Physicochemical Properties
| Molecular Formula | C36H60O8 |
| Molecular Weight | 620.8568 |
| Exact Mass | 620.428 |
| CAS # | 364779-15-7 |
| PubChem CID | 75412555 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 722.4±60.0 °C at 760 mmHg |
| Flash Point | 390.7±32.9 °C |
| Vapour Pressure | 0.0±5.3 mmHg at 25°C |
| Index of Refraction | 1.576 |
| LogP | 5.11 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 44 |
| Complexity | 1100 |
| Defined Atom Stereocenter Count | 15 |
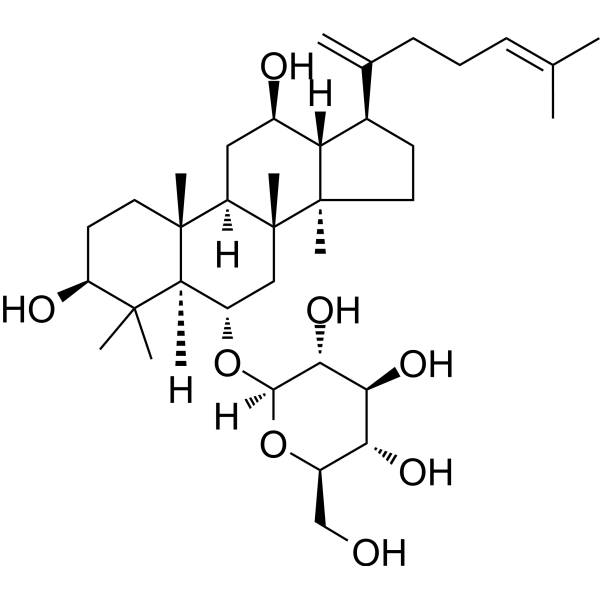
| SMILES | CC(=CCCC(=C)[C@H]1CC[C@@]2([C@@H]1[C@@H](C[C@H]3[C@]2(C[C@@H]([C@@H]4[C@@]3(CC[C@@H](C4(C)C)O)C)O[C@H]5[C@@H]([C@H]([C@@H]([C@H](O5)CO)O)O)O)C)O)C)C |
| InChi Key | AVXFIVJSCUOFNT-QXPABTKOSA-N |
| InChi Code | InChI=1S/C36H60O8/c1-19(2)10-9-11-20(3)21-12-15-35(7)27(21)22(38)16-25-34(6)14-13-26(39)33(4,5)31(34)23(17-36(25,35)8)43-32-30(42)29(41)28(40)24(18-37)44-32/h10,21-32,37-42H,3,9,11-18H2,1-2,4-8H3/t21-,22-,23+,24-,25-,26+,27+,28-,29+,30-,31+,32-,34-,35-,36-/m1/s1 |
| Chemical Name | (2R,3R,4S,5S,6R)-2-[[(3S,5R,6S,8R,9R,10R,12R,13R,14R,17S)-3,12-dihydroxy-4,4,8,10,14-pentamethyl-17-(6-methylhepta-1,5-dien-2-yl)-2,3,5,6,7,9,11,12,13,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-6-yl]oxy]-6-(hydroxymethyl)oxane-3,4,5-triol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Non-small cell lung cancer (NSCLC) cells (A549, H1299): Ginsenoside Rk3 exhibits antiproliferative activity with IC₅₀ values of 18.2 μM (A549 cells, 48 h) and 21.5 μM (H1299 cells, 48 h) [2] - NF-κB signaling pathway: Ginsenoside Rk3 inhibits NF-κB p65 nuclear translocation and transcriptional activity [2] - PI3K/Akt signaling pathway: Ginsenoside Rk3 reduces phosphorylation of PI3K (p-PI3K) and Akt (p-Akt) [2] - Apoptosis-related proteins: Ginsenoside Rk3 activates caspase-3, -9 and upregulates Bax, while downregulating Bcl-2 [2] |
| ln Vitro |
In a dose-dependent manner, ginsenoside Rk3 exhibits substantial inhibitory effect against NF-κB. HepG2 cells are first pre-treated for 1 hour at doses ranging from 0.01 to 10 μM with various ginsenosides, and then for 20 hours with TNF-α. Ginsenoside Rk3 has an IC50 of 14.24±1.30 μM and strongly suppresses TNF-α-induced NF-κB transcriptional activity. According to the data from HepG2 cells, ginsenoside Rk3 strongly suppresses TNF-α-induced NF-κB transcriptional activity in SK-Hep1 cells, with an IC50 of 15.32±0.29 μM. In line with the suppression of NF-κB, ginsenoside Rk3 substantially and dose-dependently suppresses the upregulation of IL8, CXCL1, iNOS, and ICAM1 mRNA[1]. 1. Antiproliferative activity in NSCLC cells: - Ginsenoside Rk3 (5-30 μM) inhibited proliferation of A549 (human lung adenocarcinoma) and H1299 (human lung squamous cell carcinoma) cells in a dose- and time-dependent manner [2] - MTT assay results: 10 μM Ginsenoside Rk3 reduced A549 cell viability to 68.5% (24 h) and 42.3% (48 h); 20 μM reduced viability to 31.2% (48 h) [2] - Colony formation assay: 15 μM Ginsenoside Rk3 decreased A549 colony number by 75% (vs. control) after 14 days of culture [2] 2. Apoptosis induction: - Annexin V-FITC/PI staining: 20 μM Ginsenoside Rk3 treatment for 48 h increased A549 apoptotic rate from 3.2% (control) to 62.3% (early apoptosis: 38.5%, late apoptosis: 23.8%) [2] - Western blot analysis: Ginsenoside Rk3 (10-25 μM) dose-dependently increased cleaved caspase-3 (2.8-fold at 20 μM) and cleaved caspase-9 (3.1-fold at 20 μM) levels, upregulated Bax protein (2.5-fold at 20 μM), and downregulated Bcl-2 protein (0.3-fold at 20 μM) in A549 cells [2] 3. Cell cycle arrest: - PI staining and flow cytometry: 20 μM Ginsenoside Rk3 treatment for 24 h induced G2/M phase arrest in A549 cells; G2/M phase cell proportion increased from 12.1% (control) to 38.5% [2] - This arrest was associated with reduced cyclin B1 and cdc2 (cell cycle regulatory proteins) levels (0.4-fold and 0.35-fold of control, respectively, at 20 μM) [2] 4. Signaling pathway inhibition: - Ginsenoside Rk3 (15-25 μM) inhibited TNF-α-induced NF-κB p65 nuclear translocation in A549 cells (nuclear p65 levels reduced to 0.25-fold of control at 20 μM) [2] - Reduced phosphorylation of PI3K (p-PI3K: 0.3-fold) and Akt (p-Akt: 0.28-fold) in A549 cells treated with 20 μM Ginsenoside Rk3 for 24 h [2] |
| ln Vivo |
Using an H460 xenograft model in nude mice, the inhibitory effects of ginsenoside Rk3 (Rk3) on tumor growth are investigated in vivo. The group treated with ginsenoside Rk3 shows a notable reduction in tumor development (volume) as compared to the control group. Twenty-one days after the start of treatment, the mice given 20 mg/kg Ginsenoside Rk3 showed a considerable inhibition of tumor growth of about 62.99%, which was comparable to the inhibitory impact seen in the group given 20 mg/kg Gefitinib (57.21%). Tumor growth is moderately decreased in the control group in mice given 10 and 5 mg/kg ginsenoside Rk3, with respective inhibition rates of 32.54% and 11.84%[2]. 1. Antitumor efficacy in nude mouse A549 xenograft model: - Animals: Female BALB/c nude mice (4-6 weeks old, 18-22 g), housed under specific pathogen-free (SPF) conditions [2] - Tumor establishment: A549 cells (1×10⁷ cells/mouse) were subcutaneously injected into the right dorsal flank of mice; treatment started when tumors reached ~100 mm³ [2] - Treatment groups: Control (vehicle: 0.1% DMSO in saline), Ginsenoside Rk3 10 mg/kg (i.p.), Ginsenoside Rk3 20 mg/kg (i.p.); injections given every 2 days for 21 days [2] - Tumor growth inhibition: 20 mg/kg Ginsenoside Rk3 significantly reduced tumor volume (from 100 mm³ to 285 mm³ vs. control’s 902 mm³ at day 21) and tumor weight (0.28 g vs. control’s 1.01 g), with an inhibition rate of 72.3% [2] - In vivo apoptosis: Immunohistochemistry (IHC) of tumor tissues showed increased cleaved caspase-3-positive cells (35.2% at 20 mg/kg vs. 4.1% in control) and decreased Ki-67 (proliferation marker) positive cells (18.5% at 20 mg/kg vs. 62.3% in control) [2] - No obvious toxicity: Mice in Rk3 groups showed no significant weight loss (20 mg/kg group: ±3.2% weight change vs. control) and normal serum ALT, AST, BUN, and creatinine levels [2] |
| Enzyme Assay |
1. NF-κB transcriptional activity assay (dual-luciferase reporter gene assay): - A549 cells were co-transfected with NF-κB luciferase reporter plasmid and Renilla luciferase plasmid (internal control) using transfection reagent [2] - After 24 h transfection, cells were pretreated with Ginsenoside Rk3 (10-25 μM) for 2 h, then stimulated with TNF-α (10 ng/ml) for 6 h [2] - Cells were lysed, and luciferase activity was measured using a dual-luciferase assay kit; NF-κB activity was calculated as the ratio of firefly luciferase to Renilla luciferase activity [2] - Result: 20 μM Ginsenoside Rk3 inhibited TNF-α-induced NF-κB activity by 68% (vs. TNF-α alone group) [2] 2. Caspase-3/9 activity assay: - A549 cells were treated with Ginsenoside Rk3 (10-25 μM) for 24 h, then lysed in caspase lysis buffer [2] - Lysates (50 μg protein) were mixed with caspase-3 substrate (Ac-DEVD-pNA) or caspase-9 substrate (Ac-LEHD-pNA) in reaction buffer, incubated at 37°C for 2 h [2] - Caspase activity was measured by monitoring absorbance at 405 nm; activity was expressed as fold change vs. control [2] - Result: 20 μM Ginsenoside Rk3 increased caspase-3 activity by 4.2-fold and caspase-9 activity by 3.8-fold [2] |
| Cell Assay |
1. MTT-based antiproliferation assay: - A549/H1299 cells were seeded in 96-well plates (5×10³ cells/well) and cultured in RPMI-1640 medium with 10% FBS at 37°C, 5% CO₂ [2] - Cells were treated with Ginsenoside Rk3 (5-30 μM) for 24/48/72 h; control group received 0.1% DMSO [2] - MTT solution (5 mg/ml) was added (20 μl/well) and incubated for 4 h; formazan crystals were dissolved in DMSO, and absorbance was measured at 570 nm [2] - Cell viability (%) = (Absorbance of treated group / Absorbance of control group) × 100; IC₅₀ values were calculated using dose-response curves [2] 2. Annexin V-FITC/PI apoptosis assay: - A549 cells (1×10⁶ cells/ml) were treated with Ginsenoside Rk3 (10-25 μM) for 48 h, then harvested and washed with cold PBS [2] - Cells were resuspended in binding buffer, stained with Annexin V-FITC (5 μl) and PI (5 μl) for 15 min in the dark [2] - Stained cells were analyzed by flow cytometry; apoptotic cells were gated as Annexin V-positive/PI-negative (early) or Annexin V-positive/PI-positive (late) [2] 3. Cell cycle analysis: - A549 cells treated with Ginsenoside Rk3 (20 μM) for 24 h were fixed with 70% ethanol at -20°C overnight [2] - Fixed cells were washed with PBS, treated with RNase A (100 μg/ml) for 30 min at 37°C, then stained with PI (50 μg/ml) for 15 min [2] - Cell cycle distribution (G0/G1, S, G2/M phases) was analyzed by flow cytometry; data were processed using cell cycle analysis software [2] 4. Western blot analysis: - A549 cells treated with Ginsenoside Rk3 (10-25 μM) were lysed in RIPA buffer with protease/phosphatase inhibitors [2] - Protein samples (30 μg/lane) were separated by SDS-PAGE, transferred to PVDF membranes, and blocked with 5% non-fat milk for 1 h [2] - Membranes were incubated with primary antibodies (anti-caspase-3, anti-caspase-9, anti-Bax, anti-Bcl-2, anti-NF-κB p65, anti-p-PI3K, anti-p-Akt, anti-β-actin) at 4°C overnight, then with secondary antibody for 1 h at room temperature [2] - Bands were visualized with ECL reagent; band intensity was quantified using densitometry software, normalized to β-actin [2] |
| Animal Protocol |
1. Nude mouse A549 xenograft experiment: - Animal housing: Female BALB/c nude mice (4-6 weeks old) were maintained in SPF conditions (22±2°C, 50±5% humidity, 12 h light/dark cycle) with ad libitum food and water [2] - Tumor inoculation: A549 cells were harvested in log phase, resuspended in PBS (1×10⁷ cells/100 μl), and subcutaneously injected into the right dorsal flank of each mouse [2] - Grouping and treatment: When tumors reached ~100 mm³, mice were randomly divided into 3 groups (n=6/group): - Control group: i.p. injection of vehicle (0.1% DMSO in saline) every 2 days [2] - Low-dose Rk3 group: i.p. injection of Ginsenoside Rk3 10 mg/kg every 2 days [2] - High-dose Rk3 group: i.p. injection of Ginsenoside Rk3 20 mg/kg every 2 days [2] - Sample collection and analysis: Tumor volume was measured every 3 days (volume = length × width² / 2); after 21 days, mice were euthanized, tumors were excised and weighed [2] - Tumor tissues were fixed in 4% paraformaldehyde for IHC staining (cleaved caspase-3, Ki-67); serum was collected to measure ALT, AST, BUN, and creatinine levels [2] |
| Toxicity/Toxicokinetics |
1. In vitro cytotoxicity on normal cells: - Ginsenoside Rk3 (up to 30 μM) showed low cytotoxicity on normal human bronchial epithelial cells (BEAS-2B); cell viability remained >85% after 48 h treatment (vs. A549 cell viability of 31.2% at 20 μM) [2] 2. In vivo acute toxicity: - Nude mice treated with Ginsenoside Rk3 (10-20 mg/kg, i.p., 21 days) showed no significant changes in body weight (20 mg/kg group: final weight 20.3±1.2 g vs. control’s 21.1±0.9 g) [2] - Serum biochemical indicators: ALT (28.5±3.2 U/L vs. control’s 26.8±2.5 U/L), AST (45.2±4.1 U/L vs. control’s 43.5±3.8 U/L), BUN (5.2±0.4 mmol/L vs. control’s 5.0±0.3 mmol/L), creatinine (48.3±3.5 μmol/L vs. control’s 46.8±2.9 μmol/L) were all within normal ranges [2] |
| References |
[1]. Inhibition of TNF-α-Mediated NF-κB Transcriptional Activity by Dammarane-Type Ginsenosidesfrom Steamed Flower Buds of Panax ginseng in HepG2 and SK-Hep1 Cells. Biomol Ther (Seoul). 2014 Jan;22(1):55-61. [2]. Anticancer effects of ginsenoside Rk3 on non-small cell lung cancer cells: in vitro and in vivo. Food Funct. 2017 Oct 18;8(10):3723-3736. |
| Additional Infomation |
Ginsenoside-Rk3 is a triterpenoid. Ginsenoside Rk3 has been reported in Panax notoginseng and Panax ginseng with data available. 1. Chemical background: - Ginsenoside Rk3 is a minor ginsenoside derived from heat-processed Panax ginseng (e.g., steamed or sun-dried ginseng); it is formed by the dehydration of major ginsenosides (e.g., Rg1, Rb1) during processing [2] 2. Anticancer mechanism summary: - Ginsenoside Rk3 exerts anticancer effects on NSCLC cells via multiple pathways: (1) inducing G2/M cell cycle arrest by downregulating cyclin B1/cdc2; (2) activating the mitochondrial apoptotic pathway (caspase-9/-3 activation, Bax upregulation, Bcl-2 downregulation); (3) inhibiting NF-κB and PI3K/Akt signaling pathways to suppress cell proliferation and survival [2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~161.07 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.03 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.03 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (4.03 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6107 mL | 8.0533 mL | 16.1067 mL | |
| 5 mM | 0.3221 mL | 1.6107 mL | 3.2213 mL | |
| 10 mM | 0.1611 mL | 0.8053 mL | 1.6107 mL |