Physicochemical Properties
| Molecular Formula | C49H75F3N16O12 |
| Molecular Weight | 1137.22 |
| Exact Mass | 1136.570246 |
| PubChem CID | 145712428 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 14 |
| Hydrogen Bond Acceptor Count | 18 |
| Rotatable Bond Count | 28 |
| Heavy Atom Count | 80 |
| Complexity | 2100 |
| Defined Atom Stereocenter Count | 9 |
| SMILES | CC[C@H](C)[C@@H](C(=O)O)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H]1CCCN1C(=O)[C@@H]2CCCN2C(=O)[C@H](CCC(=O)N)NC(=O)[C@H](CC3=CNC4=CC=CC=C43)N.C(=O)(C(F)(F)F)O |
| InChi Key | FNPDJQACMVRUIN-INPFXZFGSA-N |
| InChi Code | InChI=1S/C47H74N16O10.C2HF3O2/c1-4-25(2)37(45(72)73)61-41(68)32(14-8-20-55-47(52)53)58-38(65)26(3)57-40(67)31(13-7-19-54-46(50)51)59-42(69)34-15-9-21-62(34)44(71)35-16-10-22-63(35)43(70)33(17-18-36(49)64)60-39(66)29(48)23-27-24-56-30-12-6-5-11-28(27)30;3-2(4,5)1(6)7/h5-6,11-12,24-26,29,31-35,37,56H,4,7-10,13-23,48H2,1-3H3,(H2,49,64)(H,57,67)(H,58,65)(H,59,69)(H,60,66)(H,61,68)(H,72,73)(H4,50,51,54)(H4,52,53,55);(H,6,7)/t25-,26-,29-,31-,32-,33-,34-,35-,37-;/m0./s1 |
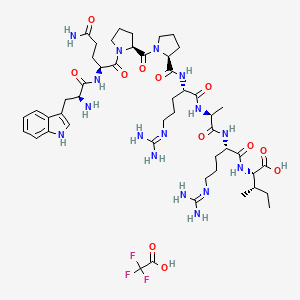
| Chemical Name | (2S,3S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-1-[(2S)-1-[(2S)-5-amino-2-[[(2S)-2-amino-3-(1H-indol-3-yl)propanoyl]amino]-5-oxopentanoyl]pyrrolidine-2-carbonyl]pyrrolidine-2-carbonyl]amino]-5-(diaminomethylideneamino)pentanoyl]amino]propanoyl]amino]-5-(diaminomethylideneamino)pentanoyl]amino]-3-methylpentanoic acid;2,2,2-trifluoroacetic acid |
| Synonyms | Fibronectin Adhesion-promoting Peptide (TFA); |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Cell Adhesion |
| ln Vitro |
GRGDS and fibronectin pro-adhesion peptide (heparin-binding peptide: WQPPRARI or Fibronectin Adhesion-promoting Peptide) micropatterning promotes endothelialization of prosthetic materials in vitro. Fibronectin pro-adhesion peptides also improve tactotactic cell movement. The anti-apoptotic activity of fibronectin requires the presence of the Fibronectin Adhesion-promoting Peptide/WQPPRARI sequence, which may be owing to the cooperative survival signaling mediated by this sequence and the RGD motif [1]. RDGS and fibronectin pro-adhesion peptides increase cell adhesion by separate activation pathways, consequently inducing diverse kinds of adhesion, which can be highlighted by study of focal and fibrillar contacts [2].
The long-term patency rates of vascular grafts and stents are limited by the lack of surface endothelialisation of the implanted materials. We have previously reported that GRGDS and Fibronectin Adhesion-promoting Peptide/WQPPRARI peptide micropatterns increase the endothelialisation of prosthetic materials in vitro. To investigate the mechanisms by which the peptide micropatterns affect endothelial cell adhesion and proliferation, a TAMRA fluorophore-tagged RGD peptide was designed. Live cell imaging revealed that the micropatterned surfaces led to directional cell spreading dependent on the location of the RGD-TAMRA spots. Focal adhesions formed within 3 h on the micropatterned surfaces near RGD-TAMRA spot edges, as expected for cell regions experiencing high tension. Similar levels of focal adhesion kinase phosphorylation were observed after 3 h on the micropatterned surfaces and on surfaces treated with RGD-TAMRA alone, suggesting that partial RGD surface coverage is sufficient to elicit integrin signaling. Lastly, endothelial cell expansion was achieved in serum-free conditions on gelatin-coated, RGD-TAMRA treated or micropatterned surfaces. These results show that these peptide micropatterns mainly impacted cell adhesion kinetics rather than cell proliferation. This insight will be useful for the optimization of micropatterning strategies to improve vascular biomaterials.[1] HSVEC behavior under physiological shear stress in vitro is investigated on PET surfaces micropatterned with both RGDS and Fibronectin Adhesion-promoting Peptide/WQPPRARI peptides. This technique allows (i) creating geometries on surface to guide cell orientation under shear stress and (ii) controlling surface chemical composition in order to modulate cell behavior. Under shear stress, endothelial cells adhere on patterned PET surfaces and present a more rapid orientation in flow direction in comparison to cells cultured on homogeneous surfaces. Micropatterned surfaces presenting a large surface area ratio of RGDS/WQPPRARI peptides induce fibrillar adhesion, while surfaces presenting an equal RGDS/WQPPRARI peptides surface area ratio preferentially induce focal adhesion [2]. |
| Cell Assay |
Covalent Grafting of Fibronectin Adhesion-promoting Peptide/WQPPRARI and RGDS Peptides with PET COOH Surface Moieties [2] Peptide grafting was achieved as previously described. The PET film was subjected to an ethanol washing and then to sonification for purification purposes. The three subsequent modification steps are detailed elsewhere.Briefly: 1. Materials were modified by means of hydrolysis and oxidation in order to promote the formation COOH functionalities on the PET surface, 2. The PET-COOH material was then immersed into a solution of EDC (0.2 M) + NHS (0.1 M) + MES buffer (0.1 M) in MilliQ Water, 3. The immobilization of cell-binding peptides was realized in a solution of Fibronectin Adhesion-promoting Peptide/WQPPRARI peptides, RGDS peptides, or Fibronectin Adhesion-promoting Peptide/WQPPRARI + RGDS (50:50, C/C) peptides (10−3 M in PBS) for 15 h at room temperature. After grafting, the materials were rinsed in MilliQ Water during 1 week under shaking. Micropatterned Surfaces Elaboration by Photolithography [2] Elaboration of micropatterned surfaces by photolithography is presented in Figure 1. Photosensitive resin was homogeneously spin-coated onto the PET-COOH (1 µm thick) and then dried. A photoresist pattern was created by first placing a mask containing a lithographically defined metal grid pattern in contact with the photoresist film, which was exposed to UV light filtered through the mask (15 s, 220 V, 60 W). The pattern was completed by immersing the substrate into a commercially available developer solution [Microposit Developer (50% in water)], which removed the exposed photoresist and left a photoresist pattern surrounded by bare substrate. PET materials were further dried and NHS molecules were covalently bound to the exposed regions of the surface. Then, Fibronectin Adhesion-promoting Peptide/WQPPRARI peptides were covalently grafted onto the polymer surface. The photoresist pattern was then removed after 1 min submersion in acetone revealing the underlying substrate. Surfaces were functionalized again by NHS molecules before grafting RGDS peptides. Patterns obtained are presented in Figure 2. |
| References |
[1]. A fluorophore-tagged RGD peptide to control endothelial cell adhesion to micropatterned surfaces. Biomaterials. 2014 Jan;35(3):879-90. [2]. Chollet C, et al. Impact of peptide micropatterning on endothelial cell actin remodeling for cell alignment under shear stress. Macromol Biosci. 2012 Dec;12(12):1648-59. [3]. Combination of Heparin Binding Peptide and Heparin Cell Surface Coatings for Mesenchymal Stem Cell Spheroid Assembly. Bioconjug Chem. 2018 Apr 18;29(4):878-884. |
| Additional Infomation | Microtissues containing multiple cell types have been used in both in vitro models and in vivo tissue repair applications. However, to improve throughput, there is a need to develop a platform that supports self-assembly of a large number of 3D microtissues containing multiple cell types in a dynamic suspension system. Thus, the objective of this study was to exploit the binding interaction between the negatively charged glycosaminoglycan, heparin, and a known heparin binding peptide to establish a method that promotes assembly of mesenchymal stem cell (MSC) spheroids into larger aggregates. We characterized heparin binding peptide (HEPpep) and heparin coatings on cell surfaces and determined the specificity of these coatings in promoting assembly of MSC spheroids in dynamic culture. Overall, combining spheroids with both coatings promoted up to 70 ± 11% of spheroids to assemble into multiaggregate structures, as compared to only 10 ± 4% assembly when cells having the heparin coating were cultured with cells coated with a scrambled peptide. These results suggest that this self-assembly method represents an exciting approach that may be applicable for a wide range of applications in which cell aggregation is desired. [3] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.8793 mL | 4.3967 mL | 8.7934 mL | |
| 5 mM | 0.1759 mL | 0.8793 mL | 1.7587 mL | |
| 10 mM | 0.0879 mL | 0.4397 mL | 0.8793 mL |