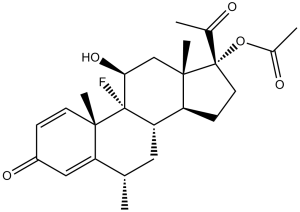
Fluorometholone Acetate (formerly NSC47438; NSC-47438; U 17,323; AI 3-52814; Flarex) is a synthetic corticosteroid that has been approved as a medication commonly used as eye drops in the treatment of steroid responsive inflammatory conditions of the eye.
Physicochemical Properties
| Molecular Formula | C24H31FO5 | |
| Molecular Weight | 418.5 | |
| Exact Mass | 418.215 | |
| CAS # | 3801-06-7 | |
| Related CAS # |
|
|
| PubChem CID | 240767 | |
| Appearance | White to off-white solid powder | |
| Density | 1.2±0.1 g/cm3 | |
| Boiling Point | 532.1±50.0 °C at 760 mmHg | |
| Melting Point | 230-232° | |
| Flash Point | 275.6±30.1 °C | |
| Vapour Pressure | 0.0±3.2 mmHg at 25°C | |
| Index of Refraction | 1.556 | |
| LogP | 2.57 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 30 | |
| Complexity | 892 | |
| Defined Atom Stereocenter Count | 8 | |
| SMILES | C[C@H]1C[C@H]2[C@@H]3CC[C@@]([C@]3(C[C@@H]([C@@]2([C@@]4(C1=CC(=O)C=C4)C)F)O)C)(C(=O)C)OC(=O)C |
|
| InChi Key | YRFXGQHBPBMFHW-SBTZIJSASA-N | |
| InChi Code | 1S/C24H31FO5/c1-13-10-19-17-7-9-23(14(2)26,30-15(3)27)22(17,5)12-20(29)24(19,25)21(4)8-6-16(28)11-18(13)21/h6,8,11,13,17,19-20,29H,7,9-10,12H2,1-5H3/t13-,17-,19-,20-,21-,22-,23-,24-/m0/s1 | |
| Chemical Name | 9-Fluoro-11beta,17-dihydroxy-6alpha-methylpregna-1,4-diene-3,20-dione 17-acetate | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Human Carbonic Anhydrase Isoform I (hCA I): Fluorometholone Acetate showed inhibitory activity against hCA I with a Ki value of 1.2 μM [2] - Human Carbonic Anhydrase Isoform II (hCA II): Fluorometholone Acetate exhibited inhibitory activity against hCA II with a Ki value of 0.8 μM [2] |
| ln Vitro |
Lineweaver-Burk plots for fluorometholone acetate yield a Ki value of 1.044 μM (noncompetitive) for hCA-I and 9.98 μM (noncompetitive) for hCA-II[2]. Inhibition of Human Carbonic Anhydrase (hCA) Activity: Fluorometholone Acetate inhibited the esterase activity of purified recombinant hCA I and hCA II in a concentration-dependent manner. At a concentration of 5 μM, it inhibited hCA I activity by 65% and hCA II activity by 78% compared to the vehicle control. The inhibition was reversible, as removing the drug via dialysis restored 80% of hCA II activity [2] |
| ln Vivo |
In New Zealand albino rabbits weighing 1.5–2.0 kg, an artificially induced inflammatory keratitis resulted in an average 47% reduction in the polymorphonuclear leukocytes invading the cornea when 0.1% fluorometholone acetate ophthalmic suspension was applied topically every hour. When given at three-hour intervals, 0.1% fluorometholone acetate, produced as a high-viscosity carbomer gel, reduced invading leukocytes in the cornea by an average of 48%; this impact was not statistically different from when the suspension was administered hourly[1]. Efficacy in Human External Ocular Inflammation: In a clinical study of 120 patients with external ocular inflammation (e.g., conjunctivitis, blepharitis), topical administration of Fluorometholone Acetate (0.1% ophthalmic suspension) 4 times daily for 14 days resulted in complete resolution of redness in 75% of patients, reduction of itching in 82% of patients, and elimination of ocular discharge in 78% of patients. The response was observed as early as day 3 in 40% of patients [3] |
| Enzyme Assay |
Human Carbonic Anhydrase (hCA) Esterase Activity Assay: The assay was performed in a 200 μL reaction system containing 50 mM Tris-HCl buffer (pH 7.4), 1 mM p-nitrophenyl acetate (substrate), and purified recombinant hCA I or hCA II. Fluorometholone Acetate was added at concentrations ranging from 0.1 to 10 μM, and the mixture was incubated at 25°C for 10 minutes. The hydrolysis of p-nitrophenyl acetate was monitored by measuring the absorbance at 405 nm every minute for 5 minutes. The enzyme activity was calculated based on the slope of the absorbance change, and the Ki value was determined by nonlinear regression analysis of the inhibition curve [2] |
| Animal Protocol |
i.p. mice and rats |
| Toxicity/Toxicokinetics |
Ocular Local Toxicity in Humans: In the clinical study [3], topical use of Fluorometholone Acetate (0.1% ophthalmic suspension) was associated with mild ocular side effects, including transient stinging (12% of patients), blurred vision (8% of patients), and increased intraocular pressure (IOP) (5% of patients). The elevated IOP returned to baseline within 7 days after discontinuing the drug, and no severe ocular or systemic toxicity (e.g., liver, kidney damage) was observed [3] |
| References |
[1]. Fluorometholone acetate. A new ophthalmic derivative of fluorometholone. Arch Ophthalmol. 1982 Apr;100(4):640-1. [2]. Some Anti-Inflammatory Agents Inhibit Esterase Activities of Human Carbonic Anhydrase Isoforms I and II: An In Vitro Study. Chem Biol Drug Des. 2015 Oct;86(4):857-63. [3]. Fluoro metholone acetate: clinical evaluation in the treatment of external ocular inflammation. Ann Ophthalmol. 1984 Dec;16(12):1110-5. |
| Additional Infomation |
Fluorometholone acetate is a steroid ester resulting from the formal condensation of the 17-hydroxy function of fluorometholone with acetic acid. Used in the treatment of steroid responsive inflammatory conditions of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the eye. It has a role as an anti-inflammatory drug. It is a steroid ester, a glucocorticoid, a 3-oxo-Delta(1),Delta(4)-steroid, a 20-oxo steroid, an 11beta-hydroxy steroid, an acetate ester and a fluorinated steroid. It is functionally related to a Delta(1)-progesterone and a fluorometholone. Fluorometholone Acetate is the acetate salt form of fluorometholone, a synthetic glucocorticoid with anti-inflammatory and anti-allergic properties. Fluorometholone acetate exerts its effect by interacting with cytoplasmic glucocorticoid receptors and subsequently activates glucocorticoid receptor mediated gene expression. The synthesis of certain anti-inflammatory proteins is induced while the synthesis of certain inflammatory mediators is inhibited. As a result, there is an overall reduction in chronic inflammation and autoimmune reactions. See also: Fluorometholone (has active moiety); Fluorometholone acetate; tobramycin (component of). Drug Classification & Background: Fluorometholone Acetate is a synthetic glucocorticoid derivative specifically developed for ophthalmic use, with improved ocular tissue penetration compared to the parent compound Fluorometholone [1] - Indication: It is indicated for the treatment of external ocular inflammation, including allergic conjunctivitis, blepharitis, and keratitis, by suppressing ocular inflammatory responses [1][3] - Mechanism of Anti-Inflammatory Action: In addition to inhibiting carbonic anhydrases, Fluorometholone Acetate exerts anti-inflammatory effects by reducing the migration of inflammatory cells (e.g., neutrophils, lymphocytes) to the ocular surface and downregulating the expression of pro-inflammatory mediators (e.g., prostaglandins, leukotrienes) [3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.97 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.97 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.97 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3895 mL | 11.9474 mL | 23.8949 mL | |
| 5 mM | 0.4779 mL | 2.3895 mL | 4.7790 mL | |
| 10 mM | 0.2389 mL | 1.1947 mL | 2.3895 mL |