Physicochemical Properties
| Molecular Formula | C16H11FNNA2O6P |
| Molecular Weight | 409.21 |
| Exact Mass | 409.0103 |
| CAS # | 1256037-62-3 |
| Related CAS # | 1256037-60-1;Foslinanib (CVM-1118); |
| PubChem CID | 49840583 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 0 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 27 |
| Complexity | 582 |
| Defined Atom Stereocenter Count | 0 |
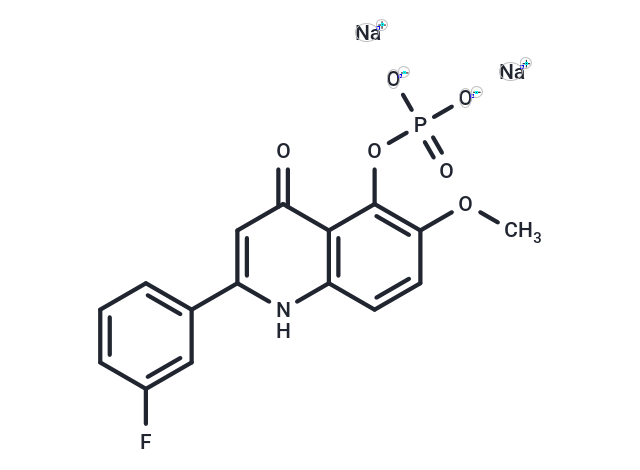
| SMILES | COC1=C(C2=C(C=C1)NC(=CC2=O)C3=CC(=CC=C3)F)OP(=O)([O-])[O-].[Na+].[Na+] |
| InChi Key | TWMCXXQLLQDSTN-UHFFFAOYSA-L |
| InChi Code | InChI=1S/C16H13FNO6P.2Na/c1-23-14-6-5-11-15(16(14)24-25(20,21)22)13(19)8-12(18-11)9-3-2-4-10(17)7-9;;/h2-8H,1H3,(H,18,19)(H2,20,21,22);;/q;2*+1/p-2 |
| Chemical Name | disodium;[2-(3-fluorophenyl)-6-methoxy-4-oxo-1H-quinolin-5-yl] phosphate |
| Synonyms | Foslinanib disodium; 1256037-62-3; TRX818 Sodium; CVM-1118 disodium; TRX-818 (disodium); 8X5F5LAF7B; UNII-8X5F5LAF7B; disodium;[2-(3-fluorophenyl)-6-methoxy-4-oxo-1H-quinolin-5-yl] phosphate; |
| HS Tariff Code | 2934.99.9001 |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Antineoplastic and anti-vasculogenic mimicry |
| ln Vitro |
The anti-cancer activity of CVM-1118 was tested using the NCI60 screening assay. The results showed that CVM-1118 inhibited cancer cell growth in approximately 87% of cell lines tested with the average GI50 value <100 nM. COMPARE analysis using the GI50 results by the NCI was also performed. Interestingly, it did not show close correlation with any standard cancer drugs presented in the NCI60 screen database, suggesting that a novel mechanism of action may be involved for the cytotoxic effect of CVM-1118 in cancer cells.[2] Previous exploration of 2-phenylquinolin-4-ones (2-PQs) has led to an anticancer drug candidate 2-(2-fluorophenyl)-6,7-methylenedioxyquinolin-4-one monosodium phosphate (CHM-1-P-Na). In order to develop additional new drug candidates, novel 2-PQs were designed, synthesized, and evaluated for cytotoxic activity. Most analogues, including 1b, 2a,b, 3a,b, 4a,b, and 5a,b, exhibited significant inhibitory activity (IC(50) of 0.03-8.2 μM) against all tested tumor cell lines. As one of the most potent analogue, 2-(3-fluorophenyl)-5-hydroxy-6-methoxyquinolin-4-one (3b) selectively inhibited 14 out of 60 cancer cell lines in a National Cancer Institute (NCI) evaluation. Preliminary mechanism of action study suggested that 3b had a significant effect on the tyrosine autophosphorylation of insulin-like growth factor-1 receptor (IGF-1R). Safety pharmacology profiling of 3b showed no significant effect on normal biological functions of most enzymes tested. Furthermore, sodium 2-(3-fluorophenyl)-6-methoxy-4-oxo-1,4-dihydroquinolin-5-yl phosphate (15), the monophosphate of 3b, exceeded the activity of doxorubicin and was comparable to CHM-1-P-Na in a Hep3B xenograft nude mice model. In summary, 15 is a promising clinical candidate and is currently under preclinical study.[1] |
| ln Vivo | Foslinanib is an orally bioavailable agent with potential antineoplastic and anti-vasculogenic mimicry activities. Upon oral administration, foslinanib targets and inhibits the formation of vasculogenic mimicry (VM; vascular mimicry). By destroying the VM channels and network, cancer cells are devoid of perfusion leading to an induction of cancer cell apoptosis and inhibition of cancer cell proliferation. VM is associated with tumor metastasis.[3] |
| Cell Assay |
Effects of CVM-1118 on tumor cell proliferation, percent viability and percent apoptosis[2] An analysis of the effect of CVM-1118 on the proliferation and percent viability, apoptosis and cell debris (representing dead cells) was performed on human melanoma cells either untreated (Control) or treated with 1, 10, 50 and 100 nM of CVM-1118 using flow cytometry with Guava Viacount and Nexin reagents. The melanoma cells (1 × 105) were treated with various concentrations of CVM-1118 for 24, 48 and 72 hr, and subsequently their proliferation, percent viability and percent apoptosis determined compared to untreated cells in triplicate. A representative finding shown in Figure 2 reveals a significant (p<0.05) reduction in proliferation by cells treated with 50 and 100 nM of CVM-1118 after 24 hr and in the cells treated with 10, 50 and 100 nM after 48 and 72 hr. While viability was significantly reduced by CVM-1118 at 10, 50 and 100 nM after 24, 48 and 72 hr of treatment, all concentrations of CVM-1118 (including 1 nM) increased the percent of apoptosis after 24 hr, and after 48 and 72 hr for the cells treated with 50 and 100 nM of CVM-1118. Inhibition of vascular mimicry by CVM-1118[2] To evaluate the effect of CVM-1118 on VM in vitro, standard VM assays were performed using 3-dimensional matrices prepared with Matrigel in 12-well culture dishes. Human melanoma cells (1 × 105 cells/well) were plated onto the matrices without (Control) or with CVM-1118 at concentrations of 1, 10 or 50 nM. VM tubular network formation was then observed after 24 hr and images captured digitally using an inverted microscope with a 10× objective (100× final magnification) and Hitachi HV-C20 CCD camera. The images from four different fields from the Control and treated cultures were then analyzed using the AngioSys software package assessing the number of junctions and tubules, as well as the total tubule length, determined for each field with the average, standard error and significance determined using Excel. Figure 3 shows representative images from the Control and each CVM-1118 treated group along with the resulting binary image generated for its subsequent analysis. The data demonstrate a dose dependent, statistically significant decrease in the number of junctions and average total tubule length (per observed field) in the samples treated with 1, 10 and 50 nM of CVM-1118 compared to the Control sample. While the ability of CVM-1118 to inhibit VM is clearly visible in the images, detailed analysis supports the concept that this inhibition is dose dependent and can be correlated with a breakdown in the cells' ability to form branching, tubular networks characteristic of VM. Pathways affected by CVM-1118[2] The key signaling pathways underlying VM described above and highlighted in Figure 1 were further interrogated after treatment with CVM-1118 in human melanoma cells. Using a combination of qRT-PCR analyses, together with protein arrays, changes were measured in human phospho-MAPK/phospho-kinases and cell stress checkpoints, in addition to apoptosis regulators. A compilation of the major findings generated in response to CVM-1118 treatment are presented as cascading and overlapping signaling pathways in Figure 4. The major overall effect of CVM-1118 on human melanoma cells at the mRNA level is down-regulation of the stem cell-associated genes Nodal (and downstream pSMAD2), Notch4 ICD and HES1, in addition to the vascular signaling-associated gene VEGF-A (annotated in Table 1) -- resulting in suppression of VM. Additional analyses of CVM-1118 treatment effects at the protein level revealed noteworthy increases in (phosphorylated, designated p-) p-HIF-1α, p-p27, and p-c-Jun, collectively demonstrating stabilization of proteins underlying the hypoxia response, cell cycle arrest, growth inhibition, and apoptotic events, respectively |
| References |
[1]. Design, synthesis, and preclinical evaluation of new 5,6- (or 6,7-) disubstituted-2-(fluorophenyl)quinolin-4-one derivatives as potent antitumor agents. J Med Chem. 2010 Nov 25;53(22):8047-58. [2]. Tumor cell vascular mimicry: Novel targeting opportunity in melanoma. Pharmacol Ther. 2016 Mar:159:83-92 [3]. https://pubchem.ncbi.nlm.nih.gov/compound/49840582 |
| Additional Infomation |
Foslinanib Disodium is the disodium salt form of foslinanib, an orally bioavailable agent with potential antineoplastic and anti-vasculogenic mimicry activities. Upon oral administration, foslinanib targets and inhibits the formation of vasculogenic mimicry (VM; vascular mimicry). By destroying the VM channels and network, cancer cells are devoid of perfusion leading to an induction of cancer cell apoptosis and inhibition of cancer cell proliferation. VM is associated with tumor metastasis. Foslinanib is under investigation in clinical trial NCT03600233 (Study of CVM-1118 for Patients With Advanced Neuroendocrine Tumors). Foslinanib is an orally bioavailable agent with potential antineoplastic and anti-vasculogenic mimicry activities. Upon oral administration, foslinanib targets and inhibits the formation of vasculogenic mimicry (VM; vascular mimicry). By destroying the VM channels and network, cancer cells are devoid of perfusion leading to an induction of cancer cell apoptosis and inhibition of cancer cell proliferation. VM is associated with tumor metastasis. In 1999, the American Journal of Pathology published an article, entitled “Vascular channel formation by human melanoma cells in vivo and in vitro: vasculogenic mimicry” by Maniotis and colleagues, which ignited a spirited debate for several years and earned the journal's distinction of a “citation classic” (Maniotis et al., 1999). Tumor cell vasculogenic mimicry (VM), also known as vascular mimicry, describes the plasticity of aggressive cancer cells forming de novo vascular networks and is associated with the malignant phenotype and poor clinical outcome. The tumor cells capable of VM share the commonality of a stem cell-like, transendothelial phenotype, which may be induced by hypoxia. Since its introduction as a novel paradigm for melanoma tumor perfusion, many studies have contributed new findings illuminating the underlying molecular pathways supporting VM in a variety of tumors, including carcinomas, sarcomas, glioblastomas, astrocytomas, and melanomas. Of special significance is the lack of effectiveness of angiogenesis inhibitors on tumor cell VM, suggesting a selective resistance by this phenotype to conventional therapy. Facilitating the functional plasticity of tumor cell VM are key proteins associated with vascular, stem cell, extracellular matrix, and hypoxia-related signaling pathways -- each deserving serious consideration as potential therapeutic targets and diagnostic indicators of the aggressive, metastatic phenotype. This review highlights seminal findings pertinent to VM, including the effects of a novel, small molecular compound, CVM-1118, currently under clinical development to target VM, and illuminates important molecular pathways involved in the suppression of this plastic, aggressive phenotype, using melanoma as a model.[2] This review of VM across a broad range of cancers provides only the highlights of key findings pertinent to its functional and translational relevance in association with an aggressive and metastatic phenotype. The molecular pathways underlying VM have illuminated new candidates for the development of innovative treatment strategies that target tumor cell plasticity and the metastatic properties affiliated with disease recurrence and drug resistance. Further confounding our ability to effectively eradicate cancer is the challenge associated with heterogeneous subpopulations comprising tumors along with a complex, diverse vasculature supply. Moreover, the unintended consequences of hypoxia induced by rapid tumor growth or by some conventional therapies may serve as a catalyst for the VM and cancer stem cell phenotype. Therefore, it seems prudent, and certainly timely, to consider the application of new agents, such as CVM-1118 -- to target VM pathways associated with the stem cell phenotype and resistant to most conventional agents. Further studies are warranted to expand the assessment of CVM-1118 in other aggressive cancers expressing VM to better understand its full potential. Targeting VM with specific molecular compounds used in a combinatorial manner with front-line therapies may hold the greatest promise in the war on cancer.[2] |
Solubility Data
| Solubility (In Vitro) | DMSO: ~7.86 mg/mL (~19.2 mM ) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4437 mL | 12.2187 mL | 24.4373 mL | |
| 5 mM | 0.4887 mL | 2.4437 mL | 4.8875 mL | |
| 10 mM | 0.2444 mL | 1.2219 mL | 2.4437 mL |