Physicochemical Properties
| Molecular Formula | C23H24O6 |
| Molecular Weight | 396.4331 |
| Exact Mass | 396.157 |
| CAS # | 31271-07-5 |
| PubChem CID | 5464078 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 648.4±55.0 °C at 760 mmHg |
| Melting Point | 207 °C |
| Flash Point | 226.4±25.0 °C |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.656 |
| LogP | 5.14 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 29 |
| Complexity | 662 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | VEZXFTKZUMARDU-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C23H24O6/c1-11(2)5-7-13-15(24)9-18-20(22(13)27)23(28)19-14(8-6-12(3)4)21(26)16(25)10-17(19)29-18/h5-6,9-10,24-27H,7-8H2,1-4H3 |
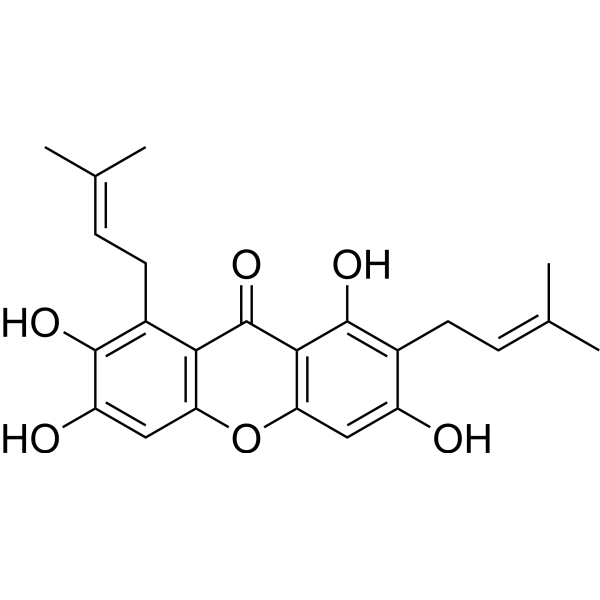
| Chemical Name | 1,3,6,7-tetrahydroxy-2,8-bis(3-methylbut-2-enyl)xanthen-9-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- 5-Hydroxytryptamine 2A (5-HT₂A) receptor (Ki = 0.3 μM)[1] - Amyloid-beta (Aβ) aggregation and production-related targets [2] |
| ln Vitro |
- Rat aortic strips: Gamma-Mangostin acts as a selective 5-HT₂A receptor antagonist, dose-dependently inhibiting 5-HT-induced vasoconstriction with maximal inhibition rate of ~80% at 1 μM[1] - SH-SY5Y cells: Gamma-Mangostin inhibits Aβ₁₋₄₂ aggregation with an IC₅₀ of 12.5 μM, and reduces Aβ₁₋₄₀/Aβ₁₋₄₂ production by suppressing cleavage of amyloid precursor protein (APP) via downregulating BACE1 expression[2] - 3T3-L1 adipocytes (insulin-resistant model): Gamma-Mangostin (10, 20 μM) enhances insulin sensitivity, promoting glucose uptake by 30-45% compared to control, and upregulates GLUT4 membrane translocation[5] |
| ln Vivo |
- STZ-induced diabetic mice: Oral administration of Gamma-Mangostin (20, 40 mg/kg/day for 4 weeks) improves liver function, reducing serum ALT and AST levels by 40-60%, and alleviates hepatic steatosis and inflammatory infiltration[3] - STZ-induced diabetic mice: Gamma-Mangostin (20, 40 mg/kg/day, oral for 4 weeks) exerts renal protective effects, decreasing serum creatinine and urea nitrogen levels by 35-55%, and attenuating renal glomerular hypertrophy, tubular damage, and interstitial fibrosis[4] - STZ-induced diabetic mice: Gamma-Mangostin (20, 40 mg/kg/day, oral for 4 weeks) synergizes with insulin to lower fasting blood glucose (by 30-50%) and glycated hemoglobin (HbA₁c) levels, and enhances insulin signaling via increasing Akt phosphorylation in liver and muscle tissues[5] |
| Enzyme Assay |
- 5-HT₂A receptor competitive binding assay: Membrane preparations from rat brain cortex expressing 5-HT₂A receptor are incubated with radiolabeled 5-HT and various concentrations of Gamma-Mangostin (0.01-10 μM) at 25°C for 60 minutes. Bound radioligand is separated by filtration, and radioactivity is measured. Ki value is calculated via nonlinear regression analysis of competition binding curves[1] - Aβ aggregation inhibition assay: Aβ₁₋₄₂ peptide is incubated with Gamma-Mangostin (5-50 μM) at 37°C for 48 hours. Thioflavin T (ThT) fluorescent probe is added, and fluorescence intensity is detected at excitation wavelength 440 nm and emission wavelength 485 nm to evaluate Aβ aggregation degree, with IC₅₀ value calculated[2] |
| Cell Assay |
- Rat aortic strip contraction assay: Isolated rat thoracic aortic strips are suspended in Krebs-Ringer bicarbonate buffer (37°C, aerated with 95% O₂ and 5% CO₂) and equilibrated for 60 minutes. Vasoconstriction is induced by 5-HT (1 μM), followed by addition of Gamma-Mangostin (0.1-1 μM). Contractile tension is recorded via a force transducer, and inhibition rate is calculated[1] - Aβ production detection assay: SH-SY5Y cells are seeded in 6-well plates and cultured to 70% confluence. Cells are treated with Gamma-Mangostin (5-20 μM) for 24 hours. Cell supernatants and lysates are collected; Aβ₁₋₄₀/Aβ₁₋₄₂ concentrations are measured by ELISA, and APP/BACE1 protein expressions are detected by Western blot[2] - Glucose uptake assay: Differentiated 3T3-L1 adipocytes are induced to insulin resistance. Cells are treated with Gamma-Mangostin (10, 20 μM) and insulin (10 nM) for 12 hours. Fluorescently labeled 2-deoxyglucose is added, and intracellular fluorescence intensity is measured to quantify glucose uptake[5] |
| Animal Protocol |
- Diabetic mice hepatoprotective study: Male mice are intraperitoneally injected with streptozotocin (STZ) to establish diabetic models. Mice are randomly divided into control group, Gamma-Mangostin low-dose group (20 mg/kg) and high-dose group (40 mg/kg). The test compound is administered orally once daily for 4 weeks. Serum ALT and AST levels are detected; liver tissues are collected for HE staining and immunohistochemical detection of inflammatory factors[3] - Diabetic mice nephroprotective study: STZ-induced diabetic mice are grouped and administered Gamma-Mangostin as described in the hepatoprotective study for 4 weeks. Serum creatinine and urea nitrogen levels are measured; kidney tissues are subjected to PAS staining and Masson staining to evaluate tissue damage and fibrosis[4] - Diabetic mice hypoglycemic study: STZ-induced diabetic mice are randomly divided into control group, insulin group, and insulin + Gamma-Mangostin groups (20, 40 mg/kg). Insulin is injected subcutaneously, and Gamma-Mangostin is administered orally once daily for 4 weeks. Fasting blood glucose is measured weekly; HbA₁c and insulin levels are detected at the end of the study, and Akt phosphorylation in liver and muscle tissues is analyzed[5] |
| Toxicity/Toxicokinetics |
- In vivo toxicity: Oral administration of Gamma-Mangostin (20, 40 mg/kg/day for 4 weeks) does not cause obvious systemic toxicity in diabetic mice, as indicated by stable body weight, no significant histological damage in heart, spleen and other major organs, and normal serum biochemical parameters[3][4][5] |
| References |
[1]. Gamma-mangostin, a novel type of 5-hydroxytryptamine 2A receptor antagonist. Naunyn Schmiedebergs Arch Pharmacol. 1998 Jan;357(1):25-31. [2]. Discovery of γ-Mangostin as an Amyloidogenesis Inhibitor. Sci Rep. 2015 Aug 27;5:13570. [3]. Hepatoprotective Effect of Gamma-mangostin for Amelioration of Impaired Liver Structure and Function in Streptozotocin-induced Diabetic Mice. 2018 IOP Conf. Ser.: Earth Environ. Sci. 217 012031. [4]. Renal protective effects of gamma-mangostin in streptozotocin-induced diabetic mice. Indian Journal of Forensic Medicine & Toxicology, 2020, 14(3): 1251-1256. [5]. Gamma-mangostin of Garcinia mangostana peels ameliorates hyperglycemia in synergism of insulin. The FASEB Journal, 33: 694.11-694.11. |
| Additional Infomation |
Gamma-mangostin is a member of the class of xanthones that is 9H-xanthene substituted by hydroxy group at positions 1, 3, 6 and 7, an oxo group at position 9 and prenyl groups at positions 2 and 8. Isolated from the stems of Cratoxylum cochinchinense, it exhibits antitumour activity. It has a role as an antineoplastic agent, a protein kinase inhibitor and a plant metabolite. It is a member of xanthones and a member of phenols. gamma-Mangostin has been reported in Garcinia xipshuanbannaensis, Hypericum perforatum, and other organisms with data available. See also: Garcinia mangostana fruit rind (part of). - Gamma-Mangostin is a natural flavonoid compound isolated from the peels of Garcinia mangostana (mangosteen)[3][5] - Its mechanisms of action include selective antagonism of 5-HT₂A receptor[1], inhibition of Aβ aggregation and APP cleavage[2], activation of insulin signaling pathway to enhance insulin sensitivity[5], and alleviation of diabetes-related hepatorenal damage via anti-inflammatory and antioxidant effects[3][4] - It has potential therapeutic value for 5-HT₂A receptor-related diseases, Alzheimer's disease, and diabetic complications (hepatic and renal diseases)[1][2][3][4][5] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~252.25 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.31 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.31 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.31 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5225 mL | 12.6126 mL | 25.2251 mL | |
| 5 mM | 0.5045 mL | 2.5225 mL | 5.0450 mL | |
| 10 mM | 0.2523 mL | 1.2613 mL | 2.5225 mL |