Physicochemical Properties
| Molecular Formula | C13H18CLFN2O6S |
| Molecular Weight | 384.81 |
| Exact Mass | 384.0558133 |
| CAS # | 3033987-42-4 |
| PubChem CID | 171663452 |
| Appearance | Solid powder ; White to off-white |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 24 |
| Complexity | 499 |
| Defined Atom Stereocenter Count | 1 |
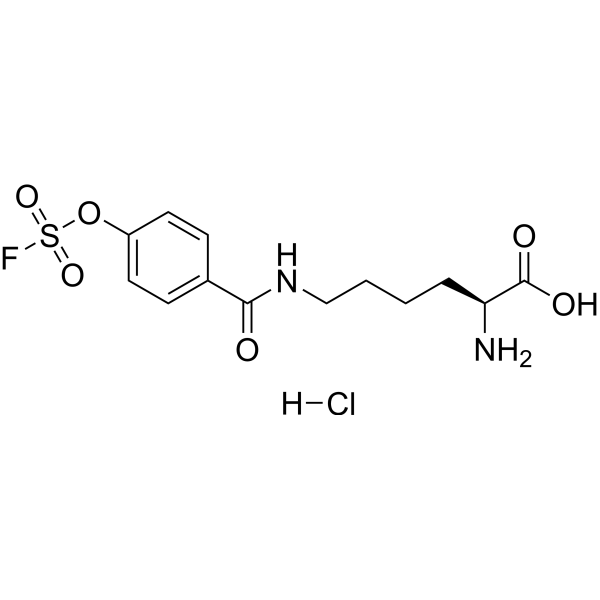
| SMILES | C1=CC(=CC=C1C(=O)NCCCC[C@@H](C(=O)O)N)OS(=O)(=O)F.Cl |
| InChi Key | WDAPBEYTBHLADN-MERQFXBCSA-N |
| InChi Code | InChI=1S/C13H17FN2O6S.ClH/c14-23(20,21)22-10-6-4-9(5-7-10)12(17)16-8-2-1-3-11(15)13(18)19;/h4-7,11H,1-3,8,15H2,(H,16,17)(H,18,19);1H/t11-;/m0./s1 |
| Chemical Name | (2S)-2-amino-6-[(4-fluorosulfonyloxybenzoyl)amino]hexanoic acid;hydrochloride |
| Synonyms | FSK (hydrochloride); FSK HCl;; 3033987-42-4 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
FSK is a latent bioreactive unnatural amino acid designed to form covalent bonds with nucleophilic amino acid residues (Lys, His, Tyr) in proximity via Sulfur(VI) Fluoride Exchange (SuFEx) chemistry. It does not target a specific protein but rather serves as a covalent bonding tool incorporated into proteins of interest. [1] |
| ln Vitro |
- FSK was incorporated into various proteins in E. coli, including ubiquitin (at position 6), eGST (at positions 65, 86, 97, 103), sfGFP, and nanobody 7D12 (at position 31). Incorporation was confirmed by fluorescence, Western blot, and mass spectrometry. High incorporation fidelity was demonstrated, with the intact mass of ubiquitin(6FSK) measured at 9590.1 Da, matching the theoretical value of 9589.9 Da. [1] - FSK enabled inter-protein cross-linking at distances unreachable with the previously developed FSY. In eGST homodimer, FSK incorporated at site 65 (with target residues Lys93, Tyr100, Lys132, Tyr135 at distances of 9.2-13.3 Å from the Cα) induced significant dimeric cross-linking, while FSY at the same site did not. In contrast, at site 103 with shorter distances (7.8 Å to His106, 6.0 Å to Lys107), FSY induced cross-linking while FSK did not. [1] - FSK was shown to react with Lys, His, and Tyr residues, confirmed by mutagenesis studies. In sGST, FSK at position 97 cross-linked with Lys44, as well as with His44, Tyr44 mutants, but not with Ala44, Ser44, or Thr44 mutants. This demonstrates its multi-targeting capability similar to FSY. [1] - FSK enabled intramolecular cross-linking in ubiquitin. Mass spectrometry of ubiquitin(18FSK) showed a major peak (75% intensity) with a mass loss of 20 Da, corresponding to cross-linking between FSK18 and Lys29, with HF loss. Tandem MS analysis confirmed the cross-linked peptide, showing FSK18 specifically reacted with Lys29. [1] - FSK incorporated into nanobody 7D12 at position 31 enabled covalent cross-linking with EGFR in vitro. SDS-PAGE and Western blot showed almost complete up-shift of the EGFR band after incubation with 7D12(31FSK), but not with 7D12(WT) or 7D12(109FSK). Tandem MS confirmed cross-linking between FSK31 of 7D12 and His359 of EGFR, as predicted from the crystal structure (distance 12.7 Å). [1] - FSK incorporated at position 62 of thioredoxin (Trx) enabled cross-linking with multiple potential substrate proteins in E. coli lysates, as visualized by Western blot. Subsequent MS analysis identified 12 substrate proteins cross-linked by FSK, with cross-linking occurring on Lys, His, or Tyr residues. [1] |
| ln Vivo |
- FSK was incorporated into various proteins in E. coli, including ubiquitin (at position 6), eGST (at positions 65, 86, 97, 103), sfGFP, and nanobody 7D12 (at position 31). Incorporation was confirmed by fluorescence, Western blot, and mass spectrometry. High incorporation fidelity was demonstrated, with the intact mass of ubiquitin(6FSK) measured at 9590.1 Da, matching the theoretical value of 9589.9 Da. [1] - FSK enabled inter-protein cross-linking at distances unreachable with the previously developed FSY. In eGST homodimer, FSK incorporated at site 65 (with target residues Lys93, Tyr100, Lys132, Tyr135 at distances of 9.2-13.3 Å from the Cα) induced significant dimeric cross-linking, while FSY at the same site did not. In contrast, at site 103 with shorter distances (7.8 Å to His106, 6.0 Å to Lys107), FSY induced cross-linking while FSK did not. [1] - FSK was shown to react with Lys, His, and Tyr residues, confirmed by mutagenesis studies. In sGST, FSK at position 97 cross-linked with Lys44, as well as with His44, Tyr44 mutants, but not with Ala44, Ser44, or Thr44 mutants. This demonstrates its multi-targeting capability similar to FSY. [1] - FSK enabled intramolecular cross-linking in ubiquitin. Mass spectrometry of ubiquitin(18FSK) showed a major peak (75% intensity) with a mass loss of 20 Da, corresponding to cross-linking between FSK18 and Lys29, with HF loss. Tandem MS analysis confirmed the cross-linked peptide, showing FSK18 specifically reacted with Lys29. [1] - FSK incorporated into nanobody 7D12 at position 31 enabled covalent cross-linking with EGFR in vitro. SDS-PAGE and Western blot showed almost complete up-shift of the EGFR band after incubation with 7D12(31FSK), but not with 7D12(WT) or 7D12(109FSK). Tandem MS confirmed cross-linking between FSK31 of 7D12 and His359 of EGFR, as predicted from the crystal structure (distance 12.7 Å). [1] - FSK incorporated at position 62 of thioredoxin (Trx) enabled cross-linking with multiple potential substrate proteins in E. coli lysates, as visualized by Western blot. Subsequent MS analysis identified 12 substrate proteins cross-linked by FSK, with cross-linking occurring on Lys, His, or Tyr residues. [1] |
| Enzyme Assay |
- No traditional enzyme inhibition assays were performed, as FSK is not an enzyme inhibitor but a tool for covalent bonding. The study focused on its reactivity with nucleophilic residues via SuFEx chemistry in the context of folded proteins. [1] |
| Cell Assay |
- HeLa Cell Incorporation Assay: HeLa-EGFP(182TAG) reporter cells were transfected with pNEU-FSKRS plasmid (expressing FSKRS and tRNA) using a transfection reagent. Cells were cultured in DMEM medium supplemented with 10% FBS, with or without 1 mM FSK. After 48 h, cells were imaged by confocal microscopy for EGFP fluorescence. Cell lysates were prepared and analyzed by Western blot using anti-EGFP antibody, with GAPDH as loading control. [1] - HEK293T Cross-linking Assay: HEK293T cells were co-transfected with pNEU-FSKRS and pcDNA3.1 plasmids encoding eGST(WT), eGST(86TAG), or eGST(86TAG/92A). Cells were grown in media containing 1 mM FSK. After 48 h, cells were lysed, and lysates were analyzed by Western blot using anti-His antibody to detect eGST and its cross-linked dimers. GAPDH was used as a reference. [1] - A431 Cell Surface Cross-linking Assay: A431 human epidermoid carcinoma cells were incubated with purified nanobody 7D12(WT) or 7D12(31FSK) (concentration not specified) for 1, 2, 4, or 8 h at 37°C. Cells were then washed, lysed, and analyzed by Western blot using anti-His antibody to detect nanobody cross-linked to EGFR. [1] - E. coli GECX Assay: E. coli cells expressing Trx with FSK incorporated at position 59 or 62 (with a C-terminal His6 tag) were grown, and cross-linking was allowed to occur spontaneously in live cells. Cells were lysed, and cross-linked complexes were pulled down using Ni-NTA resin. Pulled-down proteins were digested with trypsin and analyzed by tandem mass spectrometry to identify cross-linked peptides and substrate proteins. [1] |
| Toxicity/Toxicokinetics |
- No toxicity data (LD50, organ toxicity, protein binding, drug-drug interactions) were reported for FSK. However, the study notes that aryl fluorosulfate groups like that in FSK have "exceptional biocompatibility" and do not indiscriminately react with non-interacting proteins. [1] |
| References |
[1]. A Genetically Encoded Fluorosulfonyloxybenzoyl-l-lysine for Expansive Covalent Bonding of Proteins via SuFEx Chemistry. J Am Chem Soc. 2021 Jul 14;143(27):10341-10351. |
| Additional Infomation |
- FSK (fluorosulfonyloxybenzoyl-L-lysine) is a genetically encoded unnatural amino acid containing an aryl fluorosulfate group. It was designed to overcome the limitations of the previously developed FSY, which has a shorter and more rigid side chain. FSK features a longer and more flexible side chain (13.8 Å from Cα to F atom vs. 9.0 Å for FSY), allowing it to reach and react with nucleophilic residues at greater distances. [1] - FSK is incorporated into proteins in response to the TAG codon using an engineered orthogonal tRNA/synthetase pair (FSKRS), evolved from Methanomethylophilus alvus PyIRS. Incorporation is site-specific and requires supplementation of FSK in the growth media. [1] - The reactivity of FSK is based on SuFEx chemistry, where the aryl fluorosulfate group reacts with nucleophilic residues (Lys, His, Tyr) only when they are in close proximity, enabling proximity-dependent covalent bond formation both intra- and intermolecularly. [1] - FSK and FSY were shown to be complementary tools. FSK is effective for longer-distance cross-linking, while FSY is better suited for shorter distances. Together, they expand the range of targetable sites on proteins, which is valuable for engineering covalent protein drugs (e.g., nanobodies) to potentially overcome mutational drug resistance. [1] - FSK (and FSY) enable Genetically Encoded Chemical Cross-linking (GECX) in live cells, allowing capture and identification of weak/transient protein-protein interactions by cross-linking to residues other than Cys (Lys, His, Tyr). This significantly expands the scope of GECX compared to previous systems that targeted only Cys. [1] |
Solubility Data
| Solubility (In Vitro) |
H2O : ~62.5 mg/mL (~162.42 mM; with ultrasonication) DMSO: ~200 mg/mL (519.7 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5987 mL | 12.9934 mL | 25.9869 mL | |
| 5 mM | 0.5197 mL | 2.5987 mL | 5.1974 mL | |
| 10 mM | 0.2599 mL | 1.2993 mL | 2.5987 mL |