Physicochemical Properties
| Molecular Formula | C27H33N9O15P2 |
| Molecular Weight | 785.549820000001 |
| Exact Mass | 785.157 |
| CAS # | 146-14-5 |
| Related CAS # | Flavin adenine dinucleotide disodium salt;84366-81-4;Flavin adenine dinucleotide-13C5 ammonium |
| PubChem CID | 643975 |
| Appearance | Yellow to orange solid powder |
| Density | 2.1±0.1 g/cm3 |
| Index of Refraction | 1.850 |
| LogP | -2.93 |
| Hydrogen Bond Donor Count | 9 |
| Hydrogen Bond Acceptor Count | 20 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 53 |
| Complexity | 1560 |
| Defined Atom Stereocenter Count | 7 |
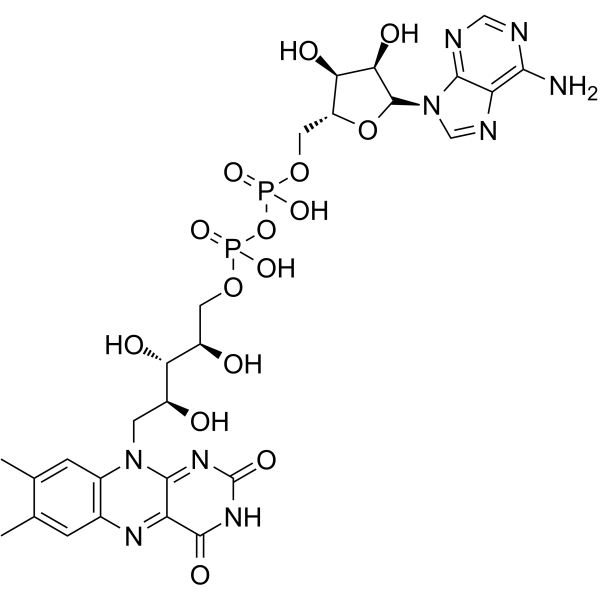
| SMILES | CC1=CC2=C(C=C1C)N(C3=NC(=O)NC(=O)C3=N2)C[C@@H]([C@@H]([C@@H](COP(=O)(O)OP(=O)(O)OC[C@@H]4[C@H]([C@H]([C@@H](O4)N5C=NC6=C(N=CN=C65)N)O)O)O)O)O |
| InChi Key | VWWQXMAJTJZDQX-UYBVJOGSSA-N |
| InChi Code | InChI=1S/C27H33N9O15P2/c1-10-3-12-13(4-11(10)2)35(24-18(32-12)25(42)34-27(43)33-24)5-14(37)19(39)15(38)6-48-52(44,45)51-53(46,47)49-7-16-20(40)21(41)26(50-16)36-9-31-17-22(28)29-8-30-23(17)36/h3-4,8-9,14-16,19-21,26,37-41H,5-7H2,1-2H3,(H,44,45)(H,46,47)(H2,28,29,30)(H,34,42,43)/t14-,15+,16+,19-,20+,21+,26+/m0/s1 |
| Chemical Name | [[(2R,3S,4R,5R)-5-(6-aminopurin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl] [(2R,3S,4S)-5-(7,8-dimethyl-2,4-dioxobenzo[g]pteridin-10-yl)-2,3,4-trihydroxypentyl] hydrogen phosphate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Poly(flavin adenine dinucleotide, FAD) features an additional polymer-type redox reaction and is a highly efficient electrocatalyst for NADH oxidation: at the lowest potential reported for a NADH sensor (0.00 V, pH 7.4) Working, the characteristics of poly(FAD) are: the electrochemical rate constant is 1.8±0.6×10-3 cm/s, which is at the level of NADH mass transfer constant. Poly(FAD) modified electrodes are characterized by greatly enhanced stability and are the most advantageous NADH sensors in analytical chemistry [2]. |
| ln Vitro | Poly(flavin adenine dinucleotide, FAD) features an additional polymer-type redox reaction and is a highly efficient electrocatalyst for NADH oxidation: at the lowest potential reported for a NADH sensor (0.00 V, pH 7.4) Working, the characteristics of poly(FAD) are: the electrochemical rate constant is 1.8±0.6×10-3 cm/s, which is at the level of NADH mass transfer constant. Poly(FAD) modified electrodes are characterized by greatly enhanced stability and are the most advantageous NADH sensors in analytical chemistry [2]. |
| ln Vivo |
Intravenous flavin adenine dinucleotide (2 mg/kg) dramatically reverses the ventricular fibrillation threshold (VFT) reduction caused by chlorpromazine (CPZ). Canine heart mitochondria are not affected by CPZ when flavin adenine dinucleotide is added. Dogs receiving a flavin adenine dinucleotide injection within 10 minutes of the administration exhibited a brief hypotension before their blood pressure returned to normal. Moreover, flavin adenine dinucleotide can stop mitochondrial dysfunction brought on by chlorpromazine [1]. In anesthetized adult mongrel dogs, prior intravenous administration of FAD (2 mg/Kg) significantly protected against chlorpromazine (CPZ, 1 mg/Kg i.v.)-induced decrease in ventricular fibrillation threshold (VFT). The VFT decreased to 56.6±5.4% of the initial level after CPZ alone, but only to 92.2±4.2% when FAD was administered before CPZ.[1] Prior administration of FAD (2 mg/Kg i.v.) also attenuated the hypotensive effect of CPZ. The drop in systolic and diastolic blood pressure 40 minutes after CPZ was significantly less severe in the FAD-pretreated group compared to the group receiving CPZ alone.[1] Heart mitochondria isolated from dogs treated with CPZ alone showed significantly decreased respiratory control index (RCI) and ADP/O. Prior administration of FAD (2 mg/Kg i.v.) partially prevented this CPZ-induced mitochondrial dysfunction, resulting in significantly higher RCI and ADP/O values compared to the CPZ-only group.[1] |
| Enzyme Assay |
Mitochondrial function assays were performed to assess the effects of FAD and CPZ. Mitochondria were isolated from the hearts of intact dogs. The respiratory control index (RCI) and ADP/O were determined using a closed assay system containing a mannitol reaction mixture, mitochondrial sample, potassium succinate, and ADP as substrates. Oxygen consumption was monitored with an oxygen electrode.[1] To assess direct in vitro effects, RCI and ADP/O were measured under three conditions using mitochondria from untreated dogs: (1) control (no drug), (2) with addition of CPZ (50 μg), and (3) with addition of FAD (100 μg) followed by CPZ (50 μg) to the reaction mixture. CPZ alone significantly decreased both RCI and ADP/O. When FAD was added before CPZ, the decreases in RCI and ADP/O were significantly attenuated, demonstrating a protective effect of FAD against CPZ-induced mitochondrial dysfunction in vitro.[1] |
| Animal Protocol |
Adult mongrel dogs (8-12 Kg) were anesthetized with intraperitoneal pentobarbital sodium (50 mg/Kg) and placed under artificial respiration. A thoracotomy was performed to expose the heart. A stimulating electrode was attached to the left ventricular apex for VFT measurement. Blood pressure was monitored via a carotid artery catheter. Femoral vein catheters were inserted for drug administration and blood sampling.[1] Dogs were divided into three groups (n=6 each). Group I (Control): received intravenous saline (2 ml/Kg), followed 10 minutes later by another saline injection (1 ml/Kg). Group II (CPZ): received intravenous saline (2 ml/Kg), followed 10 minutes later by intravenous CPZ (1 mg/Kg). Group III (FAD+CPZ): received intravenous FAD (2 mg/Kg), followed 10 minutes later by intravenous CPZ (1 mg/Kg). All solutions were administered over 1-2 minutes.[1] VFT, heart rate, and blood pressure were recorded at intervals. Blood samples were taken for serum K+ and pH measurement. Forty minutes after the first injection, the heart was isolated, and myocardial mitochondria were prepared for functional assays.[1] |
| Toxicity/Toxicokinetics |
The study reports that dogs showed a transient hypotension within 10 minutes after the intravenous injection of FAD (2 mg/Kg), after which blood pressure recovered to the initial level. No other adverse effects or toxicities of FAD were described.[1] |
| References |
[1]. Protection of chlorpromazine-induced arrhythmia by flavin-adenine-dinucleotide in canine heart. Jpn Heart J. 1979 Sep;20(5):657-65. [2]. Electropolymerized flavin adenine dinucleotide as an advanced NADH transducer. Anal Chem. 2004 Apr 1;76(7):2004-9. |
| Additional Infomation |
FAD is a flavin adenine dinucleotide in which the substituent at position 10 of the flavin nucleus is a 5'-adenosyldiphosphoribityl group. It has a role as a human metabolite, an Escherichia coli metabolite, a mouse metabolite, a prosthetic group and a cofactor. It is a vitamin B2 and a flavin adenine dinucleotide. It is a conjugate acid of a FAD(3-). A condensation product of riboflavin and adenosine diphosphate. The coenzyme of various aerobic dehydrogenases, e.g., D-amino acid oxidase and L-amino acid oxidase. (Lehninger, Principles of Biochemistry, 1982, p972) Flavin adenine dinucleotide is approved for use in Japan under the trade name Adeflavin as an ophthalmic treatment for vitamin B2 deficiency. Flavin adenine dinucleotide has been reported in Eremothecium ashbyi, Drosophila melanogaster, and other organisms with data available. FAD is a metabolite found in or produced by Saccharomyces cerevisiae. A condensation product of riboflavin and adenosine diphosphate. The coenzyme of various aerobic dehydrogenases, e.g., D-amino acid oxidase and L-amino acid oxidase. (Lehninger, Principles of Biochemistry, 1982, p972) See also: ... View More ... Drug Indication Used to treat eye diseases caused by vitamin B2 deficiency, such as keratitis and blepharitis. Flavin Adenine Dinucleotide (FAD) was investigated based on prior in vitro findings that it specifically combines with chlorpromazine (CPZ).[1] The study suggests that the arrhythmogenic and hypotensive actions of CPZ might be associated, at least in part, with impaired mitochondrial function. FAD is proposed to protect against these effects by competitively binding with CPZ, given the structural similarity between the two molecules, thereby preventing CPZ from inhibiting mitochondrial flavin-containing enzymes and other components.[1] The authors propose that FAD might be useful in the treatment of cardiac disturbances associated with overdosage of CPZ.[1] |
Solubility Data
| Solubility (In Vitro) | H2O : ~125 mg/mL (~159.12 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.2730 mL | 6.3650 mL | 12.7299 mL | |
| 5 mM | 0.2546 mL | 1.2730 mL | 2.5460 mL | |
| 10 mM | 0.1273 mL | 0.6365 mL | 1.2730 mL |