Physicochemical Properties
| Molecular Formula | C17H20N6O7S |
| Molecular Weight | 452.44 |
| Exact Mass | 452.111 |
| CAS # | 173159-57-4 |
| PubChem CID | 11419598 |
| Appearance |
Light beige solid Yellow-brownish solid, fine grained granule |
| Density | 1.471 g/cm3 |
| Melting Point | 0-6ºC |
| Index of Refraction | 1.613 |
| LogP | 2.527 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 31 |
| Complexity | 732 |
| Defined Atom Stereocenter Count | 0 |
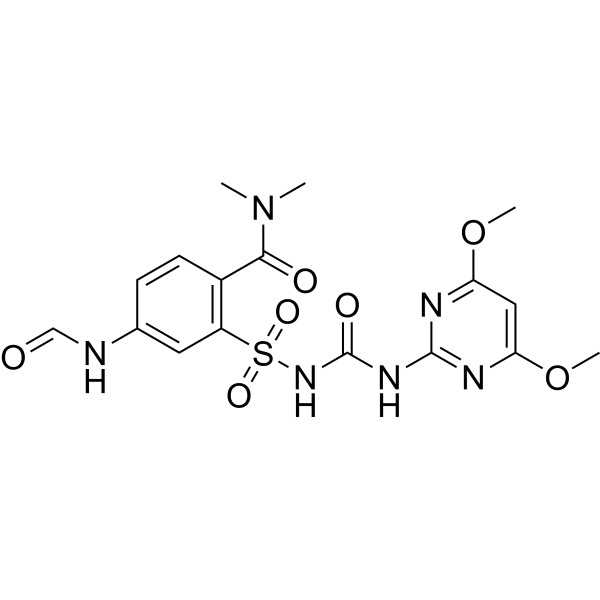
| SMILES | COC1C=C(OC)N=C(NC(NS(C2C=C(NC=O)C=CC=2C(N(C)C)=O)(=O)=O)=O)N=1 |
| InChi Key | PXDNXJSDGQBLKS-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H20N6O7S/c1-23(2)15(25)11-6-5-10(18-9-24)7-12(11)31(27,28)22-17(26)21-16-19-13(29-3)8-14(20-16)30-4/h5-9H,1-4H3,(H,18,24)(H2,19,20,21,22,26) |
| Chemical Name | 2-[(4,6-dimethoxypyrimidin-2-yl)carbamoylsulfamoyl]-4-formamido-N,N-dimethylbenzamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion 12 Sprague Dawley CRL:(IGS) CD BR rats per sex received single daily oral (gavage) doses of [14C]-phenyl AE F130360 at 10 mg/kg for up to 14 days. 3 /rats/ per sex were sacrificed 24 hours after 1, 9, and 14 days of treatment. The majority of tissues were found to have residue levels below 0.01 ug AE F130360/g 24 hours after a single oral dose. Liver contained the highest concentrations of residues (0.079 ug/g and 0.114 ug/g for males and females respectively). 24 hours after the last of 14 daily doses, residues in tissues were mostly below 0.03 ug/g, except for testes (0.073 ug/g), skin (0.042 ug/g in males and 0.166 ug/g in females), and liver (0.222 ug/g and 0.280 ug/g for males and females respectively). Elimination of [14C]-phenyl AE F130360 and its metabolites 48 hours after repeated daily dosing for 14 days was mainly in the feces where 60.99 +/- 22.19% (males) and 88.4 +/- 5.21% (females) of recovered radioactivity was found. Urine was a minor route of elimination. 4.00% (males) and 5.27% (females) was present as parent compound, AE F130360, 48 hours after daily dosing for 14 days. Cleavage product AE F153745 (4-formylamino-N,N-dimethyl-2-sulfamoyl-benzamide) accounted for 4.08% (males) and 2.41% (females), and, the free amine, AE F130619 (4-amino-2-3[-(4, 6-dimethoxypyrimidin-2-yl)-ureidosulfonyl)-N,N dimethylbenzamide), made up 3.50% (males) and 1.65% (females) of recovered label. 6 bile duct cannulated male Sprague Dawley (Crl:CD BR) rats received a single oral (gavage) dose of AE F130360 [phenyl-U-14C] at 10 mg/kg. Four animals were selected for evaluation based on the production of bile. Urine and feces along with cage debris and cage washings were collected pre-dose, 24, and 48 hours post-dosing. Bile was collected at pre-dose, 2, 4, 6, 12, 24, and 48 hours post-treatment. Animals were then sacrificed and the radioactivity as percentage of administered dose was quantified for the carcass. Approximately 17% of the administered dose was absorbed. The largest part of that was found in the urine (12.67% +/- 3.7). Bile accounted for about a quarter (4.198% +/- 1.872). The majority of the administered dose was not absorbed, 75.63% +/- 10.64 was excreted in the feces. Metabolism / Metabolites 4 Sprague Dawley CRL:CD (SD) BR rats per sex per group received a single oral (gavage) dose of [14C]-phenyl labeled AE F130360 at 10 and 1000 mg/kg. Another 2 animals per sex were dosed by gavage with [14C]-pyrimidyl labeled AE F130360 at 10 mg/kg. Urine and feces were collected separately at 6 (urine only), 12 (urine only), 24, 48, and 72 hours post-dosing for quantification of radioactivity. Expired air was not sampled since preliminary work indicated this was a minor route of excretion. The majority of the administered dose irrespective of dose level, sex, or radiolabel was excreted as unchanged parent compound in the feces. Over the 72 hour period following dosing with [14C]-phenyl labeled AE F130360 at 10 mg/kg, 73.999% (males) and 72.337% (females) of the administered dose was found in feces as AE F130360. Cleavage product AE F153745 (4-formylamino-N,N-dimethyl-2-sulfamoyl-benzamide) accounted for 8.417% and 8.671% in males and females respectively, and, a polar unknown, made up 0.176% and 0.095% respectively. The majority was excreted during the first 24 hours, except for the polar unknown, most of which was found at 48 hours. Cleavage product AE F153745 was the most prevalent metabolite in urine, accounting for 2.250% (males) and 2.300% (females) of administered phenyl radiolabel (10 mg/kg) over 72 hours. Parent compound, AE F130360, was 1.723% (males) and 2.128% (females). Free amine, AE F130619 (4-amino-2-3[-(4, 6-dimethoxypyrimidin-2-yl)-ureidosulfonyl)-N,N dimethylbenzamide), was found as 0.831% (males) and 0.777% (females) of administered dose. The polar unknown was present at 0.023% for both sexes. The majority was excreted 6 hours post-dosing. The polar unknown was found at 48 hours. At 1000 mg/kg of [14C]-phenyl labeled AE F130360, 80.360% (males) and 77.732% (females) was excreted in feces as AE F130360 over 72 hours. The polar unknown accounted for 5.856% (males) and 5.637% (females); AE F153745 for 3.381% (males) and 1.264% (females); and AE F130619 for 0.489% (males) and 2.773% (females). The majority of each compound was excreted 24 hours post dosing. ... |
| Toxicity/Toxicokinetics |
Non-Human Toxicity Values LD50 Rat oral (foramsulfuron technical) >5000 mg/kg LD50 Rat dermal (foramsulfuron technical) >2000 mg/kg LD50 Rat oral (Option corn herbicide) 3881 mg/kg LC50 Rat inhalation (Option herbicide) >5.32 mg/L (determined on milled granules) |
| References |
[1]. Proper adjuvant selection for foramsulfuron activity.Crop Protection, 2004, 23(4):361-366. |
| Additional Infomation |
Foramsulfuron is a member of the class of benzamides that is N,N-dimethylbenzamide substituted by a formylamino group at position 4 and a [(4,6-dimethoxypyrimidin-2-yl)carbamoyl]sulfamoyl group at position 2. It has a role as a xenobiotic, an environmental contaminant and a herbicide. It is a sulfonamide, a member of pyrimidines, an aromatic ether, a member of ureas and a member of benzamides. Mechanism of Action Foramsulfuron inhibits the synthesis of amino acid in plants through inhibition of acetolactate synthase (ALS). This process results in slow or stunted plant growth and/or ultimate plant death. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2102 mL | 11.0512 mL | 22.1024 mL | |
| 5 mM | 0.4420 mL | 2.2102 mL | 4.4205 mL | |
| 10 mM | 0.2210 mL | 1.1051 mL | 2.2102 mL |