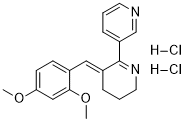
GTS-21, formerly known as DMBX-A, is a analogue of the natural product anabaseine that acts as a partial agonist at neural nicotinic acetylcholine receptors. It binds to both the α4β2 and α7 subtypes, but activates only the α7 to any significant extent. Both GTS-21 itself and its demethylated active metabolite 4-OH-GTS-21 display nootropic and neuroprotective effects, and GTS-21 is being investigated for the treatment of Alzheimer's disease, nicotine dependence, and, most significantly, for schizophrenia.
Physicochemical Properties
| Molecular Formula | C19H22CL2N2O2 |
| Molecular Weight | 381.3 |
| Exact Mass | 308.15248 |
| Elemental Analysis | C, 59.85; H, 5.82; Cl, 18.59; N, 7.35; O, 8.39 |
| CAS # | 156223-05-1 |
| Related CAS # | 148372-04-7;156223-05-1 (HCl); |
| PubChem CID | 6438361 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 485.6±45.0 °C at 760 mmHg |
| Melting Point | 216-217℃ (decomposition) |
| Flash Point | 247.5±28.7 °C |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C |
| Index of Refraction | 1.580 |
| LogP | 2.64 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 25 |
| Complexity | 447 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | COC1=CC(OC)=CC=C1/C=C2CCCN=C\2C3=CC=CN=C3.Cl[H].Cl[H] |
| InChi Key | 3-[(3E)-3-[(2,4-dimethoxyphenyl)methylidene]-5,6-dihydro-4H-pyridin-2-yl]pyridine dihydrochloride |
| InChi Code | BXKYFUGAAFLYJL-BXGYHSFXSA-N |
| Chemical Name | GTS-21 HCl GTS-21 hydrochloride GTS-21 GTS 21 GTS21 DMBX-A |
| Synonyms | GTS-21 HCl GTS-21 hydrochloride GTS-21 GTS 21 GTS21 DMBX-A |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In addition to binding to human α4β2 and a7 nAChR 18 times and 18 times lower than (-)-nicotine, respectively, GTS-21 also binds to human α4β2 nAChR (Ki=20 nM) 100 times stronger than it does to human α7-nAChR. Twice. 1]. |
| ln Vivo | The histological indicators of radiation-induced lung injury are lessened by GTS 21 (4 mg/kg; intraperitoneally; days 1, 3, 7, 14, and 21) [3]. |
| Animal Protocol |
Animal/Disease Models: C57BL6 mice were irradiated with 12 Gy to induce radiation-induced lung injury (RILI) mouse model [3] Doses: 4 mg/kg Route of Administration: intraperitoneal (ip) injection; results on days 1, 3, 7, 14 and 21 Experimental Results: Reduce pulmonary inflammatory infiltration and fibrosis in radiation-treated mice. |
| References |
[1]. Functional characterization of the novel neuronal nicotinic acetylcholine receptor ligand GTS-21 in vitro and in vivo. Pharmacol Biochem Behav. 1997;57(1-2):231-241. [2]. N-terminal domains in mouse and human 5-hydroxytryptamine3A receptors confer partial agonist and antagonist properties to benzylidene analogs of anabaseine. J Pharmacol Exp Ther. 2006;317(3):1276-1284. [3]. α7 nAchR agonist GTS 21 reduces radiation induced lung injury. Oncol Rep. 2018;40(4):2287-2297. |
Solubility Data
| Solubility (In Vitro) |
H2O : ~50 mg/mL (~131.13 mM) DMSO : ~16.5 mg/mL (~43.27 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.56 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.56 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.56 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 75 mg/mL (196.70 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6226 mL | 13.1130 mL | 26.2261 mL | |
| 5 mM | 0.5245 mL | 2.6226 mL | 5.2452 mL | |
| 10 mM | 0.2623 mL | 1.3113 mL | 2.6226 mL |