Physicochemical Properties
| Molecular Formula | C30H37F3N2O6 |
| Molecular Weight | 578.619799375534 |
| Exact Mass | 578.26 |
| Elemental Analysis | C, 62.27; H, 6.45; F, 9.85; N, 4.84; O, 16.59 |
| CAS # | 2222264-64-2 |
| PubChem CID | 139443340 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 5.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 41 |
| Complexity | 912 |
| Defined Atom Stereocenter Count | 5 |
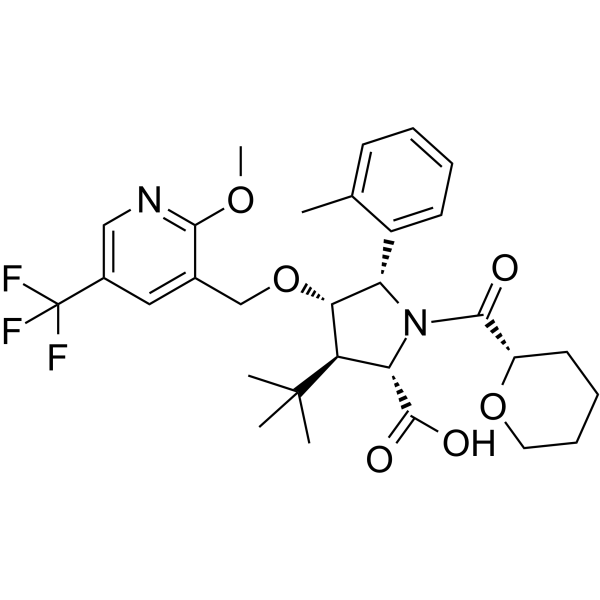
| SMILES | CC1=CC=CC=C1[C@H]2[C@H]([C@@H]([C@H](N2C(=O)[C@@H]3CCCCO3)C(=O)O)C(C)(C)C)OCC4=C(N=CC(=C4)C(F)(F)F)OC |
| InChi Key | YFEYDNAKCSOOOG-YCXOGWGTSA-N |
| InChi Code | InChI=1S/C30H37F3N2O6/c1-17-10-6-7-11-20(17)23-25(41-16-18-14-19(30(31,32)33)15-34-26(18)39-5)22(29(2,3)4)24(28(37)38)35(23)27(36)21-12-8-9-13-40-21/h6-7,10-11,14-15,21-25H,8-9,12-13,16H2,1-5H3,(H,37,38)/t21-,22+,23-,24-,25-/m0/s1 |
| Chemical Name | (2S,3R,4S,5S)-3-tert-butyl-4-[[2-methoxy-5-(trifluoromethyl)pyridin-3-yl]methoxy]-5-(2-methylphenyl)-1-[(2S)-oxane-2-carbonyl]pyrrolidine-2-carboxylic acid |
| Synonyms | GLPG3221; GLPG-3221; ABBV/GLPG-3221; 2222264-64-2; CHEMBL4588847; ABBV-3221; (2S,3R,4S,5S)-3-tert-butyl-4-[[2-methoxy-5-(trifluoromethyl)pyridin-3-yl]methoxy]-5-(2-methylphenyl)-1-[(2S)-oxane-2-carbonyl]pyrrolidine-2-carboxylic acid; (2S,3R,4S,5S)-3-tert-Butyl-4-{[2-methoxy-5-(trifluoromethyl)pyridin-3-yl]methoxy}-5-(2-methylphenyl)-1-[(2S)-oxane-2-carbonyl]pyrrolidine-2-carboxylic acid; SCHEMBL21305963; GLPG 3221 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | CFTR/cystic fibrosis transmembrane conductance regulator |
| ln Vitro | GLPG-3221 is a novel CFTR (cystic fibrosis transmembrane conductance) modulator with EC50 of 105 nM and has potential usefulness for treating cystic fibrosis. |
| ln Vivo | Treatment with 1 mg/kg of GLPG-3221 (iv) resulted in Cl and t1/2 of 0.13 L/h/kg and 3 hours, respectively [1]. The F% value of the GLPG-3221 (1 mg/kg; po) therapy was 53% [1]. |
| Enzyme Assay |
CYP3A4 Induction: [1] Cryopreserved primary human hepatocytes were thawed and cultured overnight prior to treatment. Cultured hepatocytes were treated with either test compounds (10 µM), vehicle control (0.1% v/v DMSO), or prototypical inducer of CYP3A4 (rifampin 10 µM) for 48 hours, with culture medium being refreshed every 24 hours. Following the 48 hour treatment, CYP3A4 mRNA levels measured in compound treated hepatocytes were expressed as a percentage of the response of positive control (Rifampin 10 µM). In test compound treated hepatocytes, CYP3A4 mRNA level increase by less than 20% of the response of positive control (Rifampin) is considered low risk for CYP3A4 induction. |
| Cell Assay |
Cell Surface Expression-Horse Radish Peroxidase (CSE-HRP) Assay: [1] A cellular assay for measuring the F508del CFTR cell surface expression after correction with test compounds either without or with a co-corrector (2 µM of 3-[(2R,4R)-4-({[1-(2,2- difluoro-1,3-benzodioxol-5-yl)cyclopropyl]carbonyl}amino)-7-methoxy-3,4-dihydro-2Hchromen-2-yl]benzoic acid, 1), was developed in human lung derived epithelial cell line (CFBE41o-).1 This was achieved by expressing the F508del CFTR mutation along with a horseradish peroxidase (HRP) in the fourth exofacial loop and then measuring the HRP activity using luminescence readout from these cells, CFBE41o-F508del CFTR-HRP, that were incubated overnight with the test corrector compounds.2 Briefly, for this primary assay, the CFBE41o-F508del CFTR-HRP cells were plated in 384-well plates at 4,000 cells/well along with 0.5 µg/mL doxycycline to induce the F508del CFTR-HRP expression and further incubated at 37 °C, 5% CO2 for 72 hours. The test compounds were then added at the required concentrations and further incubated for 18-24 hours at 33 °C. The highest concentration tested was 20 µM with an 8-point concentration response curve using a 3-fold dilution. Three replicate plates were run to determine one EC50. All plates contained negative controls (dimethyl sulfoxide, DMSO) and positive controls (3 µM of 3-[(2R,4R)-4-({[1-(2,2-difluoro-1,3- benzodioxol-5-yl)cyclopropyl]carbonyl}amino)-7-methoxy-3,4-dihydro-2H-chromen-2- yl]benzoic acid) (Compound 1) as well as on-plate concentration response of the positive control. Post incubation, the plates were washed 5× times with Dulbecco’s phosphate buffered saline (DPBS), followed by the addition of the HRP substrate, luminol (50 µL), and measuring the HRP activity using luminescence readout on EnVision® Multilabel Plate Reader. The raw counts from the experiment are analyzed using Accelrys® Assay Explorer v3.3. Trans-epithelial Current Clamp on Human Bronchial Epithelial Cells Conductance Assay: [1] A cell based assay using the primary human bronchial epithelial cells (hBE) was used as a secondary assay to test novel F508del CFTR correctors for their activity on primary hBE cells with F508del/F508del CFTR mutation. Primary human bronchial epithelial (hBE) cells from F508del/F508del CFTR patients were expanded from 1×106 to 250×106 cells.3 For this purpose, cells isolated from CF patients with the homozygous mutation were seeded onto 24 well Corning (Cat # 3378) filter plates that were coated with 3T3 conditioned media and grown at an air-liquid interface for 35 days using an Ultroser® G supplemented differentiation media. Apical surface mucus was removed 72 hours before the experiment by incubating the apical surface of the cells for 30 minutes with 3 mM dithiothreitol (DTT) prepared in the differentiation media, followed by aspiration of the mucus along with the media. The apical surface is washed again with phosphate buffered saline (PBS) incubated for 30 minutes followed with aspiration. The cells were then incubated with the desired dose of the corrector compounds 18-24 hours at 37 oC, 5% CO2. The corrector compounds were prepared as 10 mM stocks and the desired concentrations were prepared in differentiation media and were always applied on the basolateral side of the epithelial cells. On the day of measuring the corrector activity on the TECC, the cells were switched into a bicarbonate and serum free F-12 Coon’s medium and allowed to equilibrate for 90 minutes in a CO2 free incubator. At the time of measurement, the apical and basolateral sides of the filter were bathed with the F-12 Coon’s modification media (with 20 mM 4-(2-hydroxyethyl)-1- piperazineethanesulfonic acid (HEPES), pH 7.4 (using 1 M tris(hydroxymethyl)aminomethane (Tris)), and the measurements were made at 36.5 oC. Current responses before and after the sequential addition of benzamil (apical 6 M addition; for inhibiting epithelial ENaC channel), forskolin (apical and basolateral 10 M addition; for activating the CFTR channel), control potentiator (N-(3-carbamoyl-5,5,7,7-tetramethyl-4,7-dihydro-5H-thieno[2,3-c]pyran-2-yl)-1Hpyrazole-5-carboxamide; apical and basolateral 0.75 M addition; for potentiating the CFTR channel) and bumetanide (basolateral 20 M addition; for inhibiting the Na:2Cl:K cotransporter, an indirect measure of inhibiting the Cl- secretion driven by CFTR channel) were measured. The assay uses a TECC-24 (Transepithelial Current Clamp for 24 wells) instrument that measures the functionality of the mutated channel by measuring the equivalent CFTR current (IEQ) generated by the polarized primary epithelial cells. The instrument works by measuring the transepithelial potential difference (VT) and transepithelial conductance (GT) under current clamp conditions using a custom designed multi-channel current clamp and electrode manifold. Each measured VT values are corrected for the electrode offset potential, and each measured GT values are corrected for the combined solution series and empty filter resistances. The corrected VT and GT values were then used to calculate the equivalent current, IEQ using Ohm’s law (IEQ = VT.GT). In addition to calculating the IEQ, the area under the curve (AUC) for the time period between the forskolin peak IEQ response and at the time of bumetanide addition was also calculated using a one-third trapezoid method. The assay was run in a 24-well format and all 24-wells were measured at the same time point giving a higher throughput for this assay. |
| Animal Protocol |
Animal/Disease Models: Rat[1] Doses: 1 mg/kg Route of Administration: intravenous (iv) (iv)injection (pharmacokinetic/PK/PK analysis) Experimental Results: Cl and t1/2 were 0.13 L/h/kg and 3 hrs (hrs (hours)) respectively. |
| ADME/Pharmacokinetics | Compound 19 (ABBV-3221) was also found to have a good pharmacokinetic (PK) profile in both rats and dogs, such as high bioavailability (53% in rats; 78% in dogs) and reasonable half life (3 hr in rats; 6.4 hr in dogs). |
| References |
[1]. Discovery of ABBV/GLPG-3221, a Potent Corrector of CFTR for the Treatment of Cystic Fibrosis.ACS Med Chem Lett. 2019 Oct 31;10(11):1543-1548. |
| Additional Infomation | Cystic fibrosis (CF) is a genetic disorder that affects multiple tissues and organs. CF is caused by mutations in the CFTR gene, resulting in insufficient or impaired cystic fibrosis transmembrane conductance regulator (CFTR) protein. The deletion of phenylalanine at position 508 of the protein (F508del-CFTR) is the most common mutation observed in CF patients. The most effective treatments of these patients employ two CFTR modulator classes, correctors and potentiators. CFTR correctors increase protein levels at the cell surface; CFTR potentiators enable the functional opening of CFTR channels at the cell surface. Triple-combination therapies utilize two distinct corrector molecules (C1 and C2) to further improve the overall efficacy. We identified the need to develop a C2 corrector series that had the potential to be used in conjunction with our existing C1 corrector series and provide robust clinical efficacy for CF patients. The identification of a pyrrolidine series of CFTR C2 correctors and the structure-activity relationship of this series is described. This work resulted in the discovery and selection of (2S,3R,4S,5S)-3-(tert-butyl)-4-((2-methoxy-5-(trifluoromethyl)pyridin-3-yl)methoxy)-1-((S)-tetrahydro-2H-pyran-2-carbonyl)-5-(o-tolyl)pyrrolidine-2-carboxylic acid (ABBV/GLPG-3221), which was advanced to clinical trials.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7282 mL | 8.6412 mL | 17.2825 mL | |
| 5 mM | 0.3457 mL | 1.7282 mL | 3.4565 mL | |
| 10 mM | 0.1728 mL | 0.8641 mL | 1.7282 mL |