Physicochemical Properties
| Molecular Formula | C59H56N4O9S |
| Molecular Weight | 997.162354469299 |
| Exact Mass | 996.376 |
| CAS # | 2374144-23-5 |
| PubChem CID | 168006992 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 7.4 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 21 |
| Heavy Atom Count | 73 |
| Complexity | 1740 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | S=C(NCCCCCC(NC1=CC=CC(=C1)CN(CCCOC1C=CC=C(CC(=O)O)C=1)CC(C1C=CC=CC=1)C1C=CC=CC=1)=O)NC1C=CC2=C(C=1)C(=O)OC12C2C=CC(=CC=2OC2C=C(C=CC1=2)O)O |
| InChi Key | NSDMNSCDJXAQNR-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C59H56N4O9S/c64-45-23-26-51-53(35-45)71-54-36-46(65)24-27-52(54)59(51)50-25-22-44(34-48(50)57(69)72-59)62-58(73)60-28-9-3-8-21-55(66)61-43-19-10-14-40(31-43)37-63(29-12-30-70-47-20-11-13-39(32-47)33-56(67)68)38-49(41-15-4-1-5-16-41)42-17-6-2-7-18-42/h1-2,4-7,10-11,13-20,22-27,31-32,34-36,49,64-65H,3,8-9,12,21,28-30,33,37-38H2,(H,61,66)(H,67,68)(H2,60,62,73) |
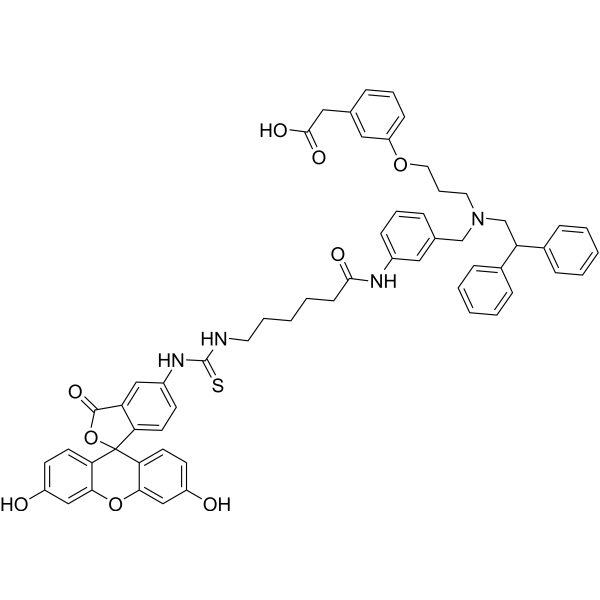
| Chemical Name | 2-[3-[3-[[3-[6-[(3',6'-dihydroxy-3-oxospiro[2-benzofuran-1,9'-xanthene]-5-yl)carbamothioylamino]hexanoylamino]phenyl]methyl-(2,2-diphenylethyl)amino]propoxy]phenyl]acetic acid |
| Synonyms | FITC-GW3965; 2374144-23-5; FITC-GW-3965 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | LXRβ/liver X receptor beta |
| ln Vitro | The liver X receptors (LXRs) of the nuclear receptor family are promising therapeutic targets of multiple diseases like lipid disorders, chronic inflammation, as well as different human cancers. To date, no LXR agonists or antagonists can be used in clinics, emphasizing the importance for discovering new LXR modulators. Fragment-based lead discovery (FBLD) is powerful for designing new scaffolds and new mechanistic drugs, but fragment screening has not been applied to LXRs yet, which might be due to the lack of a specific fragment screening method against the dynamic and hydrophobic ligand binding domain (LBD) of LXRs. Herein, a series of fluorescent tracers were designed, synthesized and tested. The tracer based on hyodeoxycholic acid exhibited a good capability for competitively detecting the ligand binding of LXRβ using a fluorescence polarization approach. Then, 1074 fragments were screened against the LBD of LXRβ (LXRβ-LBD), resulting in 27 binding hits. These fragment hits were further tested using the co-activator recruitment assay and reporter gene assay, and efforts in X-ray crystallography fortunately solved a co-crystal structure of LXRβ-LBD with the fragment F3 (tert-butyl-7-amino-3,4-dihydroisoquinoline-2(1H)-carboxylate). The fluorescence-based fragment screening tool and the newly identified LXRβ binding fragments provide the basis for developing novel LXRβ modulators [1]. |
| Enzyme Assay |
FP binding assay [1] All FP experiments were conducted with a Victor X5 microplate reader using black NBS polystyrene 384-well microplates. For the saturation experiment, fluorescent probe (10 nM) was diluted using the FP buffer (50 mM Tris, pH 8.0, 400 mM NaCl and 5 mM 2-mercaptoethanol), and LXRβ (0–20 μM) were added at different concentrations. For each LXRβ concentration, triplicated wells were used. The final concentration of DMSO in the reactions was fixed at 0.5% (v/v). The microplate was shaken gently (100 rpm) for 10 min at 25 °C using an orbital shaker and centrifuged at 1500 rpm for 1 min. After equilibration at room temperature for 30 min, the FP values were recorded using excitation and emission filters of 485 nm and 535 nm, respectively. FP competition assay [1] The previously reported LXR agonists and reverse agonists were used as positive test compounds. Final concentrations of 400 nM LXRβ and 10 nM fluorescent tracer were used in the competition binding assays. Assays were performed in triplicate with 2-fold increasing concentrations of LXR modulators. Data were fit to the following equations and the Ki values were calculated according to the method reported. Fragment screening using FP competition assays [1] The fragment library used in screening consisted of 1074 fragments including 486 fragments picked from the Ro3 fragment library of Maybridge, 246 fragments from TargetMol fragment library, and 342 fragments selected from the low molecular weight nature products of ZINC compound library. Fragment screening was performed in black 384-well plates. 10 nM fluorescent tracer 1 was mixed with 400 nM LXRβ-LBD in FP buffer, then the fragments were added at the final concentration of 1 mM. The plates were incubated for 30 min at room temperature, and the FP values were then read on a Victor X5 microplate reader (PerkinElmer). Fragments displacing the tracer by ≥ 25% were defined as the positive hits. The fragments displacing tracer by ≥ 30% were further determined for their Ki values. Co-activator recruitment assay [1] Co-activator recruitment assay was performed by fluorescence polarization assay. The buffer used was same as the FP buffer described above. 1 μM LXRβ-LBD, 0.1 μM labeled co-activator peptide and fragments or GW3965 at the various concentrations were added to the FP buffer. Polarization values were recorded using excitation and emission filters of 485 nm and 535 nm, respectively. EC50 values were determined by fitting dose-response curves with Graphpad Prism 6.0. |
| References |
[1]. Identify liver X receptor β modulator building blocks by developing a fluorescence polarization-based competition assay. Eur J Med Chem. 2019 Sep 15;178:458-467. |
| Additional Infomation | In this study, three fluorescent tracers were designed, synthesized and evaluated, and the tracer 1 was used to screen fragments for LXRβ from a library containing 1074 small fragments. A set of 27 novel fragment hits were identified, and the binding affinity, agonistic activity and the binding mode of top fragment hits were studied. Especially, fragment screening suggested Boc group a privileged building block for designing LXRβ modulators. A co-crystal structure of LXRβ-LBD in complex with a Boc-containing fragment revealed that the Boc group stabilized the His-Trp activation switch of LXRβ-LBD by forming a hydrogen-bonding interaction with His435. The fluorescent tracer and hits generated in this study could help for developing novel LXRβ modulators. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.0028 mL | 5.0142 mL | 10.0285 mL | |
| 5 mM | 0.2006 mL | 1.0028 mL | 2.0057 mL | |
| 10 mM | 0.1003 mL | 0.5014 mL | 1.0028 mL |