Physicochemical Properties
| Molecular Formula | C51H65N3O8S |
| Molecular Weight | 880.142113447189 |
| Exact Mass | 879.449 |
| CAS # | 2374144-21-3 |
| PubChem CID | 155530360 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 8.9 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 12 |
| Heavy Atom Count | 63 |
| Complexity | 1630 |
| Defined Atom Stereocenter Count | 10 |
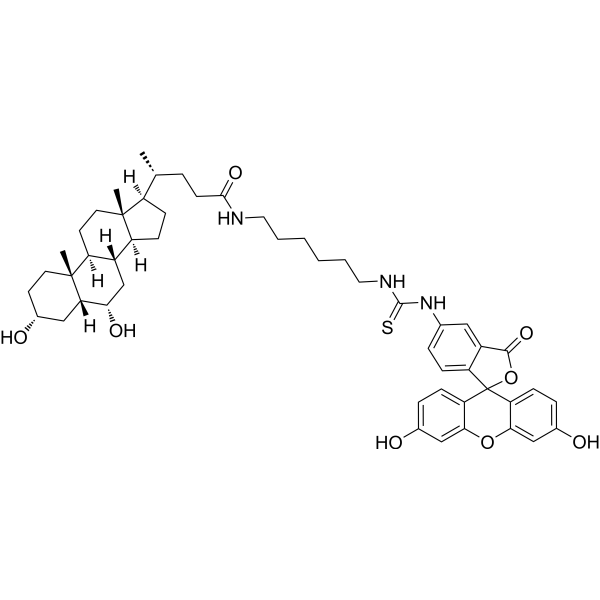
| SMILES | C[C@H](CCC(=O)NCCCCCCNC(=S)NC1=CC2=C(C=C1)C3(C4=C(C=C(C=C4)O)OC5=C3C=CC(=C5)O)OC2=O)[C@H]6CC[C@@H]7[C@@]6(CC[C@H]8[C@H]7C[C@@H]([C@H]9[C@@]8(CC[C@H](C9)O)C)O)C |
| InChi Key | MFJKSCWIKMCTMG-RGOSNDNESA-N |
| InChi Code | InChI=1S/C51H65N3O8S/c1-29(36-15-16-37-34-28-43(58)42-25-33(57)18-20-50(42,3)38(34)19-21-49(36,37)2)8-17-46(59)52-22-6-4-5-7-23-53-48(63)54-30-9-12-39-35(24-30)47(60)62-51(39)40-13-10-31(55)26-44(40)61-45-27-32(56)11-14-41(45)51/h9-14,24,26-27,29,33-34,36-38,42-43,55-58H,4-8,15-23,25,28H2,1-3H3,(H,52,59)(H2,53,54,63)/t29-,33-,34+,36-,37+,38+,42+,43+,49-,50-/m1/s1 |
| Chemical Name | (4R)-4-[(3R,5R,6S,8S,9S,10R,13R,14S,17R)-3,6-dihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl]-N-[6-[(3',6'-dihydroxy-3-oxospiro[2-benzofuran-1,9'-xanthene]-5-yl)carbamothioylamino]hexyl]pentanamide |
| Synonyms | FITC-hyodeoxycholic acid; Rimteravimab; 2374144-21-3; CHEMBL4464514; 2540797-21-3; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | FITC-labeled HDCA |
| ln Vitro | In order to confirm the binding of 10rr to LXRβ, we conducted a fluorescence polarization assay, which demonstrated that 10rr dose-dependently inhibited the binding of FITC-hyodeoxycholic acid to LXRβ with a Ki value of 2.3 μM (Fig. 5C), suggesting that 10rr binds to LXRβ directly [1]. |
| References |
[1]. Discovery of novel liver X receptor inverse agonists as lipogenesis inhibitors. Eur J Med Chem. 2020 Nov 15;206:112793. |
| Additional Infomation |
Rimteravimab is an immunoglobulin G1 (IgG1) monoclonal antibody directed against the spike (S) protein of severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), that can potentially be used for immunization against the Coronavirus disease 2019 (COVID-19). Upon administration, rimteravimab specifically targets and binds to the receptor-binding domain (RBD) of the S protein, thereby blocking viral attachment and entry into human cells and may thereby neutralize SARS-CoV-2. This may slow the progression of the disease and accelerate recovery, and may potentially provide temporary protection against infection with SARS-CoV-2. Based on the co-crystal structures of LXRβ and its agonists (spiro [pyrrolidine-3,3'-oxindole] derivatives) discovered by us previously, we designed and synthesized a compound library to explore the agonistic activities. The library was screened with luciferase reporter assays, interestingly, it resulted in the discovery of 10 LXR inverse agonists besides 5 LXR agonists. To clarify the mechanism of the actions, we conducted molecular dynamics (MD) simulations on the LXR and inverse agonists complexes, and revealed that H3, H11 and H12 configurations are the key to turn on agonism or inverse agonism status for LXR. Binding tightly with H3, pushing H11 out and destabilizing H12 could form a bigger hydrophobic groove to accommodate NCOR1 to turn on LXR inverse agonism. The inverse agonist 10rr was further studied, and found as a lipogenesis inhibitor through down-regulating LXR target genes SREBP-1c, ACC, FAS and SCD-1, and demonstrated lipid-lowering effects in 3T3-L1 cells, HepG2 cells and mice with Triton WR-1339-induced hyperlipidemia. Therefore, we have proved that LXR inverse agonists can be promising agents for hyperlipidemia treatment.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.1362 mL | 5.6809 mL | 11.3618 mL | |
| 5 mM | 0.2272 mL | 1.1362 mL | 2.2724 mL | |
| 10 mM | 0.1136 mL | 0.5681 mL | 1.1362 mL |