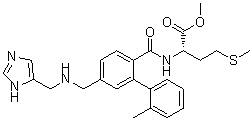
FTI-2153 is a potent and selective farnesyltransferase (FTase) inhibitor, with an IC50 of 1.4 nM and with >3000-fold more potent at blocking H-Ras (IC50, 10 nM) than Rap1A processing. FTI-2153 inhibits bipolar spindle formation during mitosis independently of transformation and Ras and p53 mutation status. FTI-2153 was reported to block bipolar spindle formation and chromosome alignment and causes prometaphase accumulation during mitosis of human lung cancer cells.
Physicochemical Properties
| Molecular Formula | C25H30N4O3S |
| Molecular Weight | 466.595704555511 |
| Exact Mass | 466.204 |
| CAS # | 344900-92-1 |
| Related CAS # | FTI-2153 TFA;2820151-01-5 |
| PubChem CID | 6918458 |
| Appearance | Off-white to light yellow solid powder |
| LogP | 4.481 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 12 |
| Heavy Atom Count | 33 |
| Complexity | 623 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | CC1=CC=CC=C1C2=C(C=CC(=C2)CNCC3=CN=CN3)C(=O)N[C@@H](CCSC)C(=O)OC |
| InChi Key | KZCMMZOLCBZLNG-QHCPKHFHSA-N |
| InChi Code | InChI=1S/C25H30N4O3S/c1-17-6-4-5-7-20(17)22-12-18(13-26-14-19-15-27-16-28-19)8-9-21(22)24(30)29-23(10-11-33-3)25(31)32-2/h4-9,12,15-16,23,26H,10-11,13-14H2,1-3H3,(H,27,28)(H,29,30)/t23-/m0/s1 |
| Chemical Name | L-Methionine,N-[[5-[[(1H-imidazol-4-ylmethyl)amino]methyl]-2'-methyl[1,1'-biphenyl]-2-yl]carbonyl]-, methyl ester |
| Synonyms | FTI2153 FTI 2153 FTI-2153 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Two human lung cancer cell lines that undergo mitosis are inhibited in the development of bipolar spindles by FTI-2153 [2]. In both transformed and untransformed cells, the fraction of pro-cells with dividing DNA shape is increased by FTI-2153 [2]. The proliferation of T-24 and Calu-1 cells was decreased by 38% and 36%, respectively, by FTI-2153 (15 μM). With an inhibition rate of 8%, the scaffolds of NIH3T3, HFF, and HT-1080 were less effective in inhibiting the growth of A-549 and OVCAR3 cells by 25% and 22%, respectively. Thus, although FTI-2153 decreased cell growth equally in T-24 and Calu-1 cells, it only inhibited Calu-8 and 13% of the cells in T-24. FTI-2153 inhibits the proliferation of both HFF and NIH3T3 cells, however only NIH3T3 cells are resistant to FTI-2153's ability to cause bipolar spindle formation [2]. 2] |
| Cell Assay |
Cell Viability Assay[2] Cell Types: NIH3T3, HFF, HT1080, T-24, OVCAR3, A-549 and Calu-1 cells. Tested Concentrations: 48 hrs (hours). Incubation Duration: 15 μM. Experimental Results: When A-549 cells were treated with FTI-2153 (15 μM, 48 hrs (hours)), the proportion of metaphase cells increased relative to other stages of mitosis. FTI-2153 accumulates cells with a rosette-like morphology during metaphase, in which chromosomes form rings around single-cell microtubules. In all cells, except T-24 and NIH3T3, FTI-2153 treatment increased the proportion of cells in metaphase mitosis and diminished the proportion of cells in telophase/mitosis. In HT1080 cells, the percentages of cells in prophase and telophase/cytokinesis were 5% and 85%, respectively, in control cells and 55% and 35%, respectively, in treated cells. Similar results were found in HFF cells. As mentioned previously, Calu-1 and A-549 cells had similarly large changes, whereas OVCAR3 had smaller changes. In contrast, FTI-2153 did not Dramatically affect the dist |
| References |
[1]. Antitumor efficacy of a novel class of non-thiol-containing peptidomimetic inhibitors of farnesyltransferase and geranylgeranyltransferase I: combination therapy with the cytotoxic agents cisplatin, Taxol, and gemcitabine. Cancer Res. 1999 Oct 1;59(19):4919-26. [2]. The farnesyltransferase inhibitor, FTI-2153, inhibits bipolar spindle formation during mitosis independently of transformation and Ras and p53 mutation status. Cell Death Differ. 2002 Jul;9(7):702-9. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~90 mg/mL (~192.88 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.36 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.36 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.36 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1432 mL | 10.7158 mL | 21.4316 mL | |
| 5 mM | 0.4286 mL | 2.1432 mL | 4.2863 mL | |
| 10 mM | 0.2143 mL | 1.0716 mL | 2.1432 mL |