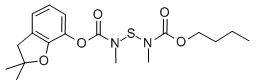
Furathiocarb is a carbamate pesticide
Physicochemical Properties
| Molecular Formula | C18H26N2O5S |
| Molecular Weight | 382.47 |
| Exact Mass | 382.156 |
| CAS # | 65907-30-4 |
| PubChem CID | 47759 |
| Appearance | Yellow, viscous liquid. |
| Density | 1.208g/cm3 |
| Boiling Point | 460ºC |
| Index of Refraction | 1.551 |
| LogP | 4.262 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 26 |
| Complexity | 502 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CCCCOC(=O)N(C)SN(C)C(=O)OC1=CC=CC2=C1OC(C)(C)C2 |
| InChi Key | HAWJXYBZNNRMNO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C18H26N2O5S/c1-6-7-11-23-16(21)19(4)26-20(5)17(22)24-14-10-8-9-13-12-18(2,3)25-15(13)14/h8-10H,6-7,11-12H2,1-5H3 |
| Chemical Name | (2,2-dimethyl-3H-1-benzofuran-7-yl) N-[butoxycarbonyl(methyl)amino]sulfanyl-N-methylcarbamate |
| Synonyms | CGA 73102; Deltanet; Promet; Promet 666SCO; 2,4-Dimethyl-5-oxo-6-oxa-3-thia-2,4-diazadecanoic Acid 2,3-Dihydro-2,2-dimethyl-7-benzofuranyl Ester |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Little information is available on the distribution of carbamates in the various organs and tissues in mammals following exposure by inhalation or the oral route. The organs in which residues have been reported are the liver, kidneys, brain, fat, and muscle.The half-life in the rat is of the order of 3 - 8 hr. It seems that the excretion of carbamates via urine is also rapid in man, and that the metabolic pathways in man are the same as those in the rat. /Carbamate pesticides/ The pharmacokinetics of furathiocarb were studied in vivo in male Sprague-Dawley rats following dermal treatment. HPLC and post-column derivatization were used for the analysis of furathiocarb and its metabolites (carbofuran, 3-hydroxycarbofuran and 3-ketocarbofuran). Carbofuran and 3-hydroxycarbofuran were detected in plasma and urine rather than furathiocarb. 3-Ketocarbofuran, another potential metabolite, was not observed in any sample. The concentration of carbofuran was higher than that of 3-hydroxycarbofuran in plasma, but the reverse was the case in urine. The corresponding area under the plasma concentration-time curve, Tmax, and Cmax values of carbofuran and 3-hydroxycarbofuran for 1,500 mg/kg doses were 2.4-8.0 mg equiv/ml, 12 hr and 0.1-0.4 mg equiv/ml, respectively. T1/2 was calculated only for 3-hydroxycarbofuran (28 hr). Two metabolites were excreted in a dose-dependent manner without saturation. Metabolism / Metabolites Metabolic transformation in rats proceeds via rapid and complete hydrolysis, followed by oxidation and conjugation. Excretion occurs mainly via kidney. The first step in the metabolism of carbamates is hydrolysis to carbamic acid, which decomposes to carbon dioxide (CO2) and the corresponding amine. The mechanism of hydrolysis is different for N -methyl and N-dimethyl derivatives. The N-methyl carbamates pass through an isocyanate intermediate, whereas in the hydrolysis of N-dimethylcarbamates, an addition product with a hydroxyl ion is formed yielding the alcohol and N-dimethyl substituted acid. The rate of hydrolysis by esterases is faster in mammals than in plants and insects. Apart from hydrolysis, oxidation also takes place including: hydroxylation of the aromatic ring, O-dealkylation, N -methyl hydroxylation, N-dealkylation, oxidation of aliphatic side chains, and sulfoxidation to the corresponding sulfone. Oxidation is associated with the mixed-function oxidase (MFO) enzymes. Conjugation leads to the formation of O- and N- glucuronides, sulfates, and mercapturic acid derivatives in mammals. Glycosides and phosphates are conjugation products more common in plants. /Carbamate Pesticides/ The pharmacokinetics of furathiocarb were studied in vivo in male Sprague-Dawley rats following dermal treatment. HPLC and post-column derivatization were used for the analysis of furathiocarb and its metabolites (carbofuran, 3-hydroxycarbofuran and 3-ketocarbofuran). Carbofuran and 3-hydroxycarbofuran were detected in plasma and urine rather than furathiocarb. 3-Ketocarbofuran, another potential metabolite, was not observed in any sample. ... The carbamates are hydrolyzed enzymatically by the liver; degradation products are excreted by the kidneys and the liver. (L793) Biological Half-Life The half-life in the rat is of the order of 3-8 hr. /Carbamate pesticides/ |
| Toxicity/Toxicokinetics |
Toxicity Summary Furathiocarb is a cholinesterase or acetylcholinesterase (AChE) inhibitor. Carbamates form unstable complexes with chlolinesterases by carbamoylation of the active sites of the enzymes. This inhibition is reversible. A cholinesterase inhibitor suppresses the action of acetylcholine esterase. Because of its essential function, chemicals that interfere with the action of acetylcholine esterase are potent neurotoxins, causing excessive salivation and eye-watering in low doses. Headache, salivation, nausea, vomiting, abdominal pain and diarrhea are often prominent at higher levels of exposure. Acetylcholine esterase breaks down the neurotransmitter acetylcholine, which is released at nerve and muscle junctions, in order to allow the muscle or organ to relax. The result of acetylcholine esterase inhibition is that acetylcholine builds up and continues to act so that any nerve impulses are continually transmitted and muscle contractions do not stop. Toxicity Data LC50 (rat) = 214 mg/m3/4h Non-Human Toxicity Values LD50 Rat oral 53 mg/kg LD50 Mouse oral 327 mg/kg LD50 Rat percutaneous >2,000 mg/kg LC50 Rat inhalation 0.214 mg/l air/4 hr |
| References | Anal. Chem., 80, 9450 (2008) |
| Additional Infomation |
Furathiocarb is a carbamate ester and a member of 1-benzofurans. It has a role as an EC 3.1.1.7 (acetylcholinesterase) inhibitor, a carbamate insecticide and an agrochemical. Furathiocarb is a carbamate pesticide. Carbamate pesticides are derived from carbamic acid and kill insects in a similar fashion as organophosphate insecticides. They are widely used in homes, gardens and agriculture. The first carbamate, carbaryl, was introduced in 1956 and more of it has been used throughout the world than all other carbamates combined. Because of carbaryl's relatively low mammalian oral and dermal toxicity and broad control spectrum, it has had wide use in lawn and garden settings. Most of the carbamates are extremely toxic to Hymenoptera, and precautions must be taken to avoid exposure to foraging bees or parasitic wasps. Some of the carbamates are translocated within plants, making them an effective systemic treatment. (L795) Mechanism of Action Carbamates are effective insecticides by virtue of their ability to inhibit acetylcholinesterase (AChE) in the nervous system. They also inhibit other esterases. The carbamylation of the enzyme is unstable, and the regeneration of AChE is relatively rapid compared with that from a phosphorylated enzyme. Thus, carbamate pesticides are less dangerous with regard to human exposure than organophosphorus pesticides. The ratio between the dose required to produce death and the dose required to produce minimum symptoms of poisoning is substantially larger for carbamate compounds than for organophosphorus compounds. /Carbamate pesticides/ |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6146 mL | 13.0729 mL | 26.1458 mL | |
| 5 mM | 0.5229 mL | 2.6146 mL | 5.2292 mL | |
| 10 mM | 0.2615 mL | 1.3073 mL | 2.6146 mL |