Physicochemical Properties
| Molecular Formula | C21H18BRNO3 |
| Molecular Weight | 412.28 |
| Exact Mass | 411.046 |
| Elemental Analysis | C, 61.18; H, 4.40; Br, 19.38; N, 3.40; O, 11.64 |
| CAS # | 881639-98-1 |
| PubChem CID | 5322399 |
| Appearance | White to off-white solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 529.6±50.0 °C at 760 mmHg |
| Flash Point | 274.1±30.1 °C |
| Vapour Pressure | 0.0±1.4 mmHg at 25°C |
| Index of Refraction | 1.637 |
| LogP | 4.37 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 26 |
| Complexity | 596 |
| Defined Atom Stereocenter Count | 3 |
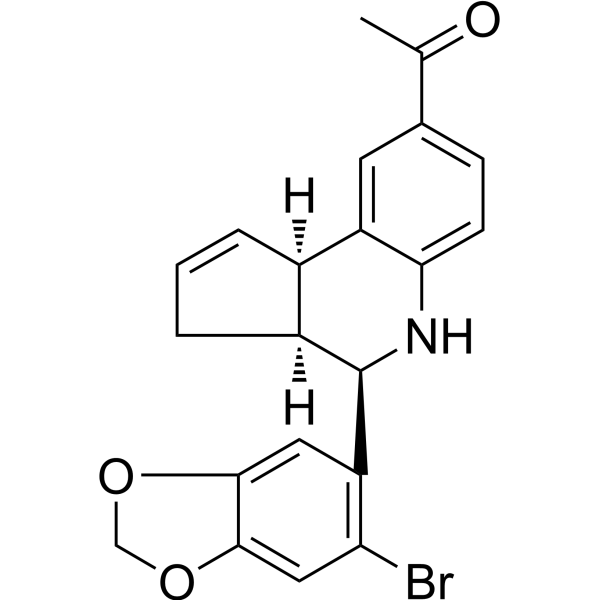
| SMILES | BrC1C=C2OCOC2=CC=1[C@@H]1NC2C=CC(=CC=2[C@@H]2C=CC[C@H]12)C(=O)C |
| InChi Key | VHSVKVWHYFBIFJ-HKZYLEAXSA-N |
| InChi Code | InChI=1S/C21H18BrNO3/c1-11(24)12-5-6-18-15(7-12)13-3-2-4-14(13)21(23-18)16-8-19-20(9-17(16)22)26-10-25-19/h2-3,5-9,13-14,21,23H,4,10H2,1H3/t13-,14+,21-/m1/s1 |
| Chemical Name | 1-[(3aS,4R,9bR)-4-(6-bromo-1,3-benzodioxol-5-yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl]ethanone |
| Synonyms | G1; G 1; G-1 |
| HS Tariff Code | 2934.99.03.00 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | G-1 is a selective GPR30 coagulant that has a high affinity and is nonsteroidal, with a Ki of 11 nM [1]. G-1 treatment (10 μM and 100 μM) for 48 and 72 hours dramatically inhibited cell proliferation (P<0.001). G-1's IC50 value at 72 hours was determined to be 20 μM. G-1 treatment of A549 cells at a dose of 20 μM resulted in a considerable increase in cells and a persistent anti-proliferative impact (P<0.001). After receiving G-1 therapy for 24 hours, cell cycle examination of H295R cells revealed that the cells were in the G2 phase of their life cycle. G-1 decreases Bcl-2 while also expressing Bax[3]. |
| ln Vivo | The Basso S Scale (BMS) score of the G-1 group was substantially greater (P<0.05) than that of the other groups, according to the results obtained 14 days after the injury. This group had fewer positive cells than the other groups (P<0.05), and there was no difference when compared (P>0.05)[1]. Beginning on day 14 of therapy, the G-1 medication resulted in a statistically significant decrease in the volume of the tumor, and after three weeks of treatment, the tumor weight of transplanted tumors was significantly lower than that of animals receiving a vehicle treatment [3]. |
| References |
[1]. G-1 exerts neuroprotective effects through G protein-coupled estrogen receptor 1 following spinal cord injury in mice. Biosci Rep. 2016 Aug 31;36(4). pii: e00373. [2]. Oxidative/antioxidative enzyme-mediated antiproliferative and proapoptotic effects of the GPER1 agonist G-1 on lung cancer cells. Oncol Lett. 2015 Nov;10(5):3177-3182. [3]. GPER agonist G-1 decreases adrenocortical carcinoma (ACC) cell growth in vitro and in vivo. Oncotarget. 2015 Aug 7;6(22):19190-203. |
| Additional Infomation |
G-1 is an agonist. G Protein-coupled Estrogen Receptor Agonist LNS8801 is an orally bioavailable selective agonist of the G protein-coupled estrogen receptor (G protein-coupled estrogen receptor 1; GPER; GPER1; GPR30), with potential immunomodulating and antineoplastic activities. Upon oral administration, LNS8801 targets, binds to and activates GPER. This activates GPER-mediated signaling and suppresses the expression of various tumor-associated genes, such as c-Myc and programmed cell death-ligand 1 (PD-L1). This leads to an inhibition of tumor cell proliferation. GPER activation may also induce immune memory. GPER is a membrane protein and a member of the G-protein coupled receptors (GPCRs) that are widely distributed in various tissues. GPER is a tumor suppressor in a wide range of cell cancer types. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~121.28 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.06 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (6.06 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.06 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4255 mL | 12.1277 mL | 24.2554 mL | |
| 5 mM | 0.4851 mL | 2.4255 mL | 4.8511 mL | |
| 10 mM | 0.2426 mL | 1.2128 mL | 2.4255 mL |