GW5074 (GW-5074; GW 5074) is a novel, potent and selective c-Raf inhibitor with potential neuroprotective activity. With an IC50 of 9 nM, it inhibits c-Raf but has no effect on JNK1/2/3, MKK6/7, CDK1/2, MEK1, c-Src, VEGFR2, p38 MAP, or c-Fms. In an animal model of Huntington's disease, GW-5074 is able to stop neurodegeneration and enhance behavioral outcomes. For the treatment of neurodegenerative diseases in people, GW 5074 has therapeutic potential.
Physicochemical Properties
| Molecular Formula | C15H8BR2INO2 | |
| Molecular Weight | 520.94 | |
| Exact Mass | 518.796 | |
| Elemental Analysis | C, 34.58; H, 1.55; Br, 30.68; I, 24.36; N, 2.69; O, 6.14 | |
| CAS # | 220904-83-6 | |
| Related CAS # | (Z)-GW 5074;1233748-60-1 | |
| PubChem CID | 5924208 | |
| Appearance | Light yellow to yellow solid powder | |
| Density | 2.2±0.1 g/cm3 | |
| Boiling Point | 561.4±50.0 °C at 760 mmHg | |
| Flash Point | 293.3±30.1 °C | |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C | |
| Index of Refraction | 1.790 | |
| LogP | 6.36 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 2 | |
| Rotatable Bond Count | 1 | |
| Heavy Atom Count | 21 | |
| Complexity | 447 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | IC1C([H])=C([H])C2=C(C=1[H])/C(=C(\[H])/C1C([H])=C(C(=C(C=1[H])Br)O[H])Br)/C(N2[H])=O |
|
| InChi Key | LMXYVLFTZRPNRV-KMKOMSMNSA-N | |
| InChi Code | InChI=1S/C15H8Br2INO2/c16-11-4-7(5-12(17)14(11)20)3-10-9-6-8(18)1-2-13(9)19-15(10)21/h1-6,20H,(H,19,21)/b10-3- | |
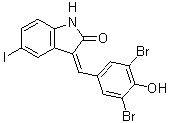
| Chemical Name | (3Z)-3-[(3,5-dibromo-4-hydroxyphenyl)methylidene]-5-iodo-1H-indol-2-one | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
C-Raf (IC50 = 9 nM) c-Raf kinase (IC50: ~100 nM) [1] - c-Raf kinase (IC50: ~80 nM) [2] - c-Raf kinase (IC50: ~120 nM) [3] |
| ln Vitro |
GW5074 is a potent and specific inhibitor of c-Raf with an IC50 value of 9 nM; it has no effect in vitro on MKK6, MKK7, p38 MAP kinase, or cdks. However, treatment with GW5074 in neuronal cultures allows for the accumulation of activating modifications on both c-Raf and B-Raf. In cerebellar granule neurons, GW5074 does not require MEK or ERK to prevent LK-induced apoptosis. Although apoptosis is inhibited by GW5074 through an Akt-independent mechanism, Akt activity is delayed during this process. Nuclear factor-kappa B, c-jun, and Ras are all impacted by GW5074. In granule cells and other neuronal types, GW5074 prevents neurotoxins from causing cell death. [1] In PC12 neuroblastoma cells, treatment with GW5074 at concentrations ranging from 0.1 μM to 1 μM for 24 hours resulted in a concentration-dependent inhibition of c-Raf kinase activity, which was evidenced by the decreased phosphorylation level of the downstream effector ERK1/2 (detected via Western blot). At 0.5 μM, GW5074 reduced the p-ERK1/2 level by approximately 45% compared with the control group; when the concentration reached 1 μM, the reduction rate increased to about 70% [1] - In vascular smooth muscle cells (VSMC), GW5074 showed antiproliferative activity. Incubating VSMC with 1 μM GW5074 for 48 hours inhibited cell proliferation by ~50%, while 5 μM GW5074 further suppressed proliferation by ~80% (evaluated by MTT assay). Additionally, treating VSMC with 2 μM GW5074 for 12 hours decreased the phosphorylation of ERK1/2 by around 60% [2] - In A549 human lung adenocarcinoma epithelial cells stimulated with lipopolysaccharide (LPS, 1 μg/mL), treatment with 5 μM GW5074 for 24 hours significantly inhibited the release of the pro-inflammatory cytokine IL-8. ELISA results indicated that the IL-8 level in the culture supernatant of drug-treated cells was ~60% lower than that in LPS-stimulated control cells. Quantitative real-time PCR (qPCR) analysis further revealed that 5 μM GW5074 reduced the IL-8 mRNA expression by ~50% in LPS-treated A549 cells [3] |
| ln Vivo |
GW5074 is preventative in a Huntington's disease experimental in vivo model. In mice, 3-NP-induced extensive bilateral striatal lesions were completely stopped by GW5074 (5 mg/Kg).[1] In mice, GW5074 reduces the airway hyperresponsiveness brought on by sidestream smoke.[3] In a mouse model of LPS-induced acute lung injury (ALI), intraperitoneal administration of GW5074 at a dose of 10 mg/kg once daily for 3 consecutive days (starting 1 hour before LPS challenge) alleviated lung inflammation. Histopathological analysis showed that the lung inflammation score of the GW5074-treated group was ~40% lower than that of the LPS-treated control group. Moreover, ELISA detection of lung tissue homogenates demonstrated that the IL-8 content in the lungs of GW5074-treated mice was ~50% less than that in control mice [3] |
| Enzyme Assay |
The Kinase Profiling service of Upstate Biotechnology typically uses purified kinase and synthetic substrates under standard conditions to perform in vitro kinase assays. In a nutshell, 5–10 mU of purified kinase are utilized for each assay. For GSK3β, cdk1, cdk2, cdk3, and cdk5, the kinase is incubated with 1 μM GW5074 for 40 min at room temperature in a buffer containing 8 mM MOPS, pH 7.2, 0.2 mM EDTA, 10 mM magnesium acetate, and [c- 33P-ATP]. With an aliquot spotted on P30 filters, washed in 50 mM phosphoric acid, and scintillation counted, 33P incorporation is measured to determine the amount of kinase activity. For [c- 33P-ATP], 50 mM Tris pH 7.5, 0.1 mM EGTA, 10 mM magnesium acetate, and [c- Raf, JNK1, JNK2, JNK3, MEK1, MKK6, and MKK7, the buffer's component parts are. These are the peptide substrates employed: MBP concentrations for c-Raf are 0.66 mg/mL, cdks are 0.1 mg/mL, JNKs are 3 μM ATF2, MEK1 is 1 μM MAPK2, MKK6 is 1 μM SAPK2a, and MKK7 is 2 μM JNK1α. For the c-Raf kinase activity assay, the reaction system (with a total volume of 50 μL) contained recombinant human c-Raf kinase, a synthetic peptide substrate (with a sequence corresponding to the ERK phosphorylation site), ATP (at a final concentration of 100 μM), and GW5074 at different concentrations (0.01 μM to 1 μM). The reaction mixture was incubated at 30°C for 60 minutes, and then a stop solution was added to terminate the reaction. The amount of phosphorylated substrate was detected using a fluorescence-based method, with an excitation wavelength of 485 nm and an emission wavelength of 535 nm. The IC50 value of GW5074 against c-Raf kinase was calculated based on the dose-response curve [1] - The c-Raf kinase activity assay was conducted in a 96-well plate. Each well contained 20 ng of recombinant c-Raf kinase, 50 μM ATP, 2 μg of substrate peptide, and GW5074 at concentrations ranging from 0.05 μM to 2 μM. The plate was incubated at 37°C for 45 minutes, followed by the addition of a kinase detection reagent. After incubating at room temperature for 30 minutes, the absorbance at 450 nm was measured using a microplate reader. The inhibition rate of GW5074 on c-Raf kinase activity was calculated by comparing the absorbance of the drug-treated group with that of the vehicle control group, and the IC50 was determined accordingly [2] - To measure c-Raf kinase activity, the assay was performed in a buffer containing 25 mM Tris-HCl (pH 7.5), 5 mM MgCl2, 1 mM DTT, 0.1 mg/mL BSA, 15 ng/well of recombinant c-Raf kinase, 75 μM ATP, 1.5 μg of substrate peptide, and GW5074 at various concentrations (0.02 μM to 1.5 μM). The reaction was initiated by adding ATP and incubated at 30°C for 50 minutes. Then, 25 μL of 2× SDS sample buffer was added to stop the reaction, and the phosphorylated substrate was detected via Western blot using a phospho-specific antibody against the substrate peptide. The band intensity was quantified with image analysis software, and the IC50 of GW5074 was calculated [3] |
| Cell Assay |
HCA is diluted from 100-fold concentrated solutions with a pH of 7.5. When cortical neurons are exposed to HCA, GW5074 is added in order to assess its effects on the cytotoxicity caused by HCA. 24 hours later, viability is evaluated. PC12 cells were cultured in DMEM medium supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin at 37°C in a humidified atmosphere with 5% CO2. When the cells reached 70%-80% confluence, they were seeded into 6-well plates at a density of 2×105 cells/well. After 24 hours of adherence, the medium was replaced with fresh medium containing GW5074 at concentrations of 0.1 μM, 0.5 μM, and 1 μM (the vehicle control contained an equal volume of DMSO). The cells were incubated for another 24 hours, then harvested and lysed with RIPA buffer containing protease and phosphatase inhibitors. The protein concentration of the cell lysate was determined using a BCA assay, and equal amounts of protein were subjected to Western blot analysis to detect the levels of phosphorylated ERK1/2 (p-ERK1/2) and total ERK1/2 [1] - Vascular smooth muscle cells (VSMC) were isolated from rat aorta and cultured in DMEM medium with 15% FBS and 1% antibiotics at 37°C with 5% CO2. For the proliferation assay, VSMC were seeded into 96-well plates at a density of 5×103 cells/well. After 24 hours of culture, the medium was changed to serum-free DMEM to synchronize the cells for 12 hours. Then, the cells were treated with GW5074 at concentrations of 0.5 μM, 1 μM, 5 μM, and 10 μM (vehicle control: DMSO) in DMEM containing 10% FBS. After 48 hours of incubation, 20 μL of MTT solution (5 mg/mL) was added to each well, and the plate was incubated at 37°C for another 4 hours. The supernatant was discarded, and 150 μL of DMSO was added to dissolve the formazan crystals. The absorbance at 570 nm was measured using a microplate reader, and the cell viability was calculated relative to the control group [2] - A549 cells were maintained in RPMI 1640 medium supplemented with 10% FBS and 1% penicillin-streptomycin at 37°C in a 5% CO2 incubator. For the inflammation-related experiment, A549 cells were seeded into 24-well plates at a density of 1×105 cells/well. After 24 hours of culture, the cells were pretreated with GW5074 at concentrations of 1 μM, 3 μM, and 5 μM for 1 hour, followed by stimulation with LPS (1 μg/mL) for 24 hours. The culture supernatant was collected to detect the IL-8 level using an ELISA kit according to the manufacturer's protocol. For qPCR analysis, total RNA was extracted from the cells using an RNA extraction kit, reverse-transcribed into cDNA, and qPCR was performed with IL-8-specific primers. The relative expression level of IL-8 mRNA was calculated using the 2-ΔΔCt method, with GAPDH as the internal reference gene [3] |
| Animal Protocol | On the sixth day after receiving injections of saline, 3-NP, or a combination of 3-NP and GW5074 over the previous five days, the locomotor activity of mice is measured using the Tru-Scan® activity monitoring system (7 mice per group). The animal is put in a Perspex arena that measures 25.9 x 25.9 cm and has infrared beams that are spaced 0.6 inches apart in the X-Y plane. A second infrared beam system at the Z plane, positioned 2.54 cm above the X-Y plane, is also installed in the arena. In this system, interruptions in the 1717-grid system caused by the infrared beams in both the X-Y and Z planes are used to accurately measure animal movement. The animal is allowed to stay in the arena for 15 minutes, during which time data is collected using a Pentium PC running Tm Scan 99 software and the Tru Scan Line interface box. We choose the following behavioral criteria: (i) Total movement distance: the total of all vectored A-Y coordinate changes in the floor plane; (ii) mean velocity: the mean velocity of all X-Y coordinate change defined movements; and (iii) total vertical plane entries: the total number of times any part of the animal entered the vertical plane (Z plane). Each movement in the floor plane is a series of coordinate changes with no rest for at least one sample interval. |
| References |
[1]. Neurochem . 2004 Aug;90(3):595-608. [2]. Eur J Pharmacol . 2006 Jul 1;540(1-3):57-9. [3]. Respir Res, 2008, 9(1), 71. |
| Additional Infomation |
In this study, GW5074 was used as a specific small-molecule inhibitor of c-Raf kinase to investigate the role of the c-Raf/ERK signaling pathway in regulating the signaling of neuroblastoma cells. The results suggested that the inhibition of c-Raf by GW5074 could downregulate the ERK signaling pathway, which might be involved in the modulation of neurocellular functions [1] - This study used GW5074 to explore the effect of c-Raf kinase inhibition on VSMC proliferation. The findings indicated that the inhibition of c-Raf mediated by GW5074 could suppress VSMC proliferation, implying that c-Raf inhibitors might have a potential therapeutic role in vascular proliferative diseases [2] - In this research, GW5074 was employed to study the involvement of the c-Raf/ERK pathway in LPS-induced lung inflammation. The results demonstrated that GW5074 could inhibit the LPS-induced production of IL-8 in A549 cells and alleviate lung inflammation in mice with ALI, suggesting that targeting the c-Raf/ERK pathway using GW5074 might be a potential strategy for the treatment of inflammatory lung diseases [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.80 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 30% propylene glycol, 5% Tween 80, 65% D5W: 30mg/mL (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9196 mL | 9.5980 mL | 19.1961 mL | |
| 5 mM | 0.3839 mL | 1.9196 mL | 3.8392 mL | |
| 10 mM | 0.1920 mL | 0.9598 mL | 1.9196 mL |