GSK2837808A is a potent, selective, and an NADH-competitive inhibitor of lactate dehydrogenase A (LDHA) with IC50s of 1.9 and 14 nM for LDHA and LDHB, respectively. GSK2837808A rapidly and profoundly inhibited lactate production rates in multiple cancer cell lines including hepatocellular and breast carcinomas. Consistent with selective inhibition of LDHA, the most sensitive breast cancer cell lines to lactate inhibition in hypoxic conditions were cells with low expression of LDHB. GSK2837808A increased rates of oxygen consumption in hepatocellular carcinoma cells at doses up to 3 microM, while higher concentrations directly inhibited mitochondrial function. Analysis of more than 500 metabolites upon LDHA inhibition in Snu398 cells revealed that intracellular concentrations of glycolysis and citric acid cycle intermediates were increased, consistent with enhanced Krebs cycle activity and blockage of cytosolic glycolysis. Treatment with these compounds also potentiated PKM2 activity and promoted apoptosis in Snu398 cells.
Physicochemical Properties
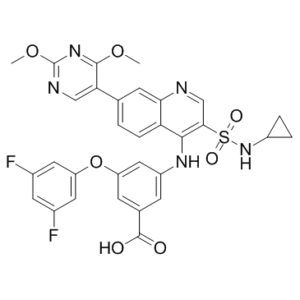
| Molecular Formula | C31H25F2N5O7S | |
| Molecular Weight | 649.63 | |
| Exact Mass | 649.144 | |
| CAS # | 1445879-21-9 | |
| Related CAS # |
|
|
| PubChem CID | 71533725 | |
| Appearance | Light yellow to yellow solid powder | |
| Density | 1.6±0.1 g/cm3 | |
| Boiling Point | 783.5±70.0 °C at 760 mmHg | |
| Flash Point | 427.6±35.7 °C | |
| Vapour Pressure | 0.0±2.9 mmHg at 25°C | |
| Index of Refraction | 1.710 | |
| LogP | 7.6 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 14 | |
| Rotatable Bond Count | 11 | |
| Heavy Atom Count | 46 | |
| Complexity | 1140 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | RZBCPMYJIARMGV-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C31H25F2N5O7S/c1-43-29-25(14-35-31(37-29)44-2)16-3-6-24-26(9-16)34-15-27(46(41,42)38-20-4-5-20)28(24)36-21-7-17(30(39)40)8-22(13-21)45-23-11-18(32)10-19(33)12-23/h3,6-15,20,38H,4-5H2,1-2H3,(H,34,36)(H,39,40) | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | In certain cancer cell lines, such as those from hepatocellular carcinoma and breast cancer, GSK2837808A quickly and significantly reduces the rates at which lactate is produced. In thirty cancer cell lines with different levels of LDHA and LDHB expression, the potency of GSK2837808A varied from 400 nM to ineffective (EC50 reported as 30 μM). There was no correlation found between the potency of GSK2837808A and the levels of LDHA, LDHB, or total LDH expression. Under hypoxic conditions, GSK2837808A suppresses lactate synthesis, but at higher doses than in normoxic conditions (EC50=10 μM). Furthermore, it lowers ECAR (EC50=10 μM). Multiple metabolic pathways in Snu398 cells are altered when LDH is inhibited by GSK2837808A [1]. |
| ln Vitro |
In certain cancer cell lines, such as those from hepatocellular carcinoma and breast cancer, GSK2837808A quickly and significantly reduces the rates at which lactate is produced. In thirty cancer cell lines with different levels of LDHA and LDHB expression, the potency of GSK2837808A varied from 400 nM to ineffective (EC50 reported as 30 μM). There was no correlation found between the potency of GSK2837808A and the levels of LDHA, LDHB, or total LDH expression. Under hypoxic conditions, GSK2837808A suppresses lactate synthesis, but at higher doses than in normoxic conditions (EC50=10 μM). Furthermore, it lowers ECAR (EC50=10 μM). Multiple metabolic pathways in Snu398 cells are altered when LDH is inhibited by GSK2837808A [1]. Compound 1 potently inhibits lactate production in a panel of cancer cell lines, with half maximal effective concentrations (EC50) ranging from 0.4 µM to >30 µM. Snu398 and HepG2 hepatocellular carcinoma cells (both with high LDHA and undetectable LDHB expression) were among the most sensitive, with lactate production EC50 values of 0.4 µM and 0.6 µM, respectively [1] In Snu398 cells, treatment with Compound 1 (1-3 µM) led to a dose-dependent increase in oxygen consumption rate (OCR) and a decrease in extracellular acidification rate (ECAR), indicating a metabolic shift from glycolysis to oxidative phosphorylation [1] Comprehensive metabolomic analysis in Snu398 cells treated with 10 µM of a related inhibitor (Compound 2) for 24 hours revealed profound alterations, including accumulation of glycolytic intermediates (some up to 40-fold), increased citric acid cycle intermediates, and upregulation of the pentose phosphate pathway [1] Compound 1 dose-dependently increased pyruvate kinase (PK) activity and promoted the formation of the active tetrameric form of PKM2 in Snu398 cells [1] Compound 1 inhibited the proliferation of Snu398 cells with an EC50 of 2.9 µM after 4-8 days of treatment and induced apoptosis, as evidenced by PARP cleavage at doses of 3-30 µM after 24 hours. Co-treatment with an NAD+ synthesis inhibitor (FK866) potentiated this effect [1] In breast cancer cell lines, sensitivity to Compound 1 in inhibiting lactate flux under conditions mimicking hypoxia/anoxia was greater in cells with low LDHB expression [1] |
| ln Vivo | Following intravenous infusion at a dose of 0.25 mg/kg in rats, the clearance rate of GSK2837808A was 69 mL/min/kg, exceeding the animal liver blood flow. Following oral administration of GSK2837808A at a dose of 50 mg/kg in rats and 100 mg/kg in mice, blood chemical levels were at or below the 2.5 ng/mL detection limit [1]. |
| Enzyme Assay |
An assay was developed where recombinant human LDHA or LDHB enzymes catalyzed the conversion of lactate to pyruvate. The level of NADH produced in this reaction was measured indirectly through a coupled enzymatic reaction involving diaphorase, which converts resazurin to resorufin. This assay was used for high-throughput screening and subsequent potency determination of identified hits. Hits were further evaluated to ensure they did not interfere with the coupling reaction and were tested for their ability to stabilize LDHA in a thermal shift assay. Compound 1 was identified as an NADH-competitive inhibitor from this campaign [1] |
| Cell Assay |
Lactate Production Assay: Cells were plated in 96-well plates and allowed to attach overnight. Compound dose responses were prepared and added to cells for a 1-hour pre-incubation. The medium was then replaced with low-glucose medium containing the same compound dilutions. After a 30-minute incubation, lactate concentration in the conditioned medium was quantified using RapidFire-mass spectrometry (MS) analysis. Cell viability in each well was assessed in parallel using a fluorescence-based viability assay, and lactate concentration was normalized to the viability signal [1] Metabolic Analysis (Metabolomics): Cells were plated in flasks and treated with either DMSO or 10 µM LDHA inhibitor for 24 hours. Conditioned medium was collected, and cells were washed, trypsinized, and pelleted. Both medium and cell pellets were submitted for MS-based analysis of over 500 metabolites, with samples prepared in quintuplicate [1] Oxygen Consumption Rate (OCR) and Extracellular Acidification Rate (ECAR) Measurement: Cells were seeded into specialized microplates and allowed to attach. Cells were equilibrated in bicarbonate-free medium. The rates of change in dissolved O2 (OCR) and pH (ECAR) in the medium surrounding the cells were measured in real-time using a flux analyzer. Compound doses were injected at indicated times during the measurement. For dose-response curves, basal levels were subtracted from values obtained after compound addition [1] Cell Proliferation and Survival Analysis: Cells were plated in multi-well plates and treated with compound or DMSO. After 4 to 8 days of incubation under specified oxygen conditions, adherent cells were trypsinized, counted, and viability was assessed using trypan blue exclusion with an automated cell counter [1] Pyruvate Kinase (PK) Activity Assay: Cells were treated with compound for 6 hours, washed, and homogenized. Cleared homogenates were used to measure PK activity with a commercial activity kit, normalized to total protein concentration [1] |
| Animal Protocol |
Pharmacokinetic Study: Compound 1 was administered to male Sprague-Dawley rats via intravenous (IV) infusion over 120 minutes into a femoral vein. Arterial blood samples were collected over time for pharmacokinetic analysis. Oral dosing was also performed in rats and mice [1] |
| ADME/Pharmacokinetics |
Following IV infusion at 0.25 mg/kg in rats, the clearance of Compound 1 was 69 mL/min/kg, which exceeds hepatic blood flow [1] Oral dosing at 50 mg/kg in rats or 100 mg/kg in mice resulted in blood compound levels at or below the detection limit of 2.5 ng/mL [1] The compound exhibited high plasma protein binding (98.7% in human plasma and 98.8% in rat plasma as determined by equilibrium dialysis) [1] |
| Toxicity/Toxicokinetics |
At doses of 10 µM and higher, Compound 1 exhibited direct inhibitory effects on mitochondrial function in permeabilized cell assays, which are likely not mediated by LDH inhibition. However, the intracellular unbound fraction available to cause this off-target effect in intact cells is likely limited due to high cytosolic protein binding [1] |
| References |
[1]. Quinoline 3-sulfonamides inhibit lactate dehydrogenase A and reverse aerobic glycolysis in cancer cells. Cancer Metab. 2013 Sep 6;1(1):19. |
| Additional Infomation |
Compound 1 is a representative of a series of quinoline-3-sulfonamides identified as potent, selective, and NADH-competitive inhibitors of LDHA. These inhibitors rapidly reverse the aerobic glycolysis (Warburg effect) phenotype in sensitive cancer cells, leading to metabolic reprogramming, impaired proliferation, and induction of apoptosis. However, optimization for high enzymatic potency and selectivity resulted in poor pharmacokinetic properties (high clearance, low oral exposure), precluding further in vivo therapeutic development with this specific chemical series [1] . |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.85 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5393 mL | 7.6967 mL | 15.3934 mL | |
| 5 mM | 0.3079 mL | 1.5393 mL | 3.0787 mL | |
| 10 mM | 0.1539 mL | 0.7697 mL | 1.5393 mL |