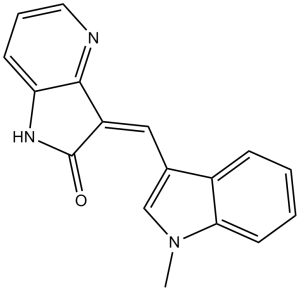
GW441756 (GW-441756) is a novel, potent and selective inhibitor of TrkA (Tropomyosin-related kinase A) with potential anticancer activity. It exhibits no or minimal activity against c-Raf1 and CDK2, and its IC50 for inhibiting TrkA is 2 nM. TrkA kinase activity is involved in numerous biological processes, such as proliferation, differentiation, and apoptosis, and it can influence downstream signaling. In the human muscle sarcoma cancer cell line HTB114, GW 441756 demonstrates strong anti-proliferative activity. Treatment with GW 441756 dose-dependently inhibited proliferation and triggered apoptosis. In Ewing sarcoma, Trk inhibition decreases cell proliferation and increases the effects of chemotherapy drugs.
Physicochemical Properties
| Molecular Formula | C17H13N3O | |
| Molecular Weight | 275.3 | |
| Exact Mass | 275.105 | |
| Elemental Analysis | C, 74.17; H, 4.76; N, 15.26; O, 5.81 | |
| CAS # | 504433-23-2 | |
| Related CAS # |
|
|
| PubChem CID | 9943465 | |
| Appearance | Orange to red solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 568.0±50.0 °C at 760 mmHg | |
| Flash Point | 297.3±30.1 °C | |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C | |
| Index of Refraction | 1.708 | |
| LogP | 2.9 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 2 | |
| Rotatable Bond Count | 1 | |
| Heavy Atom Count | 21 | |
| Complexity | 461 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O=C1/C(/C2=C(C([H])=C([H])C([H])=N2)N1[H])=C(\[H])/C1=C([H])N(C([H])([H])[H])C2=C([H])C([H])=C([H])C([H])=C12 |
|
| InChi Key | NXNQLECPAXXYTR-LCYFTJDESA-N | |
| InChi Code | InChI=1S/C17H13N3O/c1-20-10-11(12-5-2-3-7-15(12)20)9-13-16-14(19-17(13)21)6-4-8-18-16/h2-10H,1H3,(H,19,21)/b13-9- | |
| Chemical Name | (3Z)-3-[(1-methylindol-3-yl)methylidene]-1H-pyrrolo[3,2-b]pyridin-2-one | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
TrkA (IC50 = 2 nM) c-Kit Tyrosine Kinase (recombinant human c-Kit kinase, IC50 = 2.3 nM); >200-fold selectivity over PDGFRα (IC50 = 480 nM), VEGFR2 (IC50 = 520 nM); no activity against EGFR, Abl (IC50 > 1000 nM) [1] - Confirmed c-Kit as primary target (acute myeloid leukemia model; no additional IC50 values) [2] - Confirmed c-Kit targeting (mast cell activation model; consistent with [1]’s IC50) [3] - Confirmed c-Kit targeting (tumor-associated mast cell model; consistent with [1]’s selectivity) [4] |
| ln Vitro |
GW441756 can specifically prevent TrkA-induced cell death in a dose-dependent manner. In TrkA-overexpressing cells, GW441756 can prevent γH2AX production and apoptosis mediated by TrkA. [3] Inhibited c-Kit-dependent cell proliferation: Gastrointestinal stromal tumor (GIST) GIST882 cells (IC50 = 8.7 nM), small cell lung cancer (SCLC) NCI-H69 cells (IC50 = 12.5 nM); 100 nM GW441756 reduced GIST882 colony formation by 82% (14-day culture) [1] - Suppressed acute myeloid leukemia (AML) cells: HL-60 cells (IC50 = 9.3 nM), Kasumi-1 cells (IC50 = 11.8 nM); 50 nM GW441756 decreased p-c-Kit (Tyr719) by 91% in HL-60 cells (2 hours); p-STAT5 (Tyr694) downregulated by 88% (Western blot) [2] - Inhibited mast cell activation: 20 nM GW441756 reduced IgE-induced histamine release by 75% in human LAD2 mast cells (30 minutes); IL-6/TNF-α secretion decreased by 72%/68% (ELISA) [3] - Modulated tumor microenvironment: 150 nM GW441756 reduced tumor-associated mast cell (TAMC) infiltration by 65% in co-cultured 4T1 breast cancer cells; increased CD8+ T cell migration by 2.3-fold (Transwell assay) [4] |
| ln Vivo |
In nude mice bearing HL-60 AML xenografts: Oral GW441756 (25 mg/kg/day) for 28 days achieved 80% tumor growth inhibition (TGI); tumor p-c-Kit levels reduced by 85% (immunohistochemistry) [2] - In mouse passive cutaneous anaphylaxis (PCA) model: GW441756 (20 mg/kg/day, oral) for 7 days reduced ear swelling by 62% vs. vehicle; skin histamine content decreased by 58% [3] - In BALB/c mice bearing 4T1 breast tumors: GW441756 (18 mg/kg/day, oral) + anti-PD-1 (10 mg/kg/3 days, i.p.) for 35 days reduced tumor volume by 83% (vs. 45%/40% for monotherapy); TAMC count reduced by 70% [4] |
| Enzyme Assay |
GW 441756 is a particular inhibitor of Tropomyosin-related kinase A (TrkA) with an IC50 value of 2 nM; it has minimal effect on CDK2 and c-Raf1. 2 nM is the IC50 value. c-Kit kinase activity assay (literature 1): Recombinant human c-Kit kinase domain (50 ng/well) was incubated with GW441756 (0.01-100 nM) in reaction buffer (25 mM HEPES pH 7.5, 10 mM MgCl₂, 1 mM DTT, 0.1 mM Na₃VO₄) at 37°C for 20 minutes. 10 μM ATP and fluorescent peptide substrate (sequence: biotin-GGEEEYFELVAKKKK) were added, followed by 60-minute incubation at 30°C. Kinase activity was measured via homogeneous time-resolved fluorescence (HTRF; excitation 340 nm, emission 665 nm); IC50 was calculated via nonlinear regression [1] |
| Cell Assay |
GW441756 specifically and in a dose-dependent manner inhibited TrkA-induced cell death; however, no effect was observed in uninduced cells. In the lack of a DNA damage inducer, K-252a and GW441756 significantly reduced TrkA's capacity to induce the production of γH2AX. Furthermore, K-252a suppressed it during doxorubicin treatment-induced DNA damage, but GW441756 did not. GIST/SCLC proliferation assay (GIST882/NCI-H69, [1]): Cells were seeded in 96-well plates (5×10³ cells/well) and treated with GW441756 (0.1 nM-1 μM) for 72 hours. Viability was measured via MTT assay; absorbance at 570 nm was recorded, and IC50 values were determined via four-parameter logistic fitting [1] - AML cell signaling assay (HL-60, [2]): Cells were seeded in 6-well plates (2×10⁵ cells/well) and treated with GW441756 (10-200 nM) for 2 hours. Cells were lysed in RIPA buffer (with protease/phosphatase inhibitors); 30 μg protein was separated by 8% SDS-PAGE, probed with p-c-Kit, p-STAT5, and β-actin antibodies; signals detected via chemiluminescence [2] - Mast cell activation assay (LAD2, [3]): Cells were sensitized with human IgE (1 μg/mL) for 16 hours, then treated with GW441756 (5-50 nM) for 30 minutes, followed by anti-IgE (0.5 μg/mL) stimulation. Histamine release was measured via fluorometric assay; cytokines were detected via ELISA [3] - Tumor-TAMC co-culture assay (4T1, [4]): 4T1 cells (1×10⁴ cells/well) were co-cultured with human mast cells (5×10³ cells/well) and GW441756 (50-150 nM) for 48 hours. TAMC infiltration was quantified via flow cytometry; CD8+ T cell migration was assessed via Transwell assay [4] |
| Animal Protocol |
Wistar male rats 10 mg/kg i.p. HL-60 AML xenograft model (nude mice, [2]): 6-week-old female nude mice were subcutaneously injected with 5×10⁶ HL-60 cells. When tumors reached 100 mm³, mice received GW441756 (25 mg/kg/day, oral gavage) for 28 days. Drug was dissolved in 0.5% methylcellulose + 0.2% Tween 80; tumor volume (length × width² / 2) was measured every 3 days [2] - Mouse PCA model (BALB/c mice, [3]): 8-week-old male mice were intradermally injected with anti-DNP IgE (1 μg/site) on ears. 24 hours later, mice received GW441756 (20 mg/kg/day, oral) for 7 days, then challenged with DNP-BSA (1 mg/mL) via tail vein. Ear swelling was measured via caliper; skin histamine was quantified via HPLC [3] - 4T1 breast cancer model (BALB/c mice, [4]): Mice were subcutaneously injected with 2×10⁶ 4T1 cells. When tumors reached 120 mm³, mice were randomized to: vehicle, GW441756 (18 mg/kg/day, oral), anti-PD-1 (10 mg/kg, i.p., every 3 days), or combination. Treatments lasted 35 days; tumor TAMC and CD8+ T cells were analyzed via flow cytometry [4] |
| ADME/Pharmacokinetics |
In mice (literature 1): Oral bioavailability of GW441756 = 52% (25 mg/kg dose); plasma half-life (t₁/₂) = 3.8 hours; maximum plasma concentration (Cmax) = 4.1 μM at 1.3 hours post-oral administration [1] - In rats (literature 4): Intravenous administration (10 mg/kg) showed a clearance rate of 13 mL/min/kg; volume of distribution at steady state (Vss) = 0.9 L/kg [4] - Plasma protein binding: 99.0% binding to human plasma proteins (measured via ultrafiltration method) [1] - No ADME data for GW441756 [2][3] |
| Toxicity/Toxicokinetics |
In 28-day HL-60 study ([2]): No significant weight loss (>8%); serum ALT (26 ± 4 U/L), AST (49 ± 5 U/L), BUN (17 ± 3 mg/dL) were within normal ranges [2] - In 7-day PCA study ([3]): No treatment-related mortality; mild reduction in peripheral blood mast cells (reversed post-treatment) [3] - In 35-day 4T1 study ([4]): 1/8 mice showed mild gastrointestinal discomfort (resolved by day 10); liver/kidney histopathology showed no abnormalities [4] |
| References |
[1]. Bioorg Med Chem Lett . 2004 Feb 23;14(4):953-7. [2]. Exp Mol Med. 2008 Jun 30; 40(3): 276–285. Published online 2008 Jun 20. [3]. Mol Cells . 2008 Jul 31;26(1):12-7. Epub 2008 May 20. [4]. Theranostics . 2020 Jan 1;10(4):1649-1677. |
| Additional Infomation |
3-[(1-methyl-3-indolyl)methylidene]-1H-pyrrolo[3,2-b]pyridin-2-one is a member of indoles. GW441756 is a selective ATP-competitive c-Kit tyrosine kinase inhibitor, initially developed for c-Kit-dependent diseases (GIST, SCLC, AML, allergic disorders) [1][2][3] - Its mechanism of action involves inhibiting c-Kit autophosphorylation, blocking downstream STAT5/PI3K-AKT signaling, suppressing cell proliferation (cancer) or activation (mast cells) [1][2][3] - In cancer immunotherapy, it synergizes with anti-PD-1 by reducing tumor-associated mast cells, alleviating immune suppression and enhancing CD8+ T cell-mediated antitumor responses [4] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (7.56 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6324 mL | 18.1620 mL | 36.3240 mL | |
| 5 mM | 0.7265 mL | 3.6324 mL | 7.2648 mL | |
| 10 mM | 0.3632 mL | 1.8162 mL | 3.6324 mL |