Physicochemical Properties
| Exact Mass | 428.147840 |
| Elemental Analysis | C, 58.81; H, 5.64; Cl, 8.27; F, 13.29; N, 6.53; O, 7.46 |
| CAS # | 1440950-06-0 |
| Related CAS # | 1440950-13-9;1440950-06-0 (HCl); |
| PubChem CID | 71584822 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.5±0.1 g/cm3 |
| Index of Refraction | 1.675 |
| InChi Key | SYMZWNMNMYVRSG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H23F3N2O2.ClH/c1-26-12-10-18(11-13-26)25-20(27)16-4-2-15(3-5-16)14-28-19-8-6-17(7-9-19)21(22,23)24;/h2-9,18H,10-14H2,1H3,(H,25,27);1H |
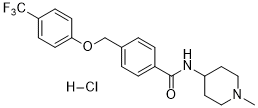
| Chemical Name | N-(1-methylpiperidin-4-yl)-4-[[4-(trifluoromethyl)phenoxy]methyl]benzamide;hydrochloride |
| Synonyms | GT-0198 hydrochloride; GT0198; GT-0198; GT-0198 HCl; GT-0198 hydrochloride; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Glycine transporter 2 (GlyT2) |
| ln Vitro | GT-0198 inhibited the function of glycine transporter 2 (GlyT2) in human GlyT2-expressing HEK293 cells and did not bind various major transporters or receptors of neurotransmitters in a competitive manner. Thus, GT-0198 is considered to be a comparatively selective GlyT2 inhibitor[1]. |
| ln Vivo | Intravenous, oral, and intrathecal injections of GT-0198 decreased the pain-related response in a model of neuropathic pain with partial sciatic nerve ligation. This result suggests that GT-0198 has an analgesic effect. The analgesic effect of GT-0198 was abolished by the intrathecal injection of strychnine, a glycine receptor antagonist. Therefore, GT-0198 is considered to exhibit its analgesic effect via the activation of a glycine receptor by glycine following presynaptic GlyT2 inhibition in the spinal cord. In summary, GT-0198 is a structurally novel GlyT2 inhibitor bearing a phenoxymethylbenzamide moiety with in vivo efficacy in behavioral models of neuropathic pain[1]. |
| Enzyme Assay |
Binding assays to various transporters and receptors of neurotransmitters[1] To evaluate the affinity of GT-0198 for various transporters and receptors, the rate of inhibition of the binding of a specific ligand to each transporter or receptor was assayed, as shown in Table 2. These assays were carried out in Sekisui Medical co., Ltd. Test substance concentration is 10 μM and positive substance concentration is 1 or 10 μM. Data are expressed as the mean values of duplicate samples. The inhibition ratios were calculated from “100 – binding ratio”. |
| Cell Assay |
Glycine uptake assays with GlyT1 and GlyT2 transfected human embryonic kidney 293 cells[1] A vector expressing human GlyT1 or GlyT2 was transfected into human embryonic kidney 293 (HEK 293) cells using Lipofectamine 2000. The transfected HEK293 cells were cultured in Dulbecco's Modified Eagle's Medium (DMEM) supplemented with 10% fetal bovine serum. Stable single-cell clones for hGlyT1 or hGlyT2 were isolated by selection with 500 μg/mL geneticin. Selected clones were evaluated in [3H]-glycine uptake assays. HEK293 cells were plated (6000 cells per well) in polylysine-coated 96-well CytoStar-T® plates and incubated overnight. The cells were then incubated at 37 °C for 2 h with 240 nM [3H]-glycine together with any compound being evaluated, in a total volume of 0.5 mL. At the end of the incubation, the radioactivity content of the wells was measured using Packard TopCount. |
| Animal Protocol |
Animals[1] Male ICR mice (5 weeks old at the start of experiments) were used. All mice were housed 3–5 animals per cage under a room temperature of 21°C–25 °C, humidity of 40%–70%, and 12-h light/dark cycle (light on at 7:00 AM) for at least 1 week before use. Mice had ad libitum access to food and water. All experiments were conducted according to the Guidelines for Animal Experiments, Research & Development Division, Toray Industries, Inc., and a pain test was performed according to the Guidelines on Ethical Standards for Investigation of Experimental Pain in Animals. Surgical operation[1] The mice were anesthetized with sodium pentobarbital (70 mg/kg, i.p.). We produced a partial sciatic nerve ligation (PSNL) model by tying a tight ligature with an 8–0 silk suture around approximately one-third to one-half the diameter of the sciatic nerve on the right side as described previously 17, 18. The sham-operated group was subjected to all procedures except ligation. In the present study, the pain test was performed 7 days after nerve ligation. Pain test[1] To evaluate the analgesic effect of the compounds, paw withdrawal responses were measured using a von Frey filament with a bending force of 0.16 g. The mice were placed individually in acrylic cages with wire mesh bottoms. After at least 60 min of acclimation, the von Frey filament was applied to the plantar surface of the hind paws for 3 s, and this was repeated 3 times. Each hind paw of the mice was tested individually. Paw withdrawal response to the von Frey filament was evaluated by scoring as follows: 0, no response; 1, slow and/or slight response to the stimulus; 2, quick withdrawal response away from the stimulus without flinching or licking; 3, intense withdrawal response away from the stimulus with brisk flinching and/or licking. The sum of 3 values served as the pain-related score. Drugs [1] For intravenous or intrathecal injection, GT-0198 was dissolved in saline and injected in a volume of 0.1 mL/10 g body weight (intravenously) or 5 μL/body (intrathecally). For oral administration, the compounds were dissolved in distilled water and injected in a volume of 0.1 mL/10 g body weight. |
| References | [1]. Analgesic effect of GT-0198, a structurally novel glycine transporter 2 inhibitor, in a mouse model of neuropathic pain. J Pharmacol Sci. 2015 Mar;127(3):377-81. |
| Additional Infomation | The GlyT2 inhibitor ALX-1393 significantly increased intercontraction interval and micturition pressure threshold in cyclophosphamide-treated rats. These results indicate that inhibition of GlyT2 leads to amelioration of cyclophosphamide-induced bladder overactivity and that GT-0198 may be a drug for the treatment of overactive bladder. In summary, we have shown that GT-0198 had an analgesic effect in an animal model of neuropathic pain and suggest that GT-0198 could reduce this pain by inhibiting spinal GlyT2. GT-0198 is a structurally novel compound, with demonstrated analgesic efficacy in a behavioral model of neuropathic pain. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |