Physicochemical Properties
| Molecular Formula | C17H24O10 |
| Molecular Weight | 388.37 |
| Exact Mass | 388.136 |
| CAS # | 24512-63-8 |
| PubChem CID | 107848 |
| Appearance | White to yellow solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 641.4±55.0 °C at 760 mmHg |
| Melting Point | 161-162ºC |
| Flash Point | 231.5±25.0 °C |
| Vapour Pressure | 0.0±4.3 mmHg at 25°C |
| Index of Refraction | 1.621 |
| LogP | -1.92 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 27 |
| Complexity | 617 |
| Defined Atom Stereocenter Count | 8 |
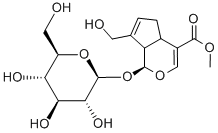
| SMILES | COC(=O)C1=CO[C@H]([C@H]2[C@@H]1CC=C2CO)O[C@H]3[C@@H]([C@H]([C@@H]([C@H](O3)CO)O)O)O |
| InChi Key | IBFYXTRXDNAPMM-BVTMAQQCSA-N |
| InChi Code | InChI=1S/C17H24O10/c1-24-15(23)9-6-25-16(11-7(4-18)2-3-8(9)11)27-17-14(22)13(21)12(20)10(5-19)26-17/h2,6,8,10-14,16-22H,3-5H2,1H3/t8-,10-,11-,12-,13+,14-,16+,17+/m1/s1 |
| Chemical Name | methyl (1S,4aS,7aS)-7-(hydroxymethyl)-1-[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-1,4a,5,7a-tetrahydrocyclopenta[c]pyran-4-carboxylate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Anti-thrombotic, anti-inflammatory, anti-diabetic, anti-atherosclerotic, anti-depressant, anti-hypertensive, toxicological, and adverse reaction properties are just a few of the actions of geniposide [1]. The production of IL-8, IL-1β, and MCP-1 in brain microvascular endothelial cells induced by OGD is significantly reduced by geniposide. This results in a significant inhibition of P2Y14 receptor expression and phosphorylation of RAF-1, MEK1/2, and ERK1/2[2]. |
| ln Vivo | In a dose-dependent way, geniposide (200 and 400 mg/kg) dramatically lowered insulin, TG, and blood glucose levels in diabetic patients. A considerable reduction in alcohol-induced blood ALT/AST and excessive rise of liver LPO levels can be achieved by using geniposide (20.0, 40.0, or 80 mg/kg), which also lowers immune protein response levels and GP and G6Pase expression at mRNA levels. By increasing the production of heme oxygenase-1 (HO-1), geniposide alleviates the transplantation-induced stress on primary cultured hippocampus neuron cells caused by 3-modenoimine hydrochloride (SIN-1) [4]. prevents IκB from degrading and turns into an extremely powerful NF-κB coupling [1]. |
| References |
[1]. Fructus Gardenia (Gardenia jasminoides J. Ellis) phytochemistry, pharmacology ofcardiovascular, and safety with the perspective of new drugs development. J Asian Nat Prod Res. 2013;15(1):94-110. [2]. Geniposide attenuates inflammatory response by suppressing P2Y14 receptor and downstream ERK1/2 signaling pathway in oxygen and glucose deprivation-induced brain microvascular endothelial cells. J Ethnopharmacol. 2016 Jun 5;185:77-86. [3]. Effect of geniposide, a hypoglycemic glucoside, on hepatic regulating enzymes in diabetic mice induced by a high-fat diet and streptozotocin. Acta Pharmacol Sin. 2009 Feb;30(2):202-8. [4]. Geniposide protects against acute alcohol-induced liver injury in mice via up-regulating the expression of the main antioxidant enzymes. Can J Physiol Pharmacol. 2015 Apr;93(4):261-7. |
| Additional Infomation |
Geniposide is a terpene glycoside. Geniposide has been reported in Rehmannia glutinosa, Gardenia jasminoides, and other organisms with data available. See also: Gardenia jasminoides whole (part of). |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~257.49 mM) H2O : ~50 mg/mL (~128.74 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.75 mg/mL (7.08 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.75 mg/mL (7.08 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.75 mg/mL (7.08 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 27.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 100 mg/mL (257.49 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5749 mL | 12.8743 mL | 25.7486 mL | |
| 5 mM | 0.5150 mL | 2.5749 mL | 5.1497 mL | |
| 10 mM | 0.2575 mL | 1.2874 mL | 2.5749 mL |