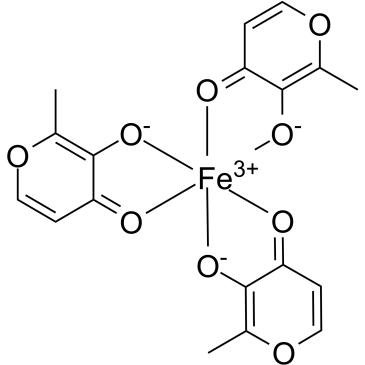
Ferric maltol (ST10; ST 10021; WHO 9974; ST-10; ST 10-021; trade names Accrufer and Feraccru) is an orally bioavailavle iron containing medication for the treatment of adults with low iron stores. It is used to provide supplemental iron to patients with an iron deficiency.
Physicochemical Properties
| Molecular Formula | C₁₈H₁₅FEO₉ |
| Molecular Weight | 431.15 |
| Exact Mass | 431.006 |
| Elemental Analysis | C, 50.14; H, 3.51; Fe, 12.95; O, 33.40 |
| CAS # | 33725-54-1 |
| PubChem CID | 169535 |
| Appearance | Typically exists as Purple to purplish red solids at room temperature |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 28 |
| Complexity | 200 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | Cl.CC1C=CC(C(C(N2CCCCC2)C)=O)=C(C)C=1 |
| InChi Key | AHPWLYJHTFAWKI-UHFFFAOYSA-K |
| InChi Code | InChI=1S/3C6H6O3.Fe/c3*1-4-6(8)5(7)2-3-9-4;/h3*2-3,8H,1H3;/q;;;+3/p-3 |
| Chemical Name | iron(3+);2-methyl-4-oxopyran-3-olate |
| Synonyms | Ferric maltol; Iron (III) maltol; ST10; ST 10021; WHO 9974ST-10; ST 10-021ST-10021; WHO-9974ST; 10 ST10-021; ST-10-021; ST10021; WHO9974; Ferric maltol; 33725-54-1; Iron (III) maltol; st10; iron maltol; MA10QYF1Z0; Ferric maltol [INN]; Ferric maltol [USAN]; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Metabolic Disease |
| ln Vitro | Serum ferritin, blood hemoglobin, reticulocyte hemoglobin, serum iron, and serum ferritin levels are all increased when iron maltol is taken [1]. |
| ln Vivo | According to research on animals, unabsorbed iron from iron maltol can at least stay in the small intestine in a chelated form, lowering the possibility of local toxicity and the risk of free iron damaging intestinal mucosa [1]. |
| Animal Protocol |
Introduction: Iron deficiency anemia affects up to three quarters of patients with inflammatory bowel disease (IBD). It can significantly impact the quality of life and the ability to work by impairing physical, emotional, and cognitive functioning. The etiology of iron deficiency anemia is multifactorial and oral or intravenous iron replacement is necessary. However, oral iron supplements are often discontinued prematurely due to poor tolerability or insufficient efficacy. Moreover, intravenous supplementation is inconvenient, associated with potentially serious safety risks, and a burden on healthcare resources.[1] Areas covered: Ferric maltol is a novel ferric iron compound with potential use as an oral therapy for iron deficiency anemia. This overview explains how the molecule’s design impacts clinical outcomes and summarizes available clinical data (ranging from early comparisons with ferrous sulfate to randomized, placebo-controlled, Phase III data in patients with IBD known to be intolerant of oral ferrous products).[1] Expert opinion: Ferric maltol offers the ability to treat iron deficiency anemia in mild-to-moderate IBD without resorting to intravenous therapy, even in those who are intolerant of oral ferrous products. This clinical benefit has the potential to change treatment pathways and increase choice, not only in IBD but also perhaps in many areas beyond gastroenterology. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Ferric maltol dissociates in the gastrointestinal tract, leading to a Tmax of 1.5-3.0 hours for iron concentrations. Mean serum iron increases by 14±6µmol/L in iron deficient patients following a single dose. a 60mg dose is approximately 14% bioavailable. 60 minutes after injection of radiolabelled ferric maltol, 11+2% of the dose is present in the bone marrow, 18±1% is present in the liver, and 2.6±1% is in the urine. Maltol has an AUC of 0.022-0.205h\*µg/mL and maltol glucuronide has an AUC of 9.83-30.9h\*µg/mL. 39.8-60% of an oral dose of ferric maltol is excreted in the urine as a glucuronide conjugate. Iron and ferric maltol are not excreted in the urine and unabsorbed ferric maltol is eliminated in the feces. Data regarding the volume of distribution of ferric maltol is not readily available. Data regarding the clearance of ferric maltol is not readily available. Metabolism / Metabolites _In vitro_, ferric maltol metabolism is predominantly glucuronidation of maltol by UGT1A6 and sulfation. Biological Half-Life Maltol has a half life of 0.7h. |
| Toxicity/Toxicokinetics |
Protein Binding Data regarding the protein binding of ferric maltol is not readily available. |
| References | [1]. Stallmach A, et al. Ferric maltol (ST10): a novel oral iron supplement for the treatment of iron deficiency anemia in inflammatory bowel disease. Expert Opin Pharmacother. 2015;16(18):2859-67. |
| Additional Infomation |
Ferric maltol is an iron(III) atom complexed with 3 maltol molecules to increase the bioavailability compared to iron(II), without depositing it in the duodenum as insoluble ferric hydroxide and phosphate. Ferric maltol has been described in literature since at least the late 1980s as a potential treatment for iron deficiency. Ferric maltol was granted FDA Approval on 25 July 2019. See also: Ferric cation (has active moiety). Drug Indication Ferric maltol is indicated to treat iron deficiency in adults. Feraccru is indicated in adults for the treatment of iron deficiency. Treatment of iron deficiency Mechanism of Action Ferric maltol dissociates as the iron atom is donated to unknown iron uptake mechanisms, possibly beta 3 integrin or divalent metal transporter 1, in the ileum and duodenum. Once the iron is in circulation, it then associates with transferrin and ferritin. Pharmacodynamics Ferric maltol is used to provide supplemental iron to patients with an iron deficiency. It has a wide therapeutic index as patients generally take 30mg twice daily, while concentrations of 20mg/kg may produce toxicity. Patients should be counselled regarding the risk of inflammatory bowel disease flares, iron overload, and accidental ingestion in children. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~12.5 mg/mL (~28.99 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1.25 mg/mL (2.90 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 1.25 mg/mL (2.90 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 12.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3194 mL | 11.5969 mL | 23.1938 mL | |
| 5 mM | 0.4639 mL | 2.3194 mL | 4.6388 mL | |
| 10 mM | 0.2319 mL | 1.1597 mL | 2.3194 mL |