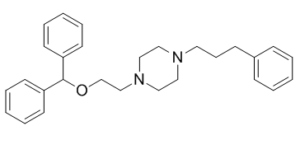
GBR-12935 is a novel, potent and selective dopamine reuptake inhibitor. It was originally developed in its 3H radiolabelled form for the purpose of mapping the distribution of dopaminergic neurons in the brain by selective labelling of dopamine transporter proteins. This has led to potential clinical uses in the diagnosis of Parkinson's disease, although selective radioligands such as Ioflupane are now available for this application. GBR-12935 is now widely used in animal research into Parkinson's disease and the dopamine pathways in the brain.
Physicochemical Properties
| Molecular Formula | C28H34N2O.C4H4O4 |
| Molecular Weight | 530.65452 |
| Exact Mass | 414.267 |
| CAS # | 76778-22-8 |
| Related CAS # | GBR 12935 dihydrochloride;67469-81-2 |
| PubChem CID | 3456 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.067g/cm3 |
| Boiling Point | 540.6ºC at 760 mmHg |
| Flash Point | 143.9ºC |
| Index of Refraction | 1.576 |
| LogP | 4.63 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 31 |
| Complexity | 440 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | N1(CCOC(C2=CC=CC=C2)C3=CC=CC=C3)CCN(CCCC4=CC=CC=C4)CC1 |
| InChi Key | RAQPOZGWANIDQT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C28H34N2O/c1-4-11-25(12-5-1)13-10-18-29-19-21-30(22-20-29)23-24-31-28(26-14-6-2-7-15-26)27-16-8-3-9-17-27/h1-9,11-12,14-17,28H,10,13,18-24H2 |
| Chemical Name | 1-(2-benzhydryloxyethyl)-4-(3-phenylpropyl)piperazine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | GBR 12909 (10-100 nM) demonstrates a strong affinity for CYP2D6, demonstrating a Kd value of 42.2 nM, a value that is less than that of the dopamine transporter. In addition to being strong and selective inhibitors of CYPZD enzyme activity, quinidine and quinine can lessen the binding impact [1]. Extracellular dopamine levels rise to almost 400% of basal values in the nucleus accumbens after GBR 12935 (10 nM; 2 minutes) is given [2]. Extracellular dopamine levels are raised by GBR 12935 (100 μM; 60 min) in contrast to locally perfused artificial cerebrospinal fluid (ACSF) [2]. In nucleus accumbens homogenates, GBR 12935 (1-9 nM) dose-dependently decreases [3H]dopamine's active absorption [2]. The increase in extracellular dopamine levels caused by GBR 12935 was dramatically decreased to basal levels when 100 μM sulpiride or raclopride were co-infused with 100 μM GBR 12935 [2]. |
| ln Vivo | GBR 12935 (1–32 mg/kg; repeated injection; 7 days) caused a larger increase in locomotor activity in C57BL/6J mice as compared to DBA/2J mice, and 10 mg/kg; injection; 7 days) produced a small number of mice allergic to the stereotypy brought on by repeated cocaine injections [3]. |
| Animal Protocol |
Animal/Disease Models: Adult male DBA/2J and C57BL/6J mice (22-30 g) [3] Doses: 1.0, 3.2, 10, 32 mg/kg Route of Administration: Repeat injection; continued for 7 days Experimental Results: C57BL/6J The locomotor activity of mice was increased to a greater extent than that of DBA/2J mice. Challenge with 10 mg/kg GBR 12935 on day eight did not induce stereotypy in mice pretreated with seven consecutive injections of 32 mg/kg cocaine or saline. |
| References |
[1]. Hiroi T, et al. Specific binding of 1-[2-(diphenylmethoxy)ethyl]-4-(3-phenyl propyl) piperazine (GBR-12935), an inhibitor of the dopamine transporter, to human CYP2D6. Biochem Pharmacol. 1997 Jun 15;53(12):1937-9. [2]. Rahman S, et al. Negative interaction of dopamine D2 receptor antagonists and GBR 12909 and GBR 12935 dopamine uptake inhibitors in the nucleus accumbens. Eur J Pharmacol. 2001 Feb 23;414(1):37-44. [3]. Tolliver BK, et al. Comparison of cocaine and GBR 12935: effects on locomotor activity and stereotypy in two inbred mouse strains. Pharmacol Biochem Behav. 1994 Jul;48(3):733-9. [4]. Darmani NA. Cocaine and selective monoamine uptake blockers (sertraline, nisoxetine, and GBR 12935) prevent the d-fenfluramine-induced head-twitch response in mice. Pharmacol Biochem Behav. 1998 May;60(1):83-90. |
| Additional Infomation | 1-[2-(benzhydryloxy)ethyl]-4-(3-phenylpropyl)piperazine is an N-alkylpiperazine that consists of piperazine bearing 2-(benzhydryloxy)ethyl and 3-phenylpropyl groups at positions 1 and 4 respectively. Potent and selective inhibitor of dopamine uptake (KD = 5.5 nM in rat striatal membranes). It has a role as a dopamine uptake inhibitor. It is a N-alkylpiperazine, an ether and a tertiary amino compound. It is a conjugate base of a 1-[2-(benzhydryloxy)ethyl]-4-(3-phenylpropyl)piperazinediium(2+). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8845 mL | 9.4224 mL | 18.8448 mL | |
| 5 mM | 0.3769 mL | 1.8845 mL | 3.7690 mL | |
| 10 mM | 0.1884 mL | 0.9422 mL | 1.8845 mL |