Physicochemical Properties
| Molecular Formula | C11H18N4O7S |
| Molecular Weight | 350.35 |
| Exact Mass | 350.089 |
| CAS # | 1452458-70-6 |
| PubChem CID | 73386853 |
| Appearance | Typically exists as solid at room temperature |
| LogP | -3.9 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 23 |
| Complexity | 592 |
| Defined Atom Stereocenter Count | 3 |
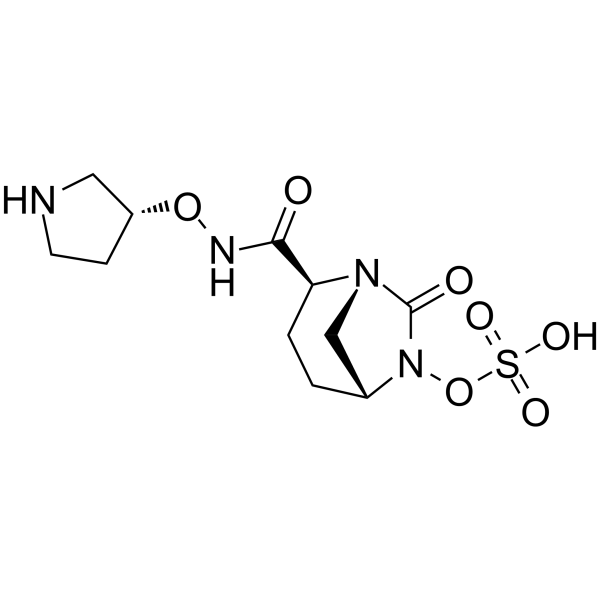
| SMILES | C1C[C@H](N2C[C@@H]1N(C2=O)OS(=O)(=O)O)C(=O)NO[C@@H]3CCNC3 |
| InChi Key | JBMFHPNYQVHTCK-HLTSFMKQSA-N |
| InChi Code | InChI=1S/C11H18N4O7S/c16-10(13-21-8-3-4-12-5-8)9-2-1-7-6-14(9)11(17)15(7)22-23(18,19)20/h7-9,12H,1-6H2,(H,13,16)(H,18,19,20)/t7-,8-,9+/m1/s1 |
| Chemical Name | [(2S,5R)-7-oxo-2-[[(3R)-pyrrolidin-3-yl]oxycarbamoyl]-1,6-diazabicyclo[3.2.1]octan-6-yl] hydrogen sulfate |
| Synonyms | FPI-1465; 1452458-70-6; SCHEMBL15207353; JBMFHPNYQVHTCK-HLTSFMKQSA-N; HY-139744; CS-0255054; (2S,5R)-7-oxo-N-[(3R)-pyrrolidin-3-yloxy]-6-(sulfooxy)-1,6-diazabicyclo[3.2.1]octane-2-carboxamide; [(2S,5R)-7-oxo-2-[[(3R)-pyrrolidin-3-yl]oxycarbamoyl]-1,6-diazabicyclo[3.2.1]octan-6-yl] hydrogen sulfate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Serine-β-Lactamases; Penicillin-binding proteins (PBPs) |
| ln Vitro |
For OXA-48, all FPI derivatives displayed roughly an order of magnitude slower on rates as well as faster off rates, leading to significantly increased Kd values (Table 1). In particular, the FPI-1465-OXA-48 carbamyl enzyme displayed a very short half-life (off rate t1/2 for avibactam vs FPI-1465: 1500 ± 200 min vs 22 ± 1 min). In the OXA-48 bound FPI cocrystal structures, we observe numerous direct and water mediated hydrogen bonds between the various C2 functional groups and conserved active site residues (Figure S2D and E). The observed higher Kd values may be due in part to an entropic cost associated with ordering the C2 side chain of the derivatives via these hydrogen bonds. Taken together, the above-mentioned structural and kinetic results demonstrate that C2 avibactam derivatives retain β-lactamase inhibitory properties. [1] Previously, FPI-1465 displayed remarkable synergy in combination with β-lactams against MBL-expressing strains. We used FPI-1465 as a model DBO compound and assessed its ability to inhibit a panel of SBLs and MBLs in vitro (Figure S3). FPI-1465 displayed no inhibitory activity against MBLs, implying a different mechanism for synergy. We next tested the minimum inhibitory concentration (MIC) of avibactam, FPI-1465, FPI-1523, and FPI-1602 alone against E. coli BW25113 pGDP-2 transformants either with an empty vector or expressing different β-lactamases. All compounds displayed consistently low MICs irrespective of β-lactamase expression (Table S3). FPI-1602 demonstrated a remarkably low MIC value of <0.5 μg/mL and was therefore selected for further MIC experiments against a panel of NDM-1 positive clinical isolates. FPI-1602 displayed marked antimicrobial activity against P. aeruginosa, E. coli, and Enterobacter spp. (Table S4). The activity against P. aeruginosa is particularly interesting as a recent publication has described a similar compound (OP0595) that does not exhibit antimicrobial activity against P. aeruginosa (MIC > 32 μg/mL). These data clearly demonstrate that avibactam derivatives act as direct antimicrobial agents and that their activity is not thwarted by β-lactamase expression. [1] In competition experiments in which BOCILLIN FL and the FPIs were added to E. coli PBP1b at the same time to start the reaction, no inhibition was observed up to 2 mM of the FPIs (Table S5). However, an extensive 48 h preincubation of FPI-1602 with the purified E. coli PBP1b membrane protein prior to addition of BOCILLIN FL resulted in a concentration-dependent inhibition of BOCILLIN FL binding (Figure S6), indicating that at high concentrations the derivatives can act as slow binding inhibitors of PBP1b. Therefore, due to difficulties in crystallizing E. coli PBP2, we reasoned that cocrystallization of E. coli PBP1b with the avibactam derivatives would lend insight into the structural basis of PBP inhibition by these compounds. The cocrystal structure of the single pass bitopic membrane protein E. coli PBP1b covalently inhibited by FPI-1465 was solved to 2.85 Å resolution in spacegroup P22121 with a single protein monomer in the ASU (for ligand electron density maps and crystallographic data statistics, see Figure S7 and Table S6). The overall orientation of FPI-1465 in the PBP1b active site is directly analogous to the carbamyl-SBL bound complexes (Figures 1b,c and 2d). The FPI-1465 N6 sulfate projects toward the conserved motif iii and makes hydrogen bonding contacts with the O-γ of T699 and T701. Also, the FPI-1465 C7 carbonyl oxygen occupies the canonical oxyanion hole constituted by the backbone amide nitrogens of T701 and S510. Finally, the C2 side chain projects away from the catalytic core (Figure 2d). This structure represents the first glimpse into DBO-mediated PBP inhibition and demonstrates the ability of avibactam derivatives to interact directly with the conserved active site motifs of PBPs, providing a molecular basis for structure-based drug design efforts. [1] In the FPI-1465-E. coli PBP1b crystal structure, the N6 nitrogen is 4.4 Å from the S572 O-γ (Figure 2d). In contrast, in the FPI-1465 bound CTX-M-15 and OXA-48 structures, the N6 nitrogen is much closer to the equivalent motif II serine O-γ (3.3 Å and 3.0 Å, Figure 1b and c). The motif II serine has an essential role as the general acid for protonation of the β-lactam or DBO nitrogen leaving group upon formation of the acyl-enzyme covalent intermediate. Therefore, we propose that the relatively poor inhibition of E. coli PBP1b by the DBOs is likely due to the suboptimal positioning of the general acid required for acylation. |
| Enzyme Assay |
Enzyme Assays. [1] For all enzyme assays the buffer consisted of 50 mM HEPES pH 7.5 and Tween20 0.01%. OXA-48 experiments were performed with the addition of 50 mM NaHCO3. Enzyme dilutions were made in BSA to 100 ng/µL. Acylation and deacylation experiments were performed as described previously. 2, 11.For all compounds described, onrates were determined using a continuous assay with nitrocefin as reporter substrate. For CTX-M-15, 100 µL enzyme (0.2 nM [final]) was added to 100 µL nitrocefin (50 µM [final]; Km = 10 µM) and inhibitor. The maximum concentration of inhibitor used for CTX-M-15 on-rates was: avibactam, 0.8 µM; FPI-1465, 9 µM; FPI-1523, 4 µM; FPI1602, 9 µM. The same methods were applied for OXA-48 (0.03 nM [final]) with nitrocefin (100 µM [final]; Km = 50 µM). For OXA-48, the maximum concentration of inhibitor used was: avibactam, 50 µM; FPI-1465, 100 µM; FPI-1523, 100 µM; FPI-1602, 100 µM. For CTX-M-15, off-rates were determined continuously using the jump dilution method12 where 1 µM enzyme was incubated with 10 µM inhibitor at 37°C for 30 minutes and then diluted 1/400 before adding 20 µL to 180 µL of nitrocefin (400 µM) in assay buffer. For OXA-48 7 µM enzyme was incubated with 10 µM inhibitor and incubated for 1 hour before 1/16000 dilution and addition to substrate (200 µM) as above. For OXA-48, discontinuous sampling was applied for all inhibitors other than FPI-1465. For concentration-response experiments assay buffer was used as above. All enzymes (1 nM) were incubated with inhibitor for 30 minutes at 37°C before dilution in nitrocefin (20 µM). Metalloenzymes were supplemented with 10 µM ZnSO4. The maximum concentration of avibactam used was 20 µM. Antimicrobial Susceptibility Testing. [1] MIC testing was done according to the Clinical Laboratory Standards Institute. 13 All experiments were performed in duplicate and strains were grown at 37°C for 18 hours. pGDP constructs were made with the noted gene under control of a bla promoter for high-level constitutive expression. PBP Binding Assays. [1] For bacterial membrane preparation, an overnight culture of E. coli K-12 (MG1665) in BHI broth was diluted in a fresh medium and was further incubated at 37°C under agitation to reach an OD600 of ~0.6-0.7. The cells were harvested by centrifugation at 3,000 g for 15 min at 4°C, washed and suspended in KPN (20 mM potassium phosphate - 140 mM NaCl, pH 7.5). Cells were first treated with lysozyme (500 µg/mL) for 1h at 37°C, before addition of a protease inhibitor cocktail, deoxyribonuclease (6 µg/mL) and ribonuclease (6 µg/mL). After 30 minutes of treatment, cells were disrupted by a French press and the bacterial lysate was centrifuged at 12,000 g for 10 min to remove unbroken cells. The supernatant was then centrifuged at 150,000 g for 40 min at 4°C using a fixed-angle rotor to collect the membranes. The membranes were suspended in a minimal volume of KPN buffer and stored at -86°C. Protein concentration was estimated by the method of Bradford with the BCA kit (Pierce) using bovine serum albumin as a standard. The relative binding affinity of test molecules for bacterial PBPs were assayed in a competition assay with the fluorescent penicillin BOCILLIN FL as the reporter molecule. Increasing concentrations of the test compounds were added to aliquots of the reaction mixture containing 30 μg of bacterial membrane preparation for 10 min at 37°C prior to the addition of BOCILLIN FL (100 µM) for an additional 20 min. Membranecontaining samples were then heated to 95°C for 3 min in electrophoretic loading buffer containing SDS before electrophoresis and separation of proteins on a SDS-polyacrylamide discontinuous gel system (5% stacking and 10% separating gels). After electrophoresis, the gels were quickly rinsed in water and incubated for 30 min in a fixing solution (50% methanol - 7% acetic acid). Gels were scanned with a Molecular Imager FX Pro instrument using the excitation and emission wavelengths of 488 nm and 530 nm, respectively, to collect the image of the PBP profile. The concentration of the test compound needed to block 50% of the subsequent binding of BOCILLIN FL to each PBP represented the IC50 value. BOCILLIN FL Competition Assays using Purified E. coli PBPs. [1] To assess the relative inhibition of E. coli PBPs by the avibactam derivatives, SDS-PAGE based concentration response experiments were performed in triplicate using BOCILLIN FL as a reporter molecule. All reagents were diluted in assay buffer prior to use. To start the reaction, various concentrations of unlabeled compound and 27.8 μM BOCILLIN FL were simultaneously added to 4.7μM of purified PBP in a final reaction volume of 36 μL. The reaction was incubated at 25°C for 20 min prior to addition of 10X SDS-PAGE loading dye. In contrast, for pre-incubation experiments various amounts of inhibitor compound was pre-incubated with 4.7μM E. coli PBP1b for 48 hours prior to an additional 20 min incubation in the presence of 27.8 S5 μM BOCILLIN FL (Figure S6). The samples were then boiled for 2 min prior to loading 10μL on a 12% SDS-PAGE precast gel. Following electrophoresis, gels were imaged under UV light using a Syngene ChemiGenius2 bio imaging System. Densitometry analysis was performed using ImageJ as previously described. 14 The individual data points were normalized to the maximum value of the fluorescence intensity, which represents total saturation of protein by BOCILLIN FL in the absence of unlabeled compound. Benzyl penicillin, and Kanamycin were used as positive and negative controls, respectively. The IC50 values are defined as the compound concentration required to reduce the residual binding of BOCILLIN FL by 50% and were calculated using SigmaPlot. |
| Cell Assay |
Microscopy. [1] Cells were cultured in a standard MIC curve, then fixed and imaged according to the methods of Czarny et al. 15 In brief, after culture densities were recorded using a spectrophotometer, cultures were diluted 1:10 in 2% glutaraldehyde buffered with 25 mM HEPES (pH 6.8) for one hour. Then, 15 uL of this solution was transferred to a 0.17 mm glass-bottom 384-well microplate, along with 5 uL of 1.5% filter-sterilized nigrosin stain. Plates were gently flushed with nitrogen gas, then heat-fixed at 50°C in a humidity-controlled incubator. Finally, plates were imaged under brightfield using a Nikon Eclipse Ti-E inverted microscope. Cell features were quantified with ImageJ14, using the analysis pipeline in Czarny et al. 15 These image features were used to cluster drug treatments using Ward's least variance, as well as compute a correlation map and Pearson correlation values for treatments. |
| References |
[1]. Structural and Kinetic Characterization of Diazabicyclooctanes as Dual Inhibitors of Both Serine-β-Lactamases and Penicillin-Binding Proteins. ACS Chem Biol. 2016 Apr 15;11(4):864-8. |
| Additional Infomation | Avibactam is a diazabicyclooctane β-lactamase inhibitor possessing outstanding but incomplete efficacy against multidrug-resistant Gram-negative pathogens in combination with β-lactam antibiotics. Significant pharmaceutical investment in generating derivatives of avibactam warrants a thorough characterization of their activity. We show here through structural and kinetic analysis that select diazabicyclooctane derivatives display effective but varied inhibition of two clinically important β-lactamases (CTX-M-15 and OXA-48). Furthermore, these derivatives exhibit considerable antimicrobial activity (MIC ≤ 2 μg/mL) against clinical isolates of Pseudomonas aeruginosa, Escherichia coli, and Enterobacter spp. Imaging of cell phenotype along with structural and biochemical experiments unambiguously demonstrate that this activity, in E. coli, is a result of targeting penicillin-binding protein 2. Our results suggest that structure-activity relationship studies for the purpose of drug discovery must consider both β-lactamases and penicillin-binding proteins as targets. We believe that this approach will yield next-generation combination or monotherapies with an expanded spectrum of activity against currently untreatable Gram-negative pathogens.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8543 mL | 14.2714 mL | 28.5429 mL | |
| 5 mM | 0.5709 mL | 2.8543 mL | 5.7086 mL | |
| 10 mM | 0.2854 mL | 1.4271 mL | 2.8543 mL |