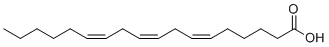
Gamolenic acid is a novel and potent omega-6 fatty acid PPAR agonist
Physicochemical Properties
| Molecular Formula | C18H30O2 |
| Molecular Weight | 278.43 |
| Exact Mass | 278.224 |
| CAS # | 506-26-3 |
| PubChem CID | 5280933 |
| Appearance | Colorless to light yellow liquid(Density:0.92 g/cm3) |
| Density | 0.9±0.1 g/cm3 |
| Boiling Point | 379.5±11.0 °C at 760 mmHg |
| Flash Point | 276.4±14.4 °C |
| Vapour Pressure | 0.0±1.8 mmHg at 25°C |
| Index of Refraction | 1.491 |
| LogP | 6.57 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 20 |
| Complexity | 301 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CCCCC/C=C\C/C=C\C/C=C\CCCCC(O)=O |
| InChi Key | VZCCETWTMQHEPK-QNEBEIHSSA-N |
| InChi Code | InChI=1S/C18H30O2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18(19)20/h6-7,9-10,12-13H,2-5,8,11,14-17H2,1H3,(H,19,20)/b7-6-,10-9-,13-12- |
| Chemical Name | (6Z,9Z,12Z)-octadeca-6,9,12-trienoic acid |
| Synonyms | Gamolenic acidγ-Linolenic Acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion The findings from a pharmacokinetic study suggest that therapeutic levels of GLA can be achieved within a week. The fasting plasma GLA levels plateaued within seven days of beginning treatment, regardless of dose. The metabolites of gamolenic acid is expected to undergo renal excretion. No pharmacokinetic data available. No pharmacokinetic data available. Metabolism / Metabolites Via elongation mediated by elongase (ELOVL5), gamolenic acid is rapidly converted to dihomo-gamma-linolenic acid (DGLA), which is further cyclooxygenated to prostaglandin E1 (PGE1) via COX-1 or COX-2 enzymatic activity depending on the cell type. PGE1 may be metabolized to smaller prostaglandin remnants, primarily dicarboxylic acids, which undergo renal excretion. DGLA may be converted to 15-(S)-hydroxy-8,11,13-eicosatrienoic acid (15-HETrE) by 15-lipoxygenase enzyme. Although the enzymatic pathway is less predominant relative to ELOVL5 in most cells, DGLA may also be converted to arachidonic acid (AA) via delta-5-desaturate activity, where hydrogen atoms are selectively removed to create new double bonds F27]. Arachidonic acid is a precursor in the biosynthesis of prostaglandin E2, thromboxanes, and leukotrienes, which are potent inflammatory mediators and play an important role in inflammatory pathways. Biological Half-Life No pharmacokinetic data available. |
| Toxicity/Toxicokinetics |
Protein Binding No pharmacokinetic data available. |
| Additional Infomation |
Pharmacodynamics Gamolenic acid is converted to PGE1, which exhibits anti-inflammatory, antithrombotic, antiproliferative, and lipid-lowering effects. PGE1 also induces smooth muscle relaxation and vasodilation. Gamolenic acid is an essential component of membrane phospholipids, including the mitochondrial membrane, where it enhances the the integrity and the fluidity of the membrane. **Bone and joint health:** In a pilot study of women with a mean age of 79.5 years and senile osteoporosis, the use of gamolenic acid in combination with calcium and eicosapentaenoic acid was associated with an increase in femoral bone density and lumbar spine density in comparison to placebo, where there were no observable changes. In clinical studies of patients with rheumatoid arthritis, treatment with gamolenic acid-containing oils resulted in an improvement in symptoms, measured by joint tenderness counts and scores, joint swelling scores, physician global assessment, and pain. **Inflammation:** A study demonstrated that oral administration of gamolenic acid suppressed human T-cell proliferation and activation by interfering with early events in the TcR/CD3-receptor–mediated signal transduction cascade. **Atherosclerosis:** In ApoE genetic knock-out mice, dietary gamolenic acid was shown to reduce the average medial layer thickness of the vessel wall and reduces the size of atherosclerotic lesions. **Diabetic complications:** In a clinical trial of patients with mild diabetic neuropathy or distal diabetic neuropathy, treatment with gamolenic acid was associated with improved symptoms in hot and cold threshold, sensation, tendon reflexes, and muscle strength. GLA ameliorated the inflammatory profile in diabetic nephropathy in rat studies. **Cancer:** In three human tumor cell lines (the neuroblastoma CHP-212, the tubal carcinoma TG, and the colon carcinoma SW-620), gamolenic acid elicited cytotoxic effects in tumours by blocking cell proliferation following incorporation into malignant cells. In both clinical and animal studies of breast cancer, gamolenic acid, in combination with tamoxifen, down-regulated the expression of estrogen receptors. **Skin disorders:** In an open study of patients with atopic dermatitis, which is a disorder related to a deficiency of delta-6-desaturase and inefficient conversion of linoleic acid to gamolenic acid, daily administration of gamolenic acid was associated with a significant increase in plasma GLA and DGLA levels in combination with an improvement of clinical signs of atopic dermatitis. **Respiratory disorders:** In patients with acute lung injury or acute respiratory distress syndrome, gamolenic acid was shown to reduce cytokine production and neutrophil recruitment into the lung. In patients with atopic asthma, gamolenic acid blocked _ex vivo_ synthesis of leukotrienes from whole blood and isolated neutrophils compared to the placebo group. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5916 mL | 17.9578 mL | 35.9157 mL | |
| 5 mM | 0.7183 mL | 3.5916 mL | 7.1831 mL | |
| 10 mM | 0.3592 mL | 1.7958 mL | 3.5916 mL |