Physicochemical Properties
| Molecular Formula | C38H64N8O15 |
| Molecular Weight | 872.95900 |
| Exact Mass | 872.449 |
| CAS # | 136466-51-8 |
| PubChem CID | 9919150 |
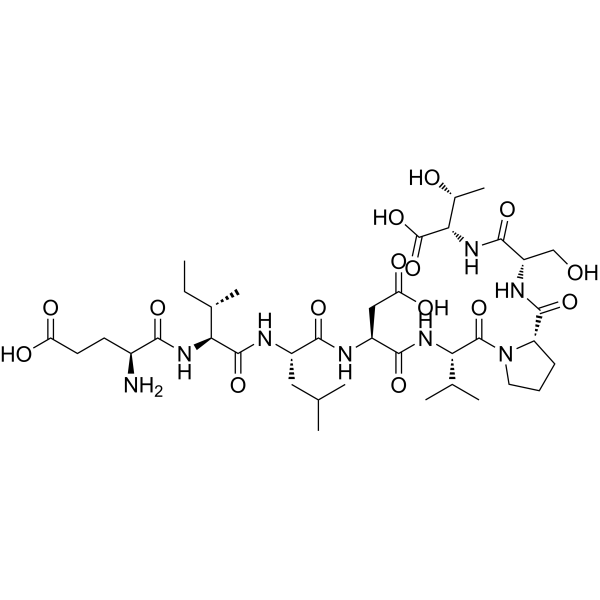
| Sequence | H-Glu-Ile-Leu-Asp-Val-Pro-Ser-Thr-OH; Glu-Ile-Leu-Asp-Val-Pro-Ser-Thr; L-alpha-glutamyl-L-isoleucyl-L-leucyl-L-alpha-aspartyl-L-valyl-L-prolyl-L-seryl-L-threonine |
| SequenceShortening | EILDVPST; H-EILDVPST-OH |
| Appearance | Typically exists as solid at room temperature |
| LogP | -3.9 |
| Hydrogen Bond Donor Count | 12 |
| Hydrogen Bond Acceptor Count | 16 |
| Rotatable Bond Count | 26 |
| Heavy Atom Count | 61 |
| Complexity | 1590 |
| Defined Atom Stereocenter Count | 10 |
| SMILES | CC[C@@H]([C@H](NC([C@@H](N)CCC(O)=O)=O)C(N[C@H](C(N[C@H](C(N[C@H](C(N1CCC[C@H]1C(N[C@H](C(N[C@H](C(O)=O)[C@H](O)C)=O)CO)=O)=O)C(C)C)=O)CC(O)=O)=O)CC(C)C)=O)C |
| InChi Key | FLNMCNFAJCMMHI-YGHIGYJTSA-N |
| InChi Code | InChI=1S/C38H64N8O15/c1-8-19(6)29(44-31(53)21(39)11-12-26(49)50)36(58)41-22(14-17(2)3)32(54)40-23(15-27(51)52)33(55)43-28(18(4)5)37(59)46-13-9-10-25(46)35(57)42-24(16-47)34(56)45-30(20(7)48)38(60)61/h17-25,28-30,47-48H,8-16,39H2,1-7H3,(H,40,54)(H,41,58)(H,42,57)(H,43,55)(H,44,53)(H,45,56)(H,49,50)(H,51,52)(H,60,61)/t19-,20+,21-,22-,23-,24-,25-,28-,29-,30-/m0/s1 |
| Chemical Name | (4S)-4-amino-5-[[(2S,3S)-1-[[(2S)-1-[[(2S)-3-carboxy-1-[[(2S)-1-[(2S)-2-[[(2S)-1-[[(1S,2R)-1-carboxy-2-hydroxypropyl]amino]-3-hydroxy-1-oxopropan-2-yl]carbamoyl]pyrrolidin-1-yl]-3-methyl-1-oxobutan-2-yl]amino]-1-oxopropan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-3-methyl-1-oxopentan-2-yl]amino]-5-oxopentanoic acid |
| Synonyms | Fibronectin CS1 Peptide; 136466-51-8; (4S)-4-amino-5-[[(2S,3S)-1-[[(2S)-1-[[(2S)-3-carboxy-1-[[(2S)-1-[(2S)-2-[[(2S)-1-[[(1S,2R)-1-carboxy-2-hydroxypropyl]amino]-3-hydroxy-1-oxopropan-2-yl]carbamoyl]pyrrolidin-1-yl]-3-methyl-1-oxobutan-2-yl]amino]-1-oxopropan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-3-methyl-1-oxopentan-2-yl]amino]-5-oxopentanoic acid; Glu-Ile-Leu-Asp-Val-Pro-Ser-Thr; Fibronectin CS-1 Fragment (1978-1985) trifluoroacetate salt; SCHEMBL8937022; CHEMBL3220763; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Cancer cell attachment |
| ln Vitro | We have investigated the antimetastatic effect of synthetic or recombinant peptides containing the functional domains of fibronectin on experimental and spontaneous lung metastases of murine tumor cells. CS1 peptide which is present within type III homology connecting segment (IIICS) as well as C-274 (cell-binding domain) were able to inhibit experimental lung metastasis when co-injected intravenously (iv) with B16-BL6 melanoma cells, while H-271 (heparin-binding domain) could not. In the spontaneous metastasis model, multiple iv administrations of CS1 or C-274 after surgical excision of primary tumors caused a significant reduction of metastatic colonies in the lung. Both CS1 and C-274 significantly inhibited cell adhesion and migration to fibronectin-coated substrates when added freely in solution. CS1 peptide also inhibited the cell adhesion and migration to laminin-coated substrates, but C-274 did not. H-271 did not have any inhibitory effect on cell adhesion or migration to either of the substrates. Similarly, CS1 inhibited tumor invasion to both Matrigel/fibronectin- and Matrigel/laminin-coated filters, whereas C-274 inhibited the invasion to only Matrigel/fibronectin-coated filter. These results indicate that CS1 peptide of fibronectin, lacking the Arg-Gly-Asp-containing domain, actively inhibits tumor metastases in spontaneous and experimental metastasis models. The use of such a peptide might offer a promising therapeutic approach for combatting or preventing cancer metastasis [1]. |
| References |
[1]. Inhibition of lung metastasis by synthetic and recombinant fragments of human fibronectin with functional domains. Jpn J Cancer Res. 1990 Oct;81(10):1003-11. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1455 mL | 5.7276 mL | 11.4553 mL | |
| 5 mM | 0.2291 mL | 1.1455 mL | 2.2911 mL | |
| 10 mM | 0.1146 mL | 0.5728 mL | 1.1455 mL |