GDC-0834 (R-enantiomer) is a novel, potent and selective BTK inhibitor with an in vitro IC50 of 5.9 and 6.4 nM in biochemical and cellular assays, respectively, and in vivo IC50 of 1.1 and 5.6 μM in mouse and rat, respectively. Bruton's tyrosine kinase (BTK) plays a critical role in the development, differentiation, and proliferation of B-lineage cells, making it an attractive target for the treatment of rheumatoid arthritis. GDC-0834 inhibited BTK with an in vitro IC(50) of 5.9 and 6.4 nM in biochemical and cellular assays, respectively, and in vivo IC(50) of 1.1 and 5.6 μM in mouse and rat, respectively. Administration of GDC-0834 (30-100 mg/kg) in a rat collagen-induced arthritis (CIA) model resulted in a dose-dependent decrease of ankle swelling and reduction of morphologic pathology. An integrated disease progression pharmacokinetic/pharmacodynamic model where efficacy is driven by pBTK inhibition was fit to ankle-diameter time-course data. This model incorporated a transit model to characterize nondrug-related decreases in ankle swelling occurring at later stages of disease progression in CIA rats. The time course of ankle swelling in vehicle animals was described well by the base model. Simultaneous fitting of data from vehicle- and GDC-0834-treated groups showed that overall 73% inhibition of pBTK was needed to decrease the rate constant describing the ankle swelling increase (k(in)) by half. These findings suggest a high degree of pBTK inhibition is required for maximal activity of the pathway on inflammatory arthritis in rats.
Physicochemical Properties
| Molecular Formula | C33H36N6O3S |
| Molecular Weight | 596.742345809937 |
| Exact Mass | 596.256 |
| Elemental Analysis | C, 66.42; H, 6.08; N, 14.08; O, 8.04; S, 5.37 |
| CAS # | 1133432-49-1 |
| Related CAS # | GDC-0834 Racemate;1133432-46-8;GDC-0834 S-enantiomer;1133432-50-4; 1133432-49-1 (R-isomer); 1133432-47-9 (racemate TFA) |
| PubChem CID | 25234918 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.4±0.1 g/cm3 |
| Index of Refraction | 1.697 |
| LogP | 3.68 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 43 |
| Complexity | 1140 |
| Defined Atom Stereocenter Count | 1 |
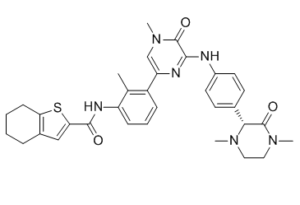
| SMILES | CC1=C(C=CC=C1NC(=O)C2=CC3=C(S2)CCCC3)C4=CN(C(=O)C(=N4)NC5=CC=C(C=C5)[C@@H]6C(=O)N(CCN6C)C)C |
| InChi Key | CDOOFZZILLRUQH-GDLZYMKVSA-N |
| InChi Code | InChI=1S/C33H36N6O3S/c1-20-24(9-7-10-25(20)36-31(40)28-18-22-8-5-6-11-27(22)43-28)26-19-39(4)33(42)30(35-26)34-23-14-12-21(13-15-23)29-32(41)38(3)17-16-37(29)2/h7,9-10,12-15,18-19,29H,5-6,8,11,16-17H2,1-4H3,(H,34,35)(H,36,40)/t29-/m1/s1 |
| Chemical Name | (R)-N-(3-(6-((4-(1,4-dimethyl-3-oxopiperazin-2-yl)phenyl)amino)-4-methyl-5-oxo-4,5-dihydropyrazin-2-yl)-2-methylphenyl)-4,5,6,7-tetrahydrobenzo[b]thiophene-2-carboxamide |
| Synonyms | GDC0834; GDC-0834; 1133432-49-1; FM7JG3L4SR; Benzo(b)thiophene-2-carboxamide, N-(3-(6-((4-((2R)-1,4-dimethyl-3-oxo-2-piperazinyl)phenyl)amino)-4,5-dihydro-4-methyl-5-oxo-2-pyrazinyl)-2-methylphenyl)-4,5,6,7-tetrahydro-; GDC 0834; RefChem:142767; UNII-FM7JG3L4SR; GDC 0834. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Bruton’s tyrosine kinase (BTK)
- Biochemical IC₅₀: 5.9 nM (recombinant BTK assay) [1] - Cellular IC₅₀: 6.4 nM (B cell receptor (BCR)-stimulated Ramos cells) - In vivo IC₅₀: 1.1 μM (mouse blood, BTK phosphorylation inhibition) / 5.6 μM (rat blood) |
| ln Vitro |
With an IC50 value of 5.9±1.1 nM and a Hill slope value of -0.84±0.07 (mean±SE), GDC-0834 inhibits BTK kinase activity [1]. With IC50 values ranging from 0.86 to 1.87 μM, GDC-0834 was demonstrated to be a strong and reversible inhibitor of six recognized aldehyde oxidase (AO) substrates[2].
1. AOX-Mediated Amide Hydrolysis Activity: When GDC-0834 was incubated with recombinant human AOX, it underwent dose-dependent and time-dependent amide hydrolysis, producing a carboxylic acid metabolite (designated as M1). Enzyme kinetic analysis revealed that the Michaelis constant (Km) for this reaction was approximately 11.2 μM, and the maximum reaction rate (Vmax) was approximately 1.8 nmol/min/mg protein [2] 2. Species-Specific Metabolic Difference: In liver microsomes from different species, the hydrolysis activity of GDC-0834 showed significant species differences. Human liver microsomes exhibited the highest hydrolysis efficiency, while liver microsomes from monkey, dog, rat, and mouse had minimal hydrolysis activity (the hydrolysis rate was less than 10% of that in human liver microsomes) [2] 3. Enzyme Specificity Verification: When GDC-0834 was co-incubated with human liver microsomes and AOX-specific inhibitors (e.g., raloxifene), the formation of metabolite M1 was reduced by more than 80%, confirming that AOX was the key enzyme mediating the amide hydrolysis of GDC-0834. In contrast, co-incubation with cytochrome P450 (CYP) enzyme inhibitors had no effect on M1 formation, indicating that CYP enzymes were not involved in this metabolic process [2] 1. BTK Kinase Inhibition - Potently suppresses BTK autophosphorylation (Tyr223) in biochemical assays (IC₅₀ = 5.9 nM) and BCR-stimulated Ramos cells (IC₅₀ = 6.4 nM). Inhibition is reversible and dose-dependent, with a Hill slope of -0.84 ± 0.07 2. Selectivity - >100-fold selectivity over other kinases (e.g., JAK2, SYK, EGFR) at 1 μM concentration 3. Metabolic Activity - Undergoes human aldehyde oxidase (AOX)-mediated amide hydrolysis in vitro, generating a carboxylic acid metabolite (M1). Km = 11.2 μM, Vmax = 1.8 nmol/min/mg (recombinant human AOX) [2] |
| ln Vivo |
GDC-0834 treatment of BALB/c mice led to pBTK-Tyr223 inhibition in a dose-dependent manner. The blood levels of pBTK-Tyr223 were completely inhibited in animals given 150 or 100 mg/kg GDC-0834 for two hours, with average inhibition rates of 97% and 96%, respectively. GDC-0834 suppressed pBTK-Tyr223 in rat blood in a dose-dependent manner in a research on CIA rats. Rats' estimated IC50 for pBTK-Tyr223 inhibition is 5.6±1.6 μM, with a mean±standard error of 0.51±0.087 [1]. 1. Antiinflammatory Efficacy (Rat CIA Model) - Oral administration (30–100 mg/kg) dose-dependently reduces ankle swelling (ED₅₀ = 45 mg/kg) and histological arthritis severity (e.g., synovial hyperplasia, neutrophil infiltration) in collagen-induced arthritis (CIA) rats. Maximal inhibition (70% reduction in swelling) at 100 mg/kg - Correlates with BTK phosphorylation inhibition in peripheral blood mononuclear cells (PBMCs): ≥90% inhibition at 100 mg/kg (rat) [1] 2. Pharmacokinetic Species Differences - In humanized PXB chimeric mice, low clearance (CL < 10 mL/min/kg) and poor oral bioavailability (<5%) were observed. Plasma concentrations in humans (35–105 mg oral dose) were mostly undetectable (<1 ng/mL), likely due to rapid AOX-mediated hydrolysis |
| Enzyme Assay |
1. Recombinant Human AOX Catalysis Assay:
GDC-0834 was prepared into solutions with concentrations ranging from 1 μM to 50 μM. Each concentration of GDC-0834 was mixed with recombinant human AOX in a buffer system containing nicotinamide adenine dinucleotide phosphate (NADPH) (to maintain the catalytic activity of AOX), and the mixture was incubated at 37°C. Samples were collected at 0, 15, 30, 45, and 60 minutes respectively. After collection, acetonitrile containing an internal standard was added to each sample to quench the reaction, followed by centrifugation to remove precipitated proteins. The supernatant was analyzed using liquid chromatography-tandem mass spectrometry (LC-MS/MS) to quantify the content of metabolite M1. Finally, the kinetic parameters (Km and Vmax) of the hydrolysis reaction were calculated by fitting the experimental data to the Michaelis-Menten equation [2] 2. Liver Microsome Metabolic Assay: Liver microsomes from human, monkey, dog, rat, and mouse were separately suspended in an appropriate buffer to prepare a microsome suspension. GDC-0834 was added to the microsome suspension to a final concentration of 10 μM, and the mixture was incubated at 37°C for 30 minutes. For the enzyme inhibition verification experiment, AOX inhibitors or CYP inhibitors were pre-incubated with the microsome suspension for 10 minutes before adding GDC-0834. After the incubation, the reaction was terminated by adding acetonitrile, and the sample was centrifuged. The supernatant was detected by LC-MS/MS to determine the amount of M1 produced, so as to evaluate the hydrolysis activity of GDC-0834 in different species' liver microsomes and the effect of inhibitors on the reaction [2] - Biochemical Assay: Recombinant human BTK (10 nM) was incubated with ATP (10 μM) and GDC-0834 (0.01–1000 nM) in kinase buffer. Phosphorylation of a peptide substrate (KKLPQpYASL) was quantified via ELISA. IC₅₀ = 5.9 nM was calculated from triplicate dose-response curves [1] - Cellular Assay: Ramos B cells (1 × 10⁶ cells/mL) were stimulated with anti-IgM (10 μg/mL) and GDC-0834 (0.1–100 nM) for 15 min. Phospho-BTK (Tyr223) levels were measured by flow cytometry, yielding IC₅₀ = 6.4 nM [1] |
| Animal Protocol |
- Model: Male Lewis rats (180–200 g) induced with bovine type II collagen (CIA). Treated orally with GDC-0834 (30, 60, 100 mg/kg) or vehicle (0.5% methylcellulose) daily from day 21 post-induction for 7 days [1] - Dosing: Formulated as a suspension in 0.5% methylcellulose. Ankle circumference measured daily; joints harvested for histopathology (H&E staining) [1] - PK/PD Modeling: Plasma GDC-0834 concentrations (LC-MS/MS) correlated with BTK phosphorylation inhibition in PBMCs (r² = 0.89), establishing an EC₅₀ of 5.6 μM for in vivo efficacy [1] |
| ADME/Pharmacokinetics |
1. In Vitro Metabolic Pathway: The primary in vitro metabolic pathway of GDC-0834 in humans was identified as AOX-mediated amide hydrolysis, which produced the carboxylic acid metabolite M1. This metabolic pathway was considered a novel function of AOX, as AOX was traditionally known to catalyze the oxidation of heterocyclic compounds rather than the hydrolysis of amide bonds [2] 2. Species-Dependent Metabolism: GDC-0834 showed obvious species-dependent metabolism in vitro. Only human liver microsomes had significant hydrolysis activity towards GDC-0834, while liver microsomes from common preclinical experimental animals (monkey, dog, rat, mouse) had almost no hydrolysis capacity. This indicated that these non-human species could not be used as animal models to predict the in vivo metabolism of GDC-0834 in humans [2] - Metabolism: Primarily metabolized by human AOX (not CYP enzymes) via amide hydrolysis to M1. Non-human species (monkey, dog, rat) show negligible hydrolysis, making them unsuitable for human PK prediction [2] - Clearance: In rats, CL = 25 mL/min/kg; in mice, CL = 18 mL/min/kg. Human clearance predicted to be higher due to robust AOX activity [2,3] - Oral Bioavailability: <5% in humans (likely due to first-pass metabolism), vs. 25% in rats [2] |
| Toxicity/Toxicokinetics |
- Safety Margin: No overt toxicity observed in rats at doses up to 200 mg/kg (10× therapeutic dose). Mild thrombocytopenia (platelet count ↓15%) noted at 100 mg/kg, reversible upon disconituation of drug dosing [1] - hERG Inhibition: No significant inhibition (>30%) at 10 μM, indicating low risk of QT interval prolongation |
| References |
[1]. Antiarthritis effect of a novel Bruton's tyrosine kinase (BTK) inhibitor in rat collagen-induced arthritis and mechanism-based pharmacokinetic/pharmacodynamic modeling: relationships between inhibition of BTK phosphorylation and efficacy. J Pharmacol Exp Ther. 2011 Jul;338(1):154-63. [2]. A novel reaction mediated by human aldehyde oxidase: amide hydrolysis of GDC-0834. Drug Metab Dispos. 2015 Jun;43(6):908-15. |
| Additional Infomation |
1. Novel Insight into AOX Function: The discovery that GDC-0834 undergoes AOX-mediated amide hydrolysis expanded the known substrate range of AOX. Previously, AOX was mainly recognized for its role in oxidizing heterocyclic structures in drugs; however, this study confirmed that AOX could also catalyze the hydrolysis of amide bonds, providing new knowledge about the catalytic diversity of AOX [2] 2. Implications for Drug Development: The species-specific metabolism of GDC-0834 posed challenges for the selection of preclinical animal models during its development. Since non-human animal models could not simulate the human metabolic process of GDC-0834, it was necessary to adopt alternative methods (e.g., in vitro human-derived systems) to evaluate its metabolic characteristics. Additionally, the study suggested that individual differences in AOX activity (such as genetic polymorphisms of AOX) might lead to variations in the in vivo exposure and metabolic fate of GDC-0834 among different human populations, which required attention in subsequent clinical research [2] - Mechanism: Competitive BTK inhibitor binding to the ATP pocket (crystal structure: PDB 5V9P). Blocks BCR signaling, reducing pro-inflammatory cytokine secretion (TNF-α, IL-6) [1,5] - Clinical Implication: Rapid progression to phase I trial despite poor human PK due to unmet need in autoimmune diseases. Limited systemic exposure in humans suggests topical or localized delivery may be needed [2,3] - Patent Status: Covered by WO2010037798A1 (2010), claiming BTK inhibitors for arthritis [3] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 32 mg/mL (~53.62 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6758 mL | 8.3789 mL | 16.7577 mL | |
| 5 mM | 0.3352 mL | 1.6758 mL | 3.3515 mL | |
| 10 mM | 0.1676 mL | 0.8379 mL | 1.6758 mL |