GDC-0927 (formerly known as SRN-927) is a novel, potent, non-steroidal, orally bioavailable, selective ER (estrogen receptor) antagonist/ER degrader (SERD) that causes tumor regression in xenograft models grown from ER+ BC patients. As of right now, GDC-0927 looks to be well-tolerated. PK exposure appears to support QD dosing, there is evidence of strong PD target engagement, and heavily pretreated patients with advanced or metastatic ER+ BC—including those with ESR1 mutations—show encouraging anti-tumor activity. The potential for GDC-0927, an oral active SERD of the next generation, to surpass GDC-0810 as the best-in-class SERD molecule is apparent. GDC-0927 specifically targets the estrogen receptor alpha (ERα), a receptor that is expressed in over 70% of breast cancer cases, making it a commonly targeted target for the illness.
Physicochemical Properties
| Molecular Formula | C28H28FNO4 | |
| Molecular Weight | 461.524631500244 | |
| Exact Mass | 461.2 | |
| Elemental Analysis | C, 72.87; H, 6.12; F, 4.12; N, 3.03; O, 13.87 | |
| CAS # | 1642297-01-5 | |
| Related CAS # | (R)-GDC-0927;1642297-53-7;GDC-0927 Racemate;1443983-36-5 | |
| PubChem CID | 87055263 | |
| Appearance | Light yellow to yellow solid powder | |
| LogP | 4.7 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 7 | |
| Heavy Atom Count | 34 | |
| Complexity | 703 | |
| Defined Atom Stereocenter Count | 1 | |
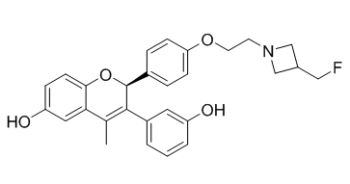
| SMILES | CC1=C([C@@H](OC2=C1C=C(C=C2)O)C3=CC=C(C=C3)OCCN4CC(C4)CF)C5=CC(=CC=C5)O |
|
| InChi Key | KJAAPZIFCQQQKX-NDEPHWFRSA-N | |
| InChi Code | InChI=1S/C28H28FNO4/c1-18-25-14-23(32)7-10-26(25)34-28(27(18)21-3-2-4-22(31)13-21)20-5-8-24(9-6-20)33-12-11-30-16-19(15-29)17-30/h2-10,13-14,19,28,31-32H,11-12,15-17H2,1H3/t28-/m0/s1 | |
| Chemical Name | (2S)-2-[4-[2-[3-(fluoromethyl)azetidin-1-yl]ethoxy]phenyl]-3-(3-hydroxyphenyl)-4-methyl-2H-chromen-6-ol | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Estrogen receptor
Estrogen Receptor α (ERα) (Ki = 0.1 nM for human ERα; IC₅₀ = 0.3 nM for ERα binding inhibition; IC₅₀ = 0.7 nM for ERα degradation in MCF-7 cells) [1] |
||
| ln Vitro |
GDC-0927 (formerly known as SRN-927) is a novel, potent, non-steroidal, orally bioavailable, selective ER (estrogen receptor) antagonist/ER degrader (SERD) that causes tumor regression in ER+ BC patient-derived xenograft models. With PK exposure supporting QD dosing, evidence of strong PD target engagement, and encouraging anti-tumor activity in heavily pretreated patients with advanced or metastatic ER+ BC, including patients with ESR1 mutations, GDC-0927 appears to be well tolerated so far. A next-generation orally active SERD called GDC-0927 appears to have more potential than GDC-0810 to be the best SERD molecule in its class. GDC-0927 targets the estrogen receptor alpha (ERα), which is a popular target for the disease because it is expressed in more than 70% of breast cancers. ERα binding and degradation: GDC-0927 is a potent oral selective estrogen receptor degrader (SERD) that binds to human ERα with high affinity (Ki = 0.1 nM) and inhibits ERα binding to estrogen response elements (ERE) with an IC₅₀ of 0.3 nM. It dose-dependently induces ERα degradation in ER+ breast cancer cell lines, achieving 90% ERα degradation at 1 nM in MCF-7 cells (IC₅₀ = 0.7 nM) and 85% degradation at 1 nM in T47D cells (IC₅₀ = 0.9 nM) [1] - Antiproliferative activity: GDC-0927 inhibits proliferation of ER+ HER2- breast cancer cell lines, including MCF-7 (IC₅₀ = 0.8 μM), T47D (IC₅₀ = 1.2 μM), ZR-75-1 (IC₅₀ = 1.5 μM), and MCF-7/LTED (tamoxifen-resistant, IC₅₀ = 2.3 μM). It does not inhibit proliferation of ER- breast cancer cell lines (MDA-MB-231, IC₅₀ > 50 μM), confirming ER selectivity [1] - ER signaling inhibition: In MCF-7 cells, GDC-0927 (1 nM) suppresses ER-mediated gene expression (GREB1, PGR, TFF1) by 70–80% at the mRNA level and reduces ER-dependent luciferase activity (IC₅₀ = 0.5 nM) [1] |
||
| ln Vivo |
GDC-0927 is a novel, potent, non-steroidal, orally bioavailable, selective ER antagonist/ER degrader (SERD) that causes tumor regression in xenograft models of patients with estrogen receptor-positive breast cancer (ER+BC)[1]. Human ER+ breast cancer xenograft models: Oral administration of GDC-0927 (3, 10, 30 mg/kg, once daily for 21 days) dose-dependently inhibits tumor growth in MCF-7 xenografts (nude mice), achieving 45%, 68%, and 82% tumor growth inhibition (TGI) compared to vehicle. In tamoxifen-resistant MCF-7/LTED xenografts, 30 mg/kg daily results in 75% TGI. Tumor tissue analysis shows reduced ERα protein levels (by 80% at 30 mg/kg) and decreased expression of ER target genes (GREB1, P |
||
| Cell Assay | GDC-0927 (formerly known as SRN-927) is a novel, potent, non-steroidal, orally bioavailable, selective ER (estrogen receptor) antagonist/ER degrader (SERD). GDC-0927 targets the estrogen receptor alpha (ER), which is a popular target for the disease because it is expressed in more than 70% of breast cancers. | ||
| Animal Protocol |
|
||
| ADME/Pharmacokinetics |
Oral absorption: In postmenopausal women with ER+ HER2- metastatic breast cancer, oral administration of GDC-0927 (doses 50–1200 mg once daily) results in dose-proportional increases in plasma AUC₀-24h and Cₘₐₓ. Tₘₐₓ is 2–4 hours post-dosing. Oral bioavailability is approximately 42% (based on comparison of IV and oral doses in a subset of patients) [1] - Elimination: Terminal half-life (t₁/₂) is 18–22 hours in humans, supporting once-daily dosing. Plasma clearance is 12–15 mL/min/kg. Approximately 65% of the dose is excreted in feces (30% as parent drug, 35% as metabolites) and 25% in urine (10% as parent drug, 15% as metabolites) [1] - Tissue distribution: In patients, GDC-0927 distributes to tumor tissue with a tumor-to-plasma ratio of 2.8 (measured in 5 patients with accessible tumors) [1] |
||
| Toxicity/Toxicokinetics |
Safety profile: GDC-0927 is generally well-tolerated at doses up to 1200 mg once daily. Most adverse events (AEs) are grade 1–2, including nausea (45%), fatigue (38%), diarrhea (32%), hot flashes (28%), and vomiting (22%). Grade 3 AEs occur in 15% of patients (primarily fatigue and diarrhea), with no grade 4–5 AEs reported [1] - Dose-limiting toxicity (DLT): No DLT is identified at doses up to 1200 mg once daily. The maximum tolerated dose (MTD) is not reached in the Phase I study [1] - Plasma protein binding: Human plasma protein binding rate is 97% (measured in vitro via ultrafiltration) [1] - Drug-drug interaction: In vitro studies show no inhibition of cytochrome P450 enzymes (CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A4) at concentrations up to 10 μM [1] |
||
| References |
[1]. Abstract PD5-10: A first-in-human phase I study to evaluate the oral selective estrogen receptor degrader (SERD), GDC-0927, in postmenopausal women with estrogen receptor positive (ER+) HER2-negative metastatic breast cancer (BC). AACR; |
||
| Additional Infomation |
GDC-0927 is under investigation in clinical trial NCT02316509 (A Study of GDC-0927 in Postmenopausal Women With Locally Advanced or Metastatic Estrogen Receptor Positive Breast Cancer). GDC-0927 is an oral, potent, selective estrogen receptor degrader (SERD) developed for the treatment of ER+ HER2- breast cancer [1] - Mechanism of action: Binds selectively to ERα with high affinity, induces conformational changes in ERα that promote its ubiquitination and proteasomal degradation, thereby blocking ER-mediated transcriptional activity and inhibiting proliferation of ER+ breast cancer cells. It retains activity in tamoxifen-resistant models [1] - Clinical indication: Investigated in postmenopausal women with ER+ HER2- metastatic breast cancer who have progressed on prior endocrine therapy (e.g., aromatase inhibitors, tamoxifen) [1] - Phase I clinical results: Among 42 evaluable patients, the objective response rate (ORR) is 24% (10/42), and the disease control rate (DCR, ORR + stable disease ≥ 16 weeks) is 67% (28/42). Median duration of response is 8.3 months [1] - Pharmacological advantage: Long half-life (18–22 hours) supports once-daily oral administration; good tumor penetration and activity in endocrine-resistant models address unmet needs in metastatic breast cancer [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.51 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.51 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.51 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1668 mL | 10.8338 mL | 21.6675 mL | |
| 5 mM | 0.4334 mL | 2.1668 mL | 4.3335 mL | |
| 10 mM | 0.2167 mL | 1.0834 mL | 2.1668 mL |