GSK-J2 is the isomer of GSK-J1 without any specific activity. GSKJ1 (GSK-J1) is a novel, highly selective and potent inhibitor of histone demethylase H3K27me3/me2-demethylases JMJD3/KDM6B and UTX/KDM6A. It that has significant activity (IC50 60 nM for human JmjD3) in vitro and in cell assays using an ester prodrug derivative (GSK-J4: 1 µM < IC50 < 10 µM; e.g. 9 µM in primary human macrophages). The KDM5/JARID1 family of Fe(II)- and α-ketoglutarate-dependent demethylases remove methyl groups from tri- and dimethylated lysine 4 of histone H3. Accumulating evidence from primary tumors and model systems supports a role for KDM5A (JARID1A/RBP2) and KDM5B (JARID1B/PLU1) as oncogenic drivers. The pyridine regio-isomer GSK-J2 displays significantly less on-target activity (IC50 > 100 µM for human JmjD3) and thus can be used as control for target effects in vitro, and as ester derivative (GSK-J5) in cells. Recent data has shown that GSK-J1 also shows some activity (IC50 950 nM for Jarid1b, IC50 1.76 uM for Jarid1c) against H3K4me3/2/1 demethylases.
Physicochemical Properties
| Molecular Formula | C22H23N5O2 | |
| Molecular Weight | 389.45 | |
| Exact Mass | 389.185 | |
| CAS # | 1394854-52-4 | |
| Related CAS # | GSK-J1;1373422-53-7;GSK-J4;1373423-53-0;GSK-J5;1394854-51-3;GSK-J1 lithium salt | |
| PubChem CID | 73010924 | |
| Appearance | White to off-white solid powder | |
| LogP | 1.833 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 7 | |
| Rotatable Bond Count | 6 | |
| Heavy Atom Count | 29 | |
| Complexity | 517 | |
| Defined Atom Stereocenter Count | 0 | |
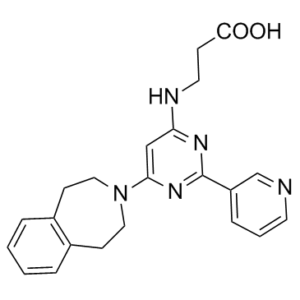
| SMILES | O=C(O)CCNC1=NC(C2=CC=CN=C2)=NC(N3CCC(C=CC=C4)=C4CC3)=C1 |
|
| InChi Key | LJIFOCRGDDQFJF-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C22H23N5O2/c28-21(29)7-11-24-19-14-20(26-22(25-19)18-6-3-10-23-15-18)27-12-8-16-4-1-2-5-17(16)9-13-27/h1-6,10,14-15H,7-9,11-13H2,(H,28,29)(H,24,25,26) | |
| Chemical Name | 3-[[2-pyridin-3-yl-6-(1,2,4,5-tetrahydro-3-benzazepin-3-yl)pyrimidin-4-yl]amino]propanoic acid | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | GSK-J2 is an isomer of GSK-J1, and it has weak activity (IC50 of > 100 μM and 49 μM, respectively) towards KDM6A and KDM6B[1]. | ||
| ln Vivo |
|
||
| Animal Protocol |
|
||
| References | [1]. Heinemann B, et al. Inhibition of demethylases by GSK-J1/J4. Nature. 2014 Oct 2;514(7520):E1-2 | ||
| Additional Infomation | 3-[[2-(3-pyridinyl)-6-(1,2,4,5-tetrahydro-3-benzazepin-3-yl)-4-pyrimidinyl]amino]propanoic acid is an organonitrogen heterocyclic compound. |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5677 mL | 12.8386 mL | 25.6772 mL | |
| 5 mM | 0.5135 mL | 2.5677 mL | 5.1354 mL | |
| 10 mM | 0.2568 mL | 1.2839 mL | 2.5677 mL |