Physicochemical Properties
| Molecular Formula | C27H41NO9 |
| Molecular Weight | 523.623 |
| Exact Mass | 523.278 |
| Elemental Analysis | C, 61.93; H, 7.89; N, 2.68; O, 27.50 |
| CAS # | 146478-73-1 |
| PubChem CID | 10673616 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.25g/cm3 |
| Boiling Point | 735.6ºC at 760mmHg |
| Flash Point | 398.7ºC |
| Vapour Pressure | 5.61E-25mmHg at 25°C |
| Index of Refraction | 1.564 |
| LogP | 2.123 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 37 |
| Complexity | 933 |
| Defined Atom Stereocenter Count | 10 |
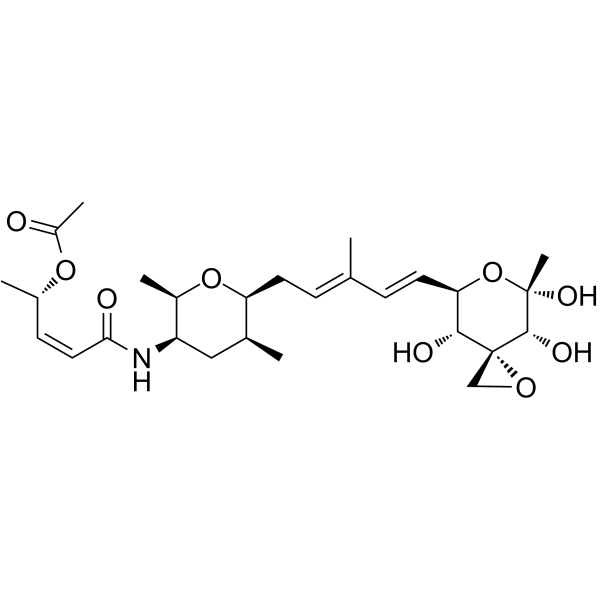
| SMILES | O1C([H])([H])[C@]21[C@@]([H])([C@@]([H])(/C(/[H])=C(\[H])/C(/C([H])([H])[H])=C(\[H])/C([H])([H])[C@@]1([H])[C@@]([H])(C([H])([H])[H])C([H])([H])[C@]([H])([C@@]([H])(C([H])([H])[H])O1)N([H])C(/C(/[H])=C(/[H])\[C@]([H])(C([H])([H])[H])OC(C([H])([H])[H])=O)=O)O[C@](C([H])([H])[H])([C@]2([H])O[H])O[H])O[H] |
| InChi Key | DQTXAXNYLWRTPB-NTVXLVODSA-N |
| InChi Code | InChI=1S/C27H41NO9/c1-15(8-11-22-24(31)27(14-34-27)25(32)26(6,33)37-22)7-10-21-16(2)13-20(18(4)36-21)28-23(30)12-9-17(3)35-19(5)29/h7-9,11-12,16-18,20-22,24-25,31-33H,10,13-14H2,1-6H3,(H,28,30)/b11-8+,12-9-,15-7+/t16-,17-,18+,20+,21-,22+,24+,25-,26-,27+/m0/s1 |
| Chemical Name | [(Z,2S)-5-[[(2R,3R,5S,6S)-2,5-dimethyl-6-[(2E,4E)-3-methyl-5-[(3R,4R,5S,7R,8R)-4,5,8-trihydroxy-5-methyl-1,6-dioxaspiro[2.5]octan-7-yl]penta-2,4-dienyl]oxan-3-yl]amino]-5-oxopent-3-en-2-yl] acetate |
| Synonyms | FR 901465; FR 901465; FR901465; 146478-73-1; (2S,3Z)-5-{[(2R,3R,5S,6S)-2,5-dimethyl-6-{(2E,4E)-3-methyl-5-[(3R,4R,5R,7S,8R)-4,7,8-trihydroxy-7-methyl-1,6-dioxaspiro[2.5]oct-5-yl]penta-2,4-dien-1-yl}tetrahydro-2H-pyran-3-yl]amino}-5-oxopent-3-en-2-yl acetate; [(Z,2S)-5-[[(2R,3R,5S,6S)-2,5-dimethyl-6-[(2E,4E)-3-methyl-5-[(3R,4R,5S,7R,8R)-4,5,8-trihydroxy-5-methyl-1,6-dioxaspiro[2.5]octan-7-yl]penta-2,4-dienyl]oxan-3-yl]amino]-5-oxopent-3-en-2-yl] acetate; [(Z,2S)-4-[[(2R,3R,5S,6S)-2,5-dimethyl-6-[(2E,4E)-3-methyl-5-[(3R,4R,5R,7S,8R)-4,7,8-trihydroxy-7-methyl-1,6-dioxaspiro[2.5]octan-5-yl]penta-2,4-dienyl]oxan-3-yl]carbamoyl]but-3-en-2-yl] acetate; 2-Pentenamide, 4-(acetyloxy)-N-[(2R,3R,5S,6S)-tetrahydro-2,5-dimethyl-6-[(2E,4E)-3-methyl-5-[(3R,4R,5R,7S,8R)-4,7,8-trihydroxy-7-methyl-1,6-dioxaspiro[2.5]oct-5-yl]-2,4-pentadien-1-yl]-2H-pyran-3-yl]-, (2Z,4S)-; CHEBI:65916; DTXSID501098324; FR 901465 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural antitumor agent; Spliceosome; promoter of SV40 DNA virus |
| ln Vitro | New antitumor substances, FR901463, FR901464 and FR901465 were isolated from the culture broth of a bacterium of Pseudomonas sp. No.2663. FR901463, FR901464 and FR901465 remarkably enhanced the transcriptional activity of the promoter of SV40 DNA virus. Further, these compounds exhibited potent antitumor activities against murine and human tumor cell lines in vitro.[3] |
| ln Vivo |
Mice with P388 leukemia and mouse ascites tumors live longer when treated with FR901465 (which has a T/C value of 127% in these mice) [1]. FR901463, FR901464 and FR901465, novel antitumor substances, were isolated from the fermentation broth of Pseudomonas sp. No. 2663. Their antitumor activities were examined in three mouse tumor systems and one human tumor system. The three FR compounds prolonged the life of mice bearing murine ascitic tumor P388 leukemia (T/C values were 160%, 145% and 127% for FR901463, FR901464 and FR901465, respectively), and inhibited the growth of a human solid tumor, A549 lung adenocarcinoma, with different effective dose ranges. [1] |
| Animal Protocol | FR901463, FR901464 and FR901465, novel antitumor substances, were isolated from the fermentation broth of Pseudomonas sp. No. 2663. Their antitumor activities were examined in three mouse tumor systems and one human tumor system. The three FR compounds prolonged the life of mice bearing murine ascitic tumor P388 leukemia (T/C values were 160%, 145% and 127% for FR901463, FR901464 and FR901465, respectively), and inhibited the growth of a human solid tumor, A549 lung adenocarcinoma, with different effective dose ranges. FR901464 exhibited most prominent effects on these tumor systems among the three FR compounds. FR901464 also inhibited the growth of murine solid tumors, Colon 38 carcinoma and Meth A fibrosarcoma. To address the involvement of transcriptional activation ability of the three FR compounds in the antitumor effect, we selected FR901464 as a candidate compound and investigated cell cycle transition, chromatin status and endogenous gene expression in FR901464-treated tumor cells having elevated transcriptional activity. FR901464 induced characteristic G1 and G2/M phase arrest in the cell cycle and internucleosomal degradation of genomic DNA with the same kinetics as activation of SV40 promoter-dependent cellular transcription in M-8 tumor cells. In contrast to the potent activation of the viral promoter, FR901464 suppressed the transcription of some inducible endogenous genes but not house keeping genes in M-8 cells. These results suggest that FR901464 may induce a dynamic change of chromatin structure, giving rise to strong antitumor activity, and therefore may represent a new type of drug for cancer chemotherapy.[1] |
| References |
[1]. New antitumor substances, FR901463, FR901464 and FR901465. II. Activities against experimental tumors in mice and mechanism of action. J Antibiot (Tokyo). 1996 Dec;49(12):1204-11. [2]. New antitumor substances, FR901463, FR901464 and FR901465. III. Structures of FR901463, FR901464 and FR901465. J Antibiot (Tokyo). 1997 Jan;50(1):96-9. [3]. New antitumor substances, FR901463, FR901464 and FR901465. I. Taxonomy, fermentation, isolation, physico-chemical properties and biological activities. J Antibiot (Tokyo). 1996 Dec;49(12):1196-203. [4]. Discovery and Development of Natural Products for Pest Management. RETURN TO BOOKPREVCHAPTERNEXT. |
| Additional Infomation | FR901465 is a spiro-epoxide with potent cytotoxic activity against human tumour cells. It is isolated from Pseudomonas sp. no.2663. It has a role as an antineoplastic agent and a bacterial metabolite. It is an acetate ester, a cyclic hemiketal, a member of pyrans, a secondary alcohol, a monocarboxylic acid amide and a spiro-epoxide. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9098 mL | 9.5489 mL | 19.0978 mL | |
| 5 mM | 0.3820 mL | 1.9098 mL | 3.8196 mL | |
| 10 mM | 0.1910 mL | 0.9549 mL | 1.9098 mL |