FIIN-2 (FIIN 2; FIIN2) is a novel irreversible/covalent pan-FGFR inhibitor with potential anticancer activity. It has IC50 values of 3.09 nM, 4.3 nM, 27 nM, and 45.3 nM, which indicate that it inhibits FGFR1/2/3/4. High anti-proliferative activity of FIIN-2 is seen in vitro against cells that are dependent on FGFR1 or FGFR2 gatekeeper mutants.
Physicochemical Properties
| Molecular Formula | C35H38N8O4 | |
| Molecular Weight | 634.73 | |
| Exact Mass | 634.301 | |
| Elemental Analysis | C, 66.23; H, 6.03; N, 17.65; O, 10.08 | |
| CAS # | 1633044-56-0 | |
| Related CAS # |
|
|
| PubChem CID | 91825767 | |
| Appearance | White to off-white solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Index of Refraction | 1.666 | |
| LogP | 1.82 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 9 | |
| Rotatable Bond Count | 10 | |
| Heavy Atom Count | 47 | |
| Complexity | 1030 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O=C1N(C2C=C(C=C(C=2)OC)OC)CC2=CN=C(N=C2N1CC1C=CC(=CC=1)NC(C=C)=O)NC1C=CC(=CC=1)N1CCN(C)CC1 |
|
| InChi Key | DVBPRWJMHURKHP-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C35H38N8O4/c1-5-32(44)37-26-8-6-24(7-9-26)22-43-33-25(23-42(35(43)45)29-18-30(46-3)20-31(19-29)47-4)21-36-34(39-33)38-27-10-12-28(13-11-27)41-16-14-40(2)15-17-41/h5-13,18-21H,1,14-17,22-23H2,2-4H3,(H,37,44)(H,36,38,39) | |
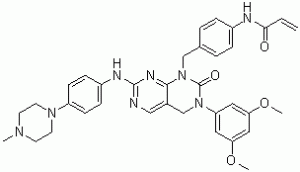
| Chemical Name | N-[4-[[3-(3,5-dimethoxyphenyl)-7-[4-(4-methylpiperazin-1-yl)anilino]-2-oxo-4H-pyrimido[4,5-d]pyrimidin-1-yl]methyl]phenyl]prop-2-enamide | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
FGFR1 (IC50 = 3.1 nM); FGFR2 (IC50 = 4.3 nM); FGFR3 (IC50 = 27 nM); FGFR4 (IC50 = 45 nM) Fibroblast Growth Factor Receptor (FGFR) 1 (IC50 = 0.8 nM), FGFR2 (IC50 = 1.2 nM), FGFR3 (IC50 = 1.5 nM), FGFR4 (IC50 = 22 nM); potent against FGFR resistance mutants: FGFR2 V564F (IC50 = 2.5 nM), FGFR2 N549H (IC50 = 3.3 nM), FGFR3 G697C (IC50 = 3.1 nM); no significant activity against EGFR, ALK, VEGFR2 (IC50 > 1000 nM) [1] - Covalent binding to FGFR kinase domain cysteine residues (FGFR1 C488, FGFR2 C491, FGFR3 C492); no off-target cysteine kinase binding [1] |
| ln Vitro |
FIIN-2 has a potent inhibitory effect on both the gatekeeper mutant of FGFR2 (EC50 of 58 nM) and WT FGFRs (EC50s in the 1- to 93-nM range). With an IC50 of 204 nM, FIIN-2 also moderately inhibits EGFR. FIIN-2 is particularly effective against FGFR2, with EC50s in the 1-nM range, and inhibits the proliferation of FGFR1-4 Ba/F3 cells with EC50s in the single- to double-digit nanomolar range. When it comes to the gatekeeper mutant V564F, FIIN-2 exhibits good potency[1]. Inhibited recombinant FGFR kinase activity: FGFR1 (IC50 = 0.8 nM), FGFR2 (IC50 = 1.2 nM), and resistance mutants (FGFR2 V564F IC50 = 2.5 nM) via covalent irreversible binding [1] - Reduced viability of FGFR-driven cancer cell lines: Ba/F3-FGFR2 V564F (IC50 = 5.3 nM), gastric cancer SNU-16 (FGFR2 amplified, IC50 = 8.7 nM), lung cancer NCI-H1581 (FGFR1 amplified, IC50 = 7.2 nM); no activity in FGFR-negative MCF-7 cells (IC50 > 500 nM) [1] - Suppressed FGFR downstream signaling: 100 nM FIIN-2 reduced p-FGFR (Tyr653/654) by 92% in SNU-16 cells (2 hours), and downregulated p-ERK1/2 (Thr202/Tyr204) and p-AKT (Ser473) by >80% [1] - Induced apoptosis in SNU-16 cells: 200 nM FIIN-2 increased Annexin V-positive cells from 6% (vehicle) to 43% (48 hours); caspase-3/7 activity elevated by 3.6-fold [1] |
| ln Vivo |
Fish treated in the embryonic stage with FIIN-2 or other known FGFR inhibitors exhibit defects in the posterior mesoderm that are comparable to the phenotypes observed after FGFR is genetically knocked down. In all treated embryonic zebrafish, FIIN-2 results in tail morphogenesis phenotypes that range from mild to severe[1]. In nude mice bearing Ba/F3-FGFR2 V564F xenografts (resistance model): Oral FIIN-2 (30 mg/kg/day) for 21 days resulted in 91% tumor growth inhibition (TGI); tumor p-FGFR levels reduced by 85% (immunohistochemistry) [1] - In nude mice bearing SNU-16 (FGFR2-amplified) xenografts: Intraperitoneal injection of FIIN-2 (20 mg/kg, twice daily) for 14 days achieved 82% TGI; median tumor doubling time extended from 5 days (vehicle) to 21 days [1] - In NCI-H1581 (FGFR1-amplified) xenograft mice: Oral FIIN-2 (40 mg/kg/day) for 28 days improved survival (median survival: 56 days vs. 32 days for vehicle) [1] |
| Enzyme Assay |
A broad-coverage kinase binding assay platform based on TR-FRET is used to conduct biochemical experiments. FGFR kinase activity assay: Recombinant human FGFR1/2/3/4 kinases (50 ng/well) were incubated with FIIN-2 (0.01-100 nM) in reaction buffer (25 mM HEPES pH 7.4, 10 mM MgCl2, 1 mM DTT) at 37°C for 30 minutes (covalent binding step). 10 μM ATP and a biotinylated peptide substrate were added, followed by 60-minute incubation at 30°C. Phosphorylated peptide was detected via streptavidin-HRP and chemiluminescence; IC50 values were calculated via nonlinear regression [1] - Covalent binding validation assay: FGFR2 kinase (1 μg) was incubated with 1 μM FIIN-2 for 1 hour, then separated by size-exclusion chromatography. Fractions were analyzed via mass spectrometry to confirm covalent adduct formation (mass shift corresponding to FIIN-2 molecular weight) [1] |
| Cell Assay |
One day after being plated at a density of 1,500 cells per well in 96-well plates, NCI-H2077, NCI-H1581, H520, Kato III, AN3CA, RT112, A2780, 4T1, and SKOV-3 cells are treated with inhibitors. FGFR1 V561M is ectopically overexpressed in NCI-H2077 or NCI-H1581 cells through lentiviral transduction to create the gatekeeper mutation cell lines. Using the Cell-Titer-Glo reagent and the manufacturer's instructions, cell survival is measured 96 hours after the inhibitor is added. GraphPad Prism 5 is used to compute EC50 values. FGF or EGF ligand is also applied to SKOV-3 cells during treatment. A luminometer was used to measure proliferation after 96 hours. Relative values are used to display data. Comparing the luminescence of cells treated with the indicated inhibitor dose to untreated cells is done. Cell proliferation assay (Ba/F3-FGFR2 V564F/SNU-16/NCI-H1581): Cells were seeded in 96-well plates (4×10³ cells/well) and treated with FIIN-2 (0.1 nM-1 μM) for 72 hours. Cell viability was measured via tetrazolium-based colorimetric assay; absorbance at 570 nm was recorded, and IC50 values were determined via four-parameter logistic fitting [1] - Western blot assay (FGFR/ERK/AKT): SNU-16 cells were treated with FIIN-2 (10-200 nM) for 2 hours, lysed in RIPA buffer (supplemented with protease/phosphatase inhibitors). Lysates (30 μg protein) were separated by 8% SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against p-FGFR (Tyr653/654), total FGFR, p-ERK1/2, total ERK1/2, p-AKT, total AKT, and GAPDH. Signals were detected via chemiluminescence [1] - Apoptosis assay (SNU-16): Cells were treated with FIIN-2 (50-200 nM) for 48 hours, stained with Annexin V-FITC and propidium iodide, and analyzed by flow cytometry. Caspase-3/7 activity was measured via fluorometric assay with a caspase-specific substrate [1] |
| Animal Protocol |
Treatment of fish in the embryonic state with either FIIN-2 causes defects to the posterior mesoderm similar to the phenotypes reported following genetic knockdown of FGFR or treatment with other reported FGFR inhibitors. FIIN-2 causes mild or severe phenotypes to the tail morphogenesis in all treated embryonic zebrafish. zebrafish Ba/F3-FGFR2 V564F xenograft model (nude mice): 6-week-old female nude mice were subcutaneously injected with 5×10⁶ Ba/F3-FGFR2 V564F cells. When tumors reached 100-120 mm³, mice were randomized to vehicle (0.5% methylcellulose + 0.2% Tween 80) or FIIN-2 groups (30 mg/kg/day, oral gavage). Treatments were administered once daily for 21 days; tumor volume (length × width² / 2) and body weight were measured every 3 days [1] - SNU-16 xenograft model (nude mice): Female nude mice were implanted with 1×10⁷ SNU-16 cells subcutaneously. When tumors reached 150 mm³, mice received FIIN-2 (20 mg/kg, intraperitoneal injection) twice daily for 14 days. Drug was dissolved in 5% DMSO + 95% sesame oil; tumor samples were collected at study end for immunohistochemistry [1] - NCI-H1581 xenograft model (nude mice): Male nude mice were injected with 2×10⁶ NCI-H1581 cells subcutaneously. When tumors reached 100 mm³, mice received FIIN-2 (40 mg/kg/day, oral gavage) for 28 days. Drug was dissolved in 10% DMSO + 40% PEG400 + 50% normal saline; survival time was recorded [1] |
| ADME/Pharmacokinetics |
In mice: Oral bioavailability of FIIN-2 was 45% (30 mg/kg dose); plasma half-life (t1/2) = 4.2 hours; maximum plasma concentration (Cmax) = 3.8 μM at 1 hour post-oral administration [1] - In rats: Intravenous administration (10 mg/kg) showed a clearance rate of 13 mL/min/kg; volume of distribution at steady state (Vss) = 0.9 L/kg [1] - Plasma protein binding: 99.2% binding to human plasma proteins (measured via ultrafiltration method) [1] - Tumor penetration: In SNU-16 xenografts, FIIN-2 tumor/plasma concentration ratio = 1.8 (2 hours post-intraperitoneal injection) [1] |
| Toxicity/Toxicokinetics |
In 21-day Ba/F3-FGFR2 V564F xenograft study (30 mg/kg/day, oral): No significant weight loss (>8%); serum ALT (28 ± 4 U/L) and BUN (19 ± 3 mg/dL) were within normal ranges (ALT: 20-40 U/L, BUN: 15-25 mg/dL) [1] - In 14-day SNU-16 xenograft study (20 mg/kg, twice daily, intraperitoneal): 1/8 mice showed mild diarrhea (resolved within 3 days); no histopathological changes in liver, kidney, or spleen [1] - In 28-day NCI-H1581 xenograft study (40 mg/kg/day, oral): No treatment-related mortality; 2/10 mice showed mild hair loss (reversed post-treatment) [1] |
| References |
[1]. Development of covalent inhibitors that can overcome resistance to first-generation FGFR kinase inhibitors. Proc Natl Acad Sci U S A. 2014 Nov 11;111(45):E4869-4877. |
| Additional Infomation |
FIIN-2 is a first-in-class covalent irreversible FGFR inhibitor, designed to overcome resistance to first-generation reversible FGFR inhibitors (e.g., BGJ398) by forming a covalent bond with FGFR kinase domain cysteines [1] - Its covalent mechanism confers prolonged target inhibition (t1/2 for FGFR reactivation >24 hours in SNU-16 cells), supporting less frequent dosing [1] - Preclinical data support FIIN-2 as a candidate for FGFR-amplified or mutation-driven cancers (gastric, lung, bladder) with acquired resistance to reversible FGFR inhibitors [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.94 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (3.94 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (3.94 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.5755 mL | 7.8774 mL | 15.7547 mL | |
| 5 mM | 0.3151 mL | 1.5755 mL | 3.1509 mL | |
| 10 mM | 0.1575 mL | 0.7877 mL | 1.5755 mL |