) demonstrates advantageous pharmacokinetic characteristics [1]. Glucocorticoid receptor agonist ((R)-16) (30 and 10 mg/kg, oral, daily, 5 weeks) significantly inhibits the production of TNF- Mouse model of α and lowers elevated levels of body fat and serum insulin [1]. Sprague-Dawley rats were given 5 mg/kg iv or 30 mg/kg po of the glucocorticoid receptor agonist ((R)-16) according to the pharmacokinetic parameters [1]. AUCinf (po) (h·ng/mL) F(po)% (R)-16 49 7.6 1.85 509 4879 48 Parameter Cl (iv) (mL/min/kg) VSS (iv) (L/kg) T1/2 (iv) (h) Cmax (po) (ng/mL) |
| References |
[1]. Nonsteroidal dissociated glucocorticoid agonists containing azaindoles as steroid A-ring mimetics. J Med Chem. 2010 Sep 23;53(18):6681-98. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 100 mg/mL (~252.28 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.31 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.31 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.31 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5228 mL | 12.6142 mL | 25.2283 mL | |
| 5 mM | 0.5046 mL | 2.5228 mL | 5.0457 mL | |
| 10 mM | 0.2523 mL | 1.2614 mL | 2.5228 mL |