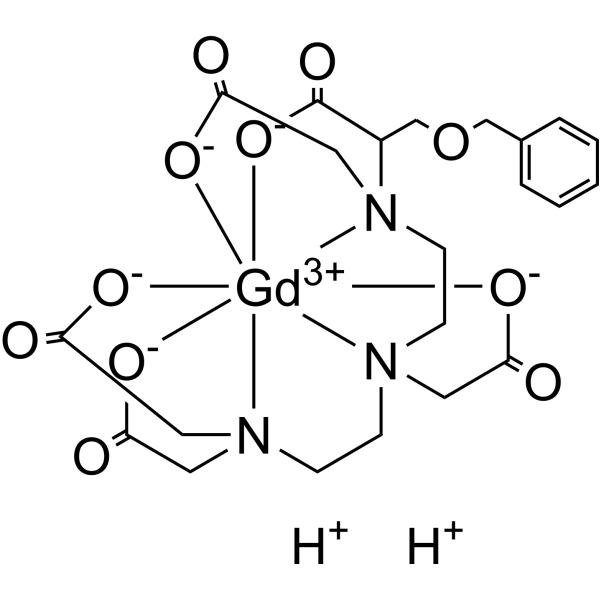
Physicochemical Properties
| Molecular Formula | C22H28GDN3O11 |
| Molecular Weight | 667.7 |
| Exact Mass | 668.096 |
| CAS # | 113662-23-0 |
| PubChem CID | 131700865 |
| Appearance | Hygroscopic powder |
| Melting Point | 124 °C |
| Vapour Pressure | 1.21E-26mmHg at 25°C |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 14 |
| Rotatable Bond Count | 17 |
| Heavy Atom Count | 37 |
| Complexity | 726 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | [Gd+3].O(CC1C=CC=CC=1)CC(C(=O)[O-])N(CC(=O)[O-])CCN(CC(=O)[O-])CCN(CC(=O)[O-])CC(=O)[O-].[H+].[H+] |
| InChi Key | MXZROTBGJUUXID-UHFFFAOYSA-K |
| InChi Code | InChI=1S/C22H31N3O11.Gd/c26-18(27)10-23(6-7-24(11-19(28)29)12-20(30)31)8-9-25(13-21(32)33)17(22(34)35)15-36-14-16-4-2-1-3-5-16;/h1-5,17H,6-15H2,(H,26,27)(H,28,29)(H,30,31)(H,32,33)(H,34,35);/q;+3/p-3 |
| Chemical Name | 2-[2-[carboxylatomethyl(carboxymethyl)amino]ethyl-[2-[carboxylatomethyl-(1-carboxy-2-phenylmethoxyethyl)amino]ethyl]amino]acetate;gadolinium(3+) |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Gadobenate ion has a rapid distribution half-life (reported as mean + or - SD) of 0.084 + or - 0.012 to 0.605 + or - 0.072 hours. Volume of distribution of the central compartment ranged from 0.074:: 0.017 to 0.158 :: 0.038 L/kg, and estimates of volume of distribution by area ranged from 0.170+ or - 0.016 to 0.282+ or - 0.079 L/kg. These latter estimates are approximately equivalent to the average volume of extracellular body water in man. In vitro studies showed no appreciable binding of gadobenate ion to human serum proteins. /Gadobenate ion/ ... The pharmacokinetics were evaluated in rats, rabbits, dogs and monkeys after iv injections of non-labelled gadobenate dimeglumine and, for biodistribution studies, (153)-Gd-labelled gadobenate dimeglumine. Assays were performed by high performance liquid chromatography, X-ray fluorescence and gamma spectrometry. The binding of gadobenate ion to animal and human serum albumin was studied by equilibrium dialysis. ... After iv injection gadobenate dimeglumine distributes into plasma and extracellular fluid as well as into the intrahepatocytic space. Gadobenate ion is cleared from plasma by renal and biliary excretion. It does not accumulate in specific tissues, except temporarily in tissues related to its elimination. Gadobenate ion is not metabolized. Its binding to plasma proteins is too weak to be detected by equilibrium dialysis. ... Gadobenate ion is eliminated predominately via the kidneys, with 78% to 96% of an administered dose recovered in the urine. Total plasma clearance and renal clearance estimates of gadobenate ion were similar, ranging from 0.093 + or - 0.010 to 0.133 + o r- 0.270 L/hr/kg and 0.082+ or - 0.007 to 0.104 + or - 0.039 L/hr/kg, respectively. The clearance is similar to that of substances that are subject to glomerular filtration. The mean elimination half-life ranged from 1.17+ or - 0.26 to 2.02 + or - 0.60 hours. A small percentage ofthe administered dose (0.6% to 4%) is eliminated via the biliary route and recovered in feces. It is not known to what extent gadobenate dimeglumine is excreted in human milk. It is known from rat experiments that less than 0.5% of the administered dose is transferred via milk from mother to neonates. For more Absorption, Distribution and Excretion (Complete) data for GADOBENATE DIMEGLUMINE (7 total), please visit the HSDB record page. Metabolism / Metabolites There was no detectable biotransformation of gadobenate ion. Dissociation of gadobenate ion in vivo has been shown to be minimal, with less than 1% of the free chelating agent being recovered alone in feces. Biological Half-Life Gadobenate ion is eliminated predominately via the kidneys, with 78% to 96% of an administered dose recovered in the urine. ... The mean elimination half-life ranged from 1.17+ or - 0.26 to 2.02 + or - 0.60 hours. ... ... Fifteen children scheduled to undergo contrast-enhanced MRI for suspected disease of the central nervous system received a single intravenous injection of 0.1 mmol/kg gadobenate dimeglumine. ... 1.2 hr for terminal elimination half-life were determined across all age groups combined. A single intravenous dose of 0.2 mmol/kg of Multihance was administered to 11 subjects (5 males and 6 females) with end-stage renal disease requiring hemodialysis ... . The mean elimination half-life on dialysis was 1.21 + or - 0.29 hours as compared with 42.4 + or - 24.4 hours when off dialysis. A single intravenous dose of 0.2 mmol/kg of Multihance was administered to 20 subjects with impaired renal function (6 men and 3 women with moderate renal impairment (urine creatinine clearance > 30 to < 60 mL/min) and 5 men and 6 women with severe renal impairment (urine creatinine clearance > 10 to < 30 mL/min)). Mean estimates of the elimination half-life were 6.1 + or - 3.0 and 9.5 + or - 3.1 hours for the moderate and severe renal impairment groups, respectively as compared with 1.0 to 2.0 hours in healthy volunteers. Gadobenate ion has a rapid distribution half-life (reported as mean + or - SD) of 0.084 + or - 0.012 to 0.605 + or - 0.072 hours. |
| Toxicity/Toxicokinetics |
Toxicity Summary IDENTIFICATION AND USE: Gadobenate dimeglumine (MultiHance) is a contract agent for MRI studies, supplied as a colorless to slightly yellow, aqueous solution intended for intravenous use only. HUMAN EXPOSURE AND TOXICITY: Gadolinium-based contrast agents (GBCAs) increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of GBCAs in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. NSF may result in fatal or debilitating systemic fibrosis affecting the skin, muscle and internal organs. The risk for NSF appears highest among patients with: chronic, severe kidney disease, or acute kidney injury. Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (older than 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing. For patients at highest risk for NSF, do not exceed the recommended MultiHance dose and allow a sufficient period of time for elimination of the drug from the body prior to re-administration. Anaphylactic and anaphylactoid reactions have been reported, involving cardiovascular, respiratory, and/or cutaneous manifestations. Some patients experienced circulatory collapse and died. Cardiac arrhythmias have been observed in patients receiving MultiHance in clinical trials. The mutagenic potential of gadobenate dimeglumine was studied by the chromosome aberration test in cultured human lymphocytes. The agent induced no increase in the incidence of aberrant cells or polyploid cells in any treatments both in the presence and absence of metabolic activation. Thus, it is concluded that gadobenate dimeglumine has shown no evidence of clastogenic or polyploidy-inducing activity under these experimental conditions. ANIMAL STUDIES: To support the clinical use of gadobenate dimeglumine for injection as an intravascular magnetic resonance imaging contrast medium through an extensive battery of toxicological safety studies. Single and multiple dose toxicity, reproduction and mutagenicity assessments were carried out in rodents and non-rodents. Initial adverse clinical signs in monkeys were associated with a systemic exposure 34 times higher than that found in humans after 0.1 mmol/kg gadobenate dimeglumine. Good systemic tolerance was observed in repeated dose toxicity studies. In experimental conditions of focal brain ischemia associated with blood-brain barrier lesions, gadobenate dimeglumine was well tolerated up to doses even 10 times higher than the maximum clinical dose (0.3 mmol/kg) intended for brain imaging procedures. Reproductive performance and physical and behavioral development of offspring were unaffected in rats. However, MultiHance has been shown to be teratogenic in rabbits when given intravenously administered at 2 mmol/kg/day (6 times the human dose based on body surface area) during organogenesis (day 6 to 18) inducing microphthalmia/small eye and/or focal retinal fold in 3 fetuses from 3 separate litters. In addition, MultiHance intravenously administered at 3 mmol/kg/day (10 times the human dose based on body surface area) has been shown to increase intrauterine deaths in rabbits. Mutagenicity tests excluded any genotoxic potential of gadobenate dimeglumine. The results were negative in the following genetic toxicity studies: 1) in vitro bacteria reverse mutation assays, 2) an in vitro gene mutation assay in mammalian cells, 3) an in vitro chromosomal aberration assay, 4) an in vitro unscheduled DNA synthesis assay, and 5) an in vivo micronucleus assay in rats. Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Gadobenate has been safely used in infants, so it does not appear to pose a substantial risk to a breastfed infant. Guidelines developed by North American professional organizations state that breastfeeding need not be disrupted after a nursing mother receives a gadolinium-containing contrast medium. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Interactions MultiHance and other drugs may compete for the canalicular multispecific organic anion transporter (MOAT also referred to as MRP2 or ABCC2). Therefore MultiHance may prolong the systemic exposure of drugs such as cisplatin, antracyclines (e.g. doxorubicin, daunorubicin), vinca alkaloids (e.g. vincristine), methotrexate, etoposide, tamoxifen, and paclitaxel. In particular, consider the potential for prolonged drug exposure in patients with decreased MOAT activity (e.g. Dubin Johnson syndrome). Non-Human Toxicity Values LD50 Mouse iv 5.7 mmol/kg (at 1 mL/min), 7.9 mmol/kg (at 0.2 mL/min) LD50 Rat iv 6.6 mmol/kg (at 6 mL/min), 9.2 mmol/kg (at 1 mL/min) LD50 Rat (male) intracisternal 0.42 mmol/kg LD50 Rat (female) intracisternal 0.25 mmol/kg |
| References |
[1]. Gadoxetic acid-enhanced MRI for the characterization of hepatocellular carcinoma: A systematic review and meta-analysis. J Magn Reson Imaging. 2017 Jan;45(1):281-290. |
| Additional Infomation |
Gadobenate Dimeglumine is a gadolinium-based paramagnetic contrast agent. When placed in a magnetic field, gadobenate dimeglumine produces a large magnetic moment and so a large local magnetic field, which can enhance the relaxation rate of nearby protons; as a result, the signal intensity of tissue images observed with magnetic resonance imaging (MRI) may be enhanced. Because this agent is preferentially taken up by normal functioning hepatocytes, normal hepatic tissue is enhanced with MRI while tumor tissue is unenhanced. In addition, because gadobenate dimeglumine is excreted in the bile, it may be used to visualize the biliary system using MRI. See also: Gadobenic Acid (preferred). Therapeutic Uses Contrast Media MultiHance is indicated for intravenous use in magnetic resonance imaging (MRI) of the central nervous system (CNS) in adults and children over 2 years of age to visualize lesions with abnormal blood brain barrier or abnormal vascularity of the brain, spine, and associated tissues. /Included in US product labeling/ MultiHance is indicated for use in magnetic resonance angiography (MRA) to evaluate adults with known or suspected renal or aorto-ilio-femoral occlusive vascular disease. /Included in US product label/ A study was conducted to compare gadobenate dimeglumine (Multihance) with other commercially available MRI contrast agents for the detection of intracranial metastases ... A retrospective assessment was performed on MR images from 22 patients enrolled in a prior phase II clinical trial of gadobenate dimeglumine. Each patient underwent two examinations: a first examination with one of three "comparator" agents (gadopentetate dimeglumine, gadodiamide, and gadoterate meglumine) at a dosage of either 0.1 or 0.2 mmol/kg, and then a similar examination with gadobenate dimeglumine at equal dosage. All images were evaluated randomly for lesion number and location in unpaired and then paired fashion by two independent, masked neuroradiologists. A third assessor performed quantitative assessments on the available complete sets of digitally recorded images (10 cases) ... The findings for the comparator agents were pooled. Sensitivity for lesion detection with gadobenate dimeglumine (93%-100%) was markedly superior to that of comparator-enhanced examinations (65%-73%). The increase of lesion-to-brain contrast of the main lesion was consistently greater with gadobenate dimeglumine than with comparator agents relative to unenhanced contrast (+43% vs. +27%) ... Gadobenate dimeglumine proved to be a more efficacious agent than comparator contrast agents for the detection of intracranial metastatic lesions: superior efficacy was noted by both reviewers for total lesion count as well as for sensitivity and positive predictive value for lesion detection. The higher relaxivity of gadobenate dimeglumine might explain the superior sensitivity of gadobenate dimeglumine-enhanced MRI for the detection of central nervous system metastases. For more Therapeutic Uses (Complete) data for GADOBENATE DIMEGLUMINE (9 total), please visit the HSDB record page. Drug Warnings /BOXED WARNING/ WARNING: NEPHROGENIC SYSTEMIC FIBROSIS. Gadolinium-based contrast agents (GBCAs) increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of GBCAs in these patients unless the diagnostic information is essential and not available with non-contrasted MRI or other modalities. NSF may result in fatal or debilitating systemic fibrosis affecting the skin, muscle and internal organs. The risk for NSF appears highest among patients with: chronic, severe kidney disease (GFR <30 mL/min/1.73 sq m), or acute kidney injury. Screen patients for acute kidney injury and other conditions that may reduce renal function. For patients at risk for chronically reduced renal function (e.g. age > 60 years, hypertension or diabetes), estimate the glomerular filtration rate (GFR) through laboratory testing. For patients at highest risk for NSF, do not exceed the recommended MultiHance dose and allow a sufficient period of time for elimination of the drug from the body prior to re-administration. Gadolinium-based contrast agents (GBCAs) increase the risk for nephrogenic systemic fibrosis (NSF) among patients with impaired elimination of the drugs. Avoid use of GBCAs among these patients unless the diagnostic information is essential and not available with non-contrast enhanced MRI or other modalities. The GBCA-associated NSF risk appears highest for patients with chronic, severe kidney disease (GFR <30 mL/min/1.73 sq m) as well as patients with acute kidney injury. The risk appears lower for patients with chronic, moderate kidney disease (GFR 30-59 mL/min/1.73 sq m) and little, if any, for patients with chronic, mild kidney disease (GFR 60-89 mL/min/1.73 sq m). NSF may result in fatal or debilitating fibrosis affecting the skin, muscle and internal organs. ... Screen patients for acute kidney injury and other conditions that may reduce renal function. Features of acute kidney injury consist of rapid (over hours to days) and usually reversible decrease in kidney function, commonly in the setting of surgery, severe infection, injury or drug-induced kidney toxicity. Serum creatinine levels and estimated GFR may not reliably assess renal function in the setting of acute kidney injury. For patients at risk for chronically reduced renal function (e.g., age > 60 years, diabetes mellitus or chronic hypertension), estimate the GFR through laboratory testing. Among the factors that may increase the risk for NSF are repeated or higher than recommended doses of a GBCA and the degree of renal impairment at the time of exposure. Record the specific GBCA and the dose administered to a patient. For patients at highest risk for NSF, do not exceed the recommended MultiHance dose and allow a sufficient period of time for elimination of the drug prior to re-administration. For patients receiving hemodialysis, physicians may consider the prompt initiation of hemodialysis following the administration of a GBCA in order to enhance the contrast agent's elimination. The usefulness of hemodialysis in the prevention of NSF is unknown Anaphylactic and anaphylactoid reactions have been reported, involving cardiovascular, respiratory, and/or cutaneous manifestations. Some patients experienced circulatory collapse and died. In most cases, initial symptoms occurred within minutes of MultiHance administration and resolved with prompt emergency treatment. Prior to MultiHance administration, ensure the availability of personnel trained and medications to treat hypersensitivity reactions. If such a reaction occurs stop MultiHance and immediately begin appropriate therapy. Additionally, consider the risk for hypersensitivity reactions, especially in patients with a history of hypersensitivity reactions or a history of asthma or other allergic disorders. Observe patients for signs and symptoms of a hypersensitivity reaction during and for up to 2 hours after MultiHance administration. FDA Pregnancy Risk Category: C /RISK CANNOT BE RULED OUT. Adequate, well controlled human studies are lacking, and animal studies have shown risk to the fetus or are lacking as well. There is a chance of fetal harm if the drug is given during pregnancy; but the potential benefits may outweigh the potential risk./ For more Drug Warnings (Complete) data for GADOBENATE DIMEGLUMINE (15 total), please visit the HSDB record page. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4977 mL | 7.4884 mL | 14.9768 mL | |
| 5 mM | 0.2995 mL | 1.4977 mL | 2.9954 mL | |
| 10 mM | 0.1498 mL | 0.7488 mL | 1.4977 mL |