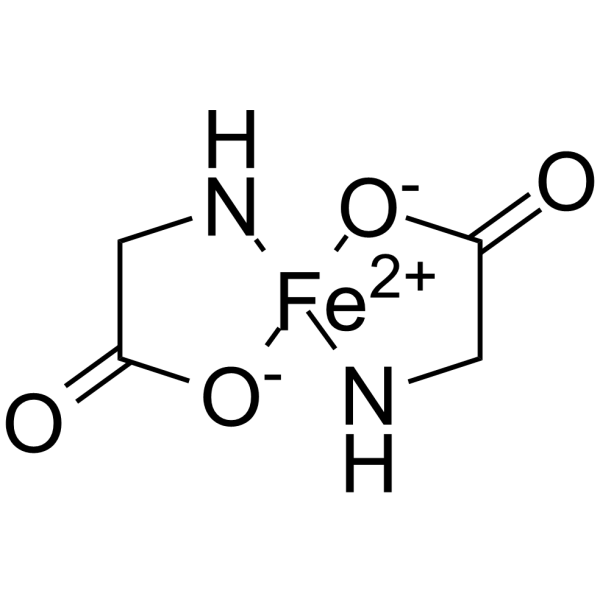
Physicochemical Properties
| Molecular Formula | C4H6FEN2O4 |
| Molecular Weight | 201.95 |
| Exact Mass | 203.983 |
| CAS # | 20150-34-9 |
| PubChem CID | 9837124 |
| Appearance | Light brown to green solid powder |
| Boiling Point | 240.9ºC at 760 mmHg |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 11 |
| Complexity | 37.4 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | GIPOFCXYHMWROH-UHFFFAOYSA-L |
| InChi Code | InChI=1S/2C2H5NO2.Fe/c2*3-1-2(4)5;/h2*1,3H2,(H,4,5);/q;;+2/p-2 |
| Chemical Name | 2-aminoacetate;iron(2+) |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Ferrous bisglycinate (25–200 μM; 2 h) has no effect on the viability of Caco-2 cells[2]. The Caco-2 cells' ferritin level is increased by ferrous bisglycinate (25 μM; 2 h)[2]. |
| ln Vivo | Mice treated with ferrous bisglycinate (500 mg/kg iron; po) do not develop colitis[3]. |
| Cell Assay |
Cell Viability Assay[2] Cell Types: Caco-2 cells Tested Concentrations: 0, 25, 50, 100, 200 μM Incubation Duration: 2 hrs (hours) Experimental Results: Did not affect the viability of wild-type and divalent metal transporter 1 (DMT1) knockout Caco-2 cells . Western Blot Analysis[2] Cell Types: Caco-2 cells Tested Concentrations: 25 μM Incubation Duration: 2 hrs (hours) Experimental Results: Increased ferritin content and diminished DMT1 expression levels Dramatically in the wild-type cells. |
| Animal Protocol |
Animal/Disease Models: Female C57BL/6 mice are induced colitis by dextran sodium sulfate (DSS)[3] Doses: 500 mg/kg iron Route of Administration: Po (add to the diet) for 10 days Experimental Results: Had the best survival rates (100%). Caused the least body lost (9% body loss). |
| References |
[1]. Treatment of mild non-chemotherapy-induced iron deficiency anemia in cancer patients: comparison between oral ferrous bisglycinate chelate and ferrous sulfate. Biomed Pharmacother. 2012 Sep; 66(6): 414-8. [2]. Iron Transport from Ferrous Bisglycinate and Ferrous Sulfate in DMT1-Knockout Human Intestinal Caco-2 Cells. Nutrients. 2019 Feb 26; 11(3): 485. [3]. Iron Supplements Modulate Colon Microbiota Composition and Potentiate the Protective Effects of Probiotics in Dextran Sodium Sulfate-induced Colitis. Inflamm Bowel Dis. 2017 May; 23(5): 753-766. |
| Additional Infomation |
Ferrous bisglycinate is a chelate that is used as a source of dietary iron. Forming a ring structure when reacting with glycine, ferrous bisglycinate acts as both a chelate and a nutritionally functional. It is found in foods for food enrichment or in supplements for the treatment of iron deficiency or iron deficiency anemia. See also: ... View More ... |
Solubility Data
| Solubility (In Vitro) | 0.1 M HCL: 2 mg/mL (9.90 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2 mg/mL (9.90 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.9517 mL | 24.7586 mL | 49.5172 mL | |
| 5 mM | 0.9903 mL | 4.9517 mL | 9.9034 mL | |
| 10 mM | 0.4952 mL | 2.4759 mL | 4.9517 mL |