GGTI-298 (GGTI298) is a potent geranylgeranyltransferase-I (GGTase-I) inhibitor with potential anticancer actrivity. GGTI-298 inhibits the G(1)-S transition and MAP kinase activation in transformed adrenocortical cells that overexpress Ki-Ras. GGTI-298 induces hypophosphorylation of retinoblastoma and partner switching of cyclin-dependent kinase inhibitors. A potential mechanism for GGTI-298 antitumor activity. Human tumor cells are stopped in G0/G1 by GGTI-298, which also causes p21(WAF1/CIP1/SDI1) to be induced without the need for p53. GGTI-298 induces G0-G1 block and apoptosis whereas FTI-277 causes G2-M enrichment in A549 cells.
Physicochemical Properties
| Molecular Formula | C27H33N3O3S |
| Molecular Weight | 479.634 |
| Exact Mass | 479.22 |
| Elemental Analysis | C, 67.61; H, 6.93; N, 8.76; O, 10.01; S, 6.69 |
| CAS # | 180977-44-0 |
| Related CAS # | GGTI298 Trifluoroacetate; 1217457-86-7; 180977-44-0; 205590-41-6 (HCl) |
| PubChem CID | 9811606 |
| Appearance | White solid powder |
| LogP | 6.474 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 34 |
| Complexity | 661 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | S([H])C([H])([H])[C@@]([H])(C([H])([H])N([H])C1C([H])=C([H])C(C(N([H])[C@]([H])(C(=O)OC([H])([H])[H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])[H])=O)=C(C=1[H])C1=C([H])C([H])=C([H])C2=C([H])C([H])=C([H])C([H])=C12)N([H])[H].FC(C(=O)O[H])(F)F |
| InChi Key | XVWPFYDMUFBHBF-CLOONOSVSA-N |
| InChi Code | InChI=1S/C27H33N3O3S/c1-17(2)13-25(27(32)33-3)30-26(31)23-12-11-20(29-15-19(28)16-34)14-24(23)22-10-6-8-18-7-4-5-9-21(18)22/h4-12,14,17,19,25,29,34H,13,15-16,28H2,1-3H3,(H,30,31)/t19-,25+/m1/s1 |
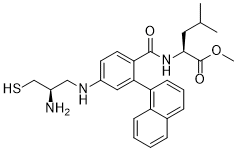
| Chemical Name | methyl (2S)-2-[[4-[[(2R)-2-amino-3-sulfanylpropyl]amino]-2-naphthalen-1-ylbenzoyl]amino]-4-methylpentanoate |
| Synonyms | GGTI 298; GGTI298; GGTI-298; GGTI298; 180977-44-0; Ggti 298; (S)-methyl 2-(4-(((R)-2-amino-3-mercaptopropyl)amino)-2-(naphthalen-1-yl)benzamido)-4-methylpentanoate; ELA97V8Q7P; CHEMBL282748; L-Leucine, N-[4-[[(2R)-2-amino-3-mercaptopropyl]amino]-2-(1-naphthalenyl)benzoyl]-, methyl ester; GGTI-298 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Rap1A ( pIC50 = 3 μM ); Histamine H1 receptor ( Ha-Ras > 20 μM ) | ||
| ln Vitro | Both RhoA parent (GGTI298) and ROCK parent (H1152) significantly reduce cAMP agonist-stimulated IK(ap), while night also reduces the colocalization of KCNN4c with the apical membrane marker malt agglutinin in T84WT cells [1]. Knockdown of NF-κB eliminates NF-κB activation, thereby sensitizing cells to activation of DR5 induced by the combined induction of GGTI298 and TRAIL. GGTI298/TRAIL activates NF-κB and inhibits Akt. Knocking out DR5 can prevent the reduction of IκBα and p-Akt induced by GGTI298/TRAIL, indicating that DR5 mediates the reduction of IκBα and p-Akt induced by GGTI298/TRAIL. In contrast, DR4 knockdown further promotes GGTI298/TRAIL-induced p-Akt reduction [2]. | ||
| ln Vivo |
In vivo model ileal loop experiments showed that when TRAM-34, GGTI298 or H1152 were injected into the loop together with cholera toxin, fluid volume concentration was reduced in a dose-dependent manner [1]. A major barrier towards the study of the effects of drugs on Giant Cell Tumor of Bone (GCT) has been the lack of an animal model. In this study, we created an animal model in which GCT stromal cells survived and functioned as proliferating neoplastic cells. A proliferative cell line of GCT stromal cells was used to create a stable and luciferase-transduced cell line, Luc-G33. The cell line was characterized and was found that there were no significant differences on cell proliferation rate and recruitment of monocytes when compared with the wild type GCT stromal cells. We delivered the Luc-G33 cells either subcutaneously on the back or to the tibiae of the nude mice. The presence of viable Luc-G33 cells was assessed using real-time live imaging by the IVIS 200 bioluminescent imaging (BLI) system. The tumor cells initially propagated and remained viable on site for 7 weeks in the subcutaneous tumor model. We also tested in vivo antitumor effects of Zoledronate (ZOL) and Geranylgeranyl transferase-I inhibitor (GGTI-298) alone or their combinations in Luc-G33-transplanted nude mice. ZOL alone at 400 µg/kg and the co-treatment of ZOL at 400 µg/kg and GGTI-298 at 1.16 mg/kg reduced tumor cell viability in the model. Furthermore, the anti-tumor effects by ZOL, GGTI-298 and the co-treatment in subcutaneous tumor model were also confirmed by immunohistochemical (IHC) staining. In conclusion, we established a nude mice model of GCT stromal cells which allows non-invasive, real-time assessments of tumor development and testing the in vivo effects of different adjuvants for treating GCT.[4] |
||
| Enzyme Assay | The specified cells are lysed in Reporter Lysis Buffer and then put through a luciferase activity assay in a luminometer using the Luciferase Assay System. The relative luciferase activity is calibrated to the protein concentration. | ||
| Cell Assay |
In 96-well cell culture plates, cells are seeded, and the following day, they are treated with the specified agents. The sulforhodamine B assay is used to calculate the number of viable cells. Cell Survival Assay[2] Cells were seeded in 96-well cell culture plates and treated the next day with the agents indicated. The viable cell number was determined using the sulforhodamine B assay, as previously described. Detection of Apoptosis[2] Apoptosis was evaluated by Annexin V staining using Annexin V-PE apoptosis detection kit purchased commercially following the manufacturer's instructions. Caspase activation was also detected by Western blotting (as described below) as an additional indicator of apoptosis. Western Blot Analysis[2] Whole-cell protein lysates were prepared and analyzed by Western blotting as described previously. |
||
| Animal Protocol |
|
||
| References |
[1]. The Epac1 signaling pathway regulates Cl- secretion via modulation of apical KCNN4c channels in diarrhea. J Biol Chem. 2013 Jul 12;288(28):20404-15. [2]. Dissecting the roles of DR4, DR5 and c-FLIP in the regulation of geranylgeranyltransferase I inhibition-mediated augmentation of TRAIL-induced apoptosis. Mol Cancer. 2010 Jan 29;9:23. [3]. Platelet-derived growth factor receptor tyrosine phosphorylation requires protein geranylgeranylation but not farnesylation. J Biol Chem. 1996 Nov 1;271(44):27402-7. [4]. A mouse model of luciferase-transfected stromal cells of giant cell tumor of bone. Connect Tissue Res. 2015 Nov;56(6):493-503. |
||
| Additional Infomation | (2S)-2-[[[4-[[(2R)-2-amino-3-mercaptopropyl]amino]-2-(1-naphthalenyl)phenyl]-oxomethyl]amino]-4-methylpentanoic acid methyl ester is a leucine derivative. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0849 mL | 10.4247 mL | 20.8494 mL | |
| 5 mM | 0.4170 mL | 2.0849 mL | 4.1699 mL | |
| 10 mM | 0.2085 mL | 1.0425 mL | 2.0849 mL |