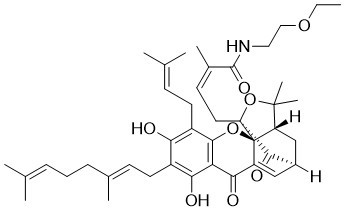
GNA002 is a novel and potent EZH2 (Enhancer of zeste homolog 2) inhibitor (IC50 of 1.1 μM) with anticancer activity. It acts by covalently binding to cysteine residues of EZH2 and triggering its ubiquitination and subsequent degradation by the protein quality control E3 ligase, CHIP.
Physicochemical Properties
| Molecular Formula | C42H55NO8 |
| Molecular Weight | 701.888013124466 |
| Exact Mass | 701.392 |
| CAS # | 1385035-79-9 |
| PubChem CID | 162642890 |
| Appearance | Light yellow to yellow ointment |
| LogP | 7.9 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 14 |
| Heavy Atom Count | 51 |
| Complexity | 1530 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CCOCCNC(=O)/C(=C/CC12C(=O)C3CC(C14C(=C3)C(=O)C5=C(C(=C(C(=C5O4)CC=C(C)C)O)C/C=C(\C)/CCC=C(C)C)O)C(O2)(C)C)/C |
| InChi Key | HJJVIXXMFVHPER-PIZOZCJOSA-N |
| InChi Code | InChI=1S/C42H55NO8/c1-10-49-21-20-43-39(48)27(7)18-19-41-38(47)28-22-31-36(46)33-35(45)29(17-15-26(6)13-11-12-24(2)3)34(44)30(16-14-25(4)5)37(33)50-42(31,41)32(23-28)40(8,9)51-41/h12,14-15,18,22,28,32,44-45H,10-11,13,16-17,19-21,23H2,1-9H3,(H,43,48)/b26-15+,27-18+ |
| Chemical Name | (E)-4-[7-[(2E)-3,7-dimethylocta-2,6-dienyl]-6,8-dihydroxy-17,17-dimethyl-5-(3-methylbut-2-enyl)-10,14-dioxo-3,16-dioxapentacyclo[11.4.1.02,11.02,15.04,9]octadeca-4,6,8,11-tetraen-15-yl]-N-(2-ethoxyethyl)-2-methylbut-2-enamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | The proliferation of multiple nail lines is significantly inhibited by GNA002 (10 μM; 72 hours); the IC50s for MV4-11 and RS4-11, respectively, are 0.070 μM and 0.103 μM. In the case of human cancer, GNA002 (2 μM; 24 hours) and GNA002 (0.1-4 μM; 48 hours) both efficiently inhibit the H3K27 trimethylation of Cal-27 in the head and neck bones caused by EZH2 [1]. The capacity of cells to cause cell death is greater [1]. Examine[1] |
| ln Vivo | Both the H3K27Me3 in tumor tissue and the Cal-27-derived tumor volume were markedly decreased by GNA002 (Manhattan; 100 mg/kg; daily). Additionally, GNA002 levels were markedly suppressed in the periphery of Daudi and Pfeiffer cells, which are xenografted A549 lung cancer cells. At least in xenograft experimental models, GNA002 reduces the aberrant core function of EZH2, which in turn inhibits tumor growth in vivo [1]. |
| Cell Assay |
Test[1] Cell Types: Various cancer cell lines Tested Concentrations: 10 μM Incubation Duration: 72 hrs (hours) Experimental Results: Inhibits the proliferation of various cancer cell lines, the IC50 of MV4-11 and RS4-11 are 0.070 μM and 0.103 μM respectively. Apoptosis analysis [1] Cell Types: HN-4 and Cal-27 head and neck cancer cells Tested Concentrations: 2 μM Incubation Duration: 24 hrs (hours) Experimental Results: Induction of apoptosis in human cancer cells. Western Blot Analysis[1] Cell Types: Cal-27 Head and Neck Cancer Cells Tested Concentrations: 0.1, 0.2, 0.5, 1, 2, 4 μM Incubation Duration: 48 hrs (hours) Experimental Results: diminished H3K27Me3 levels. |
| Animal Protocol |
Animal/Disease Models: Male BALB/C nude mice, 30-35 days old, weighing 18-22 g, carrying Cal-27 xenograft tumors [1] Doses: 100 mg/kg Doses: oral; daily Experimental Results: Cal-27 The size and weight of tumors formed by cells are diminished. |
| References |
[1]. A covalently bound inhibitor triggers EZH2 degradation through CHIP-mediated ubiquitination. EMBO J. 2017 May, 36(9):1243-1260. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~25 mg/mL (~35.62 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (1.78 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 1.25 mg/mL (1.78 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.4247 mL | 7.1236 mL | 14.2472 mL | |
| 5 mM | 0.2849 mL | 1.4247 mL | 2.8494 mL | |
| 10 mM | 0.1425 mL | 0.7124 mL | 1.4247 mL |