Physicochemical Properties
| Molecular Formula | C28H19FN2O4 |
| Molecular Weight | 466.459870576859 |
| Exact Mass | 466.132 |
| CAS # | 916232-21-8 |
| PubChem CID | 121513876 |
| Appearance | Pink to red solid powder |
| LogP | 5.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 35 |
| Complexity | 844 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C(C1C=CC(N2C(=O)C(=CC3=CC=C(C4C(F)=CC=CC=4)O3)C(CC3C=CC=CC=3)=N2)=CC=1)O |
| InChi Key | IZPMWFSVTDOCDI-HAVVHWLPSA-N |
| InChi Code | InChI=1S/C28H19FN2O4/c29-24-9-5-4-8-22(24)26-15-14-21(35-26)17-23-25(16-18-6-2-1-3-7-18)30-31(27(23)32)20-12-10-19(11-13-20)28(33)34/h1-15,17H,16H2,(H,33,34)/b23-17+ |
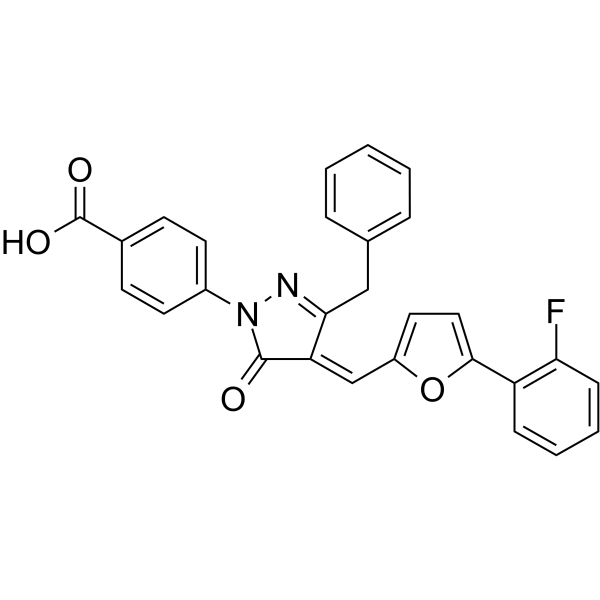
| Chemical Name | 4-[(4E)-3-benzyl-4-[[5-(2-fluorophenyl)furan-2-yl]methylidene]-5-oxopyrazol-1-yl]benzoic acid |
| Synonyms | GS143; GS 143; GS-143 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In vitro, Th2 cells are resistant to factor-induced apoptosis when exposed to GS143 (10 μM or 20 μM), but not Th1 cells. In a dose-dependent manner, GS143 strongly suppresses the Th2 pathway's ability to produce IL-4 [1]. TNFα-induced NF-κB regulation is inhibited by GS143, with an average IC50 value of 10.5 μM. With an IC50 value of 6.1 μM, GS143 also suppresses TNFα-induced ICAM-1 expression in HT-29 circulation. LPS-activated THP-1 cells showed inhibited production of TNFα and IL-1β, with IC50 values of 2.1 μM and 5.3 μM, respectively. p53 and β-catenin are not inhibited by GS143[2]. |
| ln Vivo | Intranasal scent administered once to BALB/c mice containing 16–32 μg/body of GS143 causes transcription factor receptors to become activated by NF-κB. Additionally, GS143 therapy causes the development of Th2 cytokines and eotaxin in the airways, as well as transcription factor activation of eosinophils and eosinophils into the airways [1]. |
| Animal Protocol |
Animal/Disease Models: balb/c (Bagg ALBino) mouse (7-8 weeks) injected with ovalbumin [1] Doses: 16 μg/body or 32 μg/body Route of Administration: intranasal administration; primary Experimental Results:inhibition of lungs of sensitized mice Antigen-induced NF-κB activation. |
| References |
[1]. GS143, an IkappaB ubiquitination inhibitor, inhibits allergic airway inflammation in mice. Biochem Biophys Res Commun. 2008 Sep 26;374(3):507-11. [2]. A novel small-molecule inhibitor of NF-kappaB signaling. Biochem Biophys Res Commun. 2008 Apr 18;368(4):1007-13. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~37.5 mg/mL (~80.39 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.08 mg/mL (4.46 mM) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 2: 1.88 mg/mL (4.03 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 18.8 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1438 mL | 10.7190 mL | 21.4381 mL | |
| 5 mM | 0.4288 mL | 2.1438 mL | 4.2876 mL | |
| 10 mM | 0.2144 mL | 1.0719 mL | 2.1438 mL |