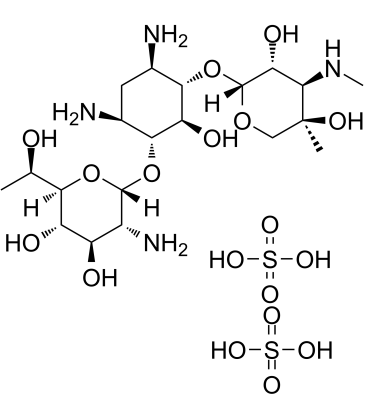
Geneticin G418 (G-418 disulfate) is a potent aminoglycoside antibiotic of the gentamicin B1 class, blocking polypeptide synthesis by inhibiting the elongation step in both prokaryotic and eukaryotic cells.
Physicochemical Properties
| Molecular Formula | C20H44N4O18S2 |
| Molecular Weight | 692.71 |
| Exact Mass | 692.209 |
| Elemental Analysis | C, 34.68; H, 6.40; N, 8.09; O, 41.57; S, 9.26 |
| CAS # | 108321-42-2 |
| Related CAS # | G-418;49863-47-0; 49863-47-0; 108321-42-2 (sulfate) |
| PubChem CID | 206347 |
| Appearance | White to off-white solid powder |
| Boiling Point | 1012.1ºC at 760mmHg |
| Flash Point | 565.9ºC |
| Vapour Pressure | 0mmHg at 25°C |
| Source | Micromonospora rhodorangea |
| Hydrogen Bond Donor Count | 14 |
| Hydrogen Bond Acceptor Count | 22 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 44 |
| Complexity | 763 |
| Defined Atom Stereocenter Count | 15 |
| SMILES | S(=O)(=O)(O[H])O[H].S(=O)(=O)(O[H])O[H].O([C@@]1([H])C([H])(C([H])(C([H])([C@]([H])(C([H])(C([H])([H])[H])O[H])O1)O[H])O[H])N([H])[H])C1([H])C([H])(C([H])([H])C([H])(C([H])(C1([H])O[H])O[C@@]1([H])C([H])(C([H])([C@](C([H])([H])[H])(C([H])([H])O1)O[H])N([H])C([H])([H])[H])O[H])N([H])[H])N([H])[H] |
| InChi Key | UHEPSJJJMTWUCP-DHDYTCSHSA-N |
| InChi Code | InChI=1S/C20H40N4O10.2H2O4S/c1-6(25)14-11(27)10(26)9(23)18(32-14)33-15-7(21)4-8(22)16(12(15)28)34-19-13(29)17(24-3)20(2,30)5-31-19;2*1-5(2,3)4/h6-19,24-30H,4-5,21-23H2,1-3H3;2*(H2,1,2,3,4)/t6-,7+,8-,9-,10-,11+,12+,13-,14-,15-,16+,17-,18-,19-,20+;;/m1../s1 |
| Chemical Name | (2R,3S,4R,5R,6S)-5-amino-6-(((1R,2S,3S,4R,6S)-4,6-diamino-3-(((2R,3R,4R,5R)-3,5-dihydroxy-5-methyl-4-(methylamino)tetrahydro-2H-pyran-2-yl)oxy)-2-hydroxycyclohexyl)oxy)-2-((R)-1-hydroxyethyl)tetrahydro-2H-pyran-3,4-diol bis(sulfate) |
| Synonyms | Geneticin G418; Geneticin G-418; Geneticin G 418; Antibiotic G418; |
| HS Tariff Code | 2934.99.03.00 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Aminoglycoside | |
| ln Vitro |
An aminoglycoside neomycin analogue called G418 sulfate inhibits protein synthesis and is widely used to select mammalian cell lines with neomycin resistance (NR). In eukaryotic vectors, the neomycin resistance (neo) gene is commonly employed as a dominant selectable gene. Asialoorosomucoid (AsOR) and G418 can be covalently bound to create a conjugate that is toxic and specific to hepatocytes[2]. MDA-MB231 cells were transfected with human GD3 synthase cDNA, and cells that stably overexpressed the enzyme were chosen using G-418 bulk selection[2]. Numerous pro- and eukaryotes have been demonstrated to be inhibited by the aminoglycoside antibiotic G418 at concentrations ranging from 1 to 300 micrograms/ml[4]. |
|
| ln Vivo |
|
|
| Cell Assay | Acanthamoeba is a widely distributed opportunistic parasite which causes a vision-threatening keratitis and a life-threatening encephalitis. The cyst stage of this amoeba is especially resistant to currently used therapeutics and so alternative agents are urgently required. Growing evidence supports the existence of a programmed cell death system (PCD) in Acanthamoeba and while some features are shared by higher eukaryote cells, others differ. It is hoped that by understanding these differences we can exploit them as targets for novel drug intervention to activate PCD pathways in the amoebae but not the invaded human tissue. Here, we use the aminoglycoside G418 to activate PCD in Acanthamoeba. This drug caused a shape change in the treated amoebae. Cells rounded up and contracted, and after 6 h fragments of cells resembling the 'apoptotic bodies' of vertebrate cells were observed. G418 causes an increase in intracellular calcium from a resting level of 24 nM to 60 nM after 6 h of treatment. Mitochondrial function as assayed by the ΔΨm reporting dye JC-1 and CTC a redox dye becomes inhibited during treatment and we have found that cytochrome c is released from the mitochondria. Cells stained with Hoechst showed first an alteration in chromatin structure and then a vesiculation of the nucleus with G418 treatment, although we found no obvious breakdown in genomic DNA in the early stages of PCD [5]. | |
| Animal Protocol | Mouse RML and 22L prion inocula were obtained from RML- or 22L-infected CAD5 cells (34). The cells were grown to confluency, scraped in 1 ml PBS per 10 cm dish, and then homogenized using a Minilys bead homogenizer and CK14 homogenization tubes (Bertin). Benzonase (50 units/ml; EMD Millipore #70746-4) was added into the scraped culture, and then the culture was homogenized for three cycles of 30 s at maximum speed, with 5 min of incubation on ice between each cycle. Cell homogenates were then stored at −80 °C. Syrian hamster 263K prion inoculum was prepared in a similar way from 263K-infected CAD5-PrP−/− cells stably expressing HaPrP.[6] | |
| References |
[1]. An antibiotic selection marker for nematode transgenesis. Nat Methods. 2010;7(9):721-723. [2]. A novel G418 conjugate results in targeted selection of genetically protected hepatocytes without bystander toxicity. Bioconjug Chem. 2007;18(6):1965-1971. [3]. Disialyl GD2 ganglioside suppresses ICAM-1-mediated invasiveness in human breast cancer MDA-MB231 cells. Int J Biol Sci. 2017;13(3):265-275. Published 2017 Feb 12. [4]. A new selective agent for eukaryotic cloning vectors. Am J Trop Med Hyg. 1980;29(5 Suppl):1089-1092. [5]. G418 induces programmed cell death in Acanthamoeba through the elevation of intracellular calcium and cytochrome c translocation. Parasitol Res. 2019 Feb;118(2):641-651.[6]. J Biol Chem. 2021 Sep;297(3):101073. doi: 10.1016/j.jbc.2021.101073. Epub 2021 Aug 12. |
|
| Additional Infomation | The study of prions and the discovery of candidate therapeutics for prion disease have been facilitated by the ability of prions to replicate in cultured cells. Paradigms in which prion proteins from different species are expressed in cells with low or no expression of endogenous prion protein (PrP) have expanded the range of prion strains that can be propagated. In these systems, cells stably expressing a PrP of interest are typically generated via coexpression of a selectable marker and treatment with an antibiotic. Here, we report the unexpected discovery that the aminoglycoside G418 (Geneticin) interferes with the ability of stably transfected cultured cells to become infected with prions. In G418-resistant lines of N2a or CAD5 cells, the presence of G418 reduced levels of protease-resistant PrP following challenge with the RML or 22L strains of mouse prions. G418 also interfered with the infection of cells expressing hamster PrP with the 263K strain of hamster prions. Interestingly, G418 had minimal to no effect on protease-resistant PrP levels in cells with established prion infection, arguing that G418 selectively interferes with de novo prion infection. As G418 treatment had no discernible effect on cellular PrP levels or its localization, this suggests that G418 may specifically target prion assemblies or processes involved in the earliest stages of prion infection.[6] |
Solubility Data
| Solubility (In Vitro) | Water : 100~125 mg/mL(~180.45 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 50 mg/mL (72.18 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. Solubility in Formulation 2: Saline: 30mg/ml (43.31mM) (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4436 mL | 7.2180 mL | 14.4361 mL | |
| 5 mM | 0.2887 mL | 1.4436 mL | 2.8872 mL | |
| 10 mM | 0.1444 mL | 0.7218 mL | 1.4436 mL |