Physicochemical Properties
| Molecular Formula | C27H37CLN2O4 |
| Molecular Weight | 489.05 |
| Exact Mass | 488.244185 |
| Elemental Analysis | C, 66.31; H, 7.63; Cl, 7.25; N, 5.73; O, 13.09 |
| CAS # | 2560549-35-9 |
| Related CAS # | 2560549-35-9 (HCl); 1593673-23-4 |
| PubChem CID | 169490895 |
| Appearance | Typically exists as solids at room temperature |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 34 |
| Complexity | 645 |
| Defined Atom Stereocenter Count | 1 |
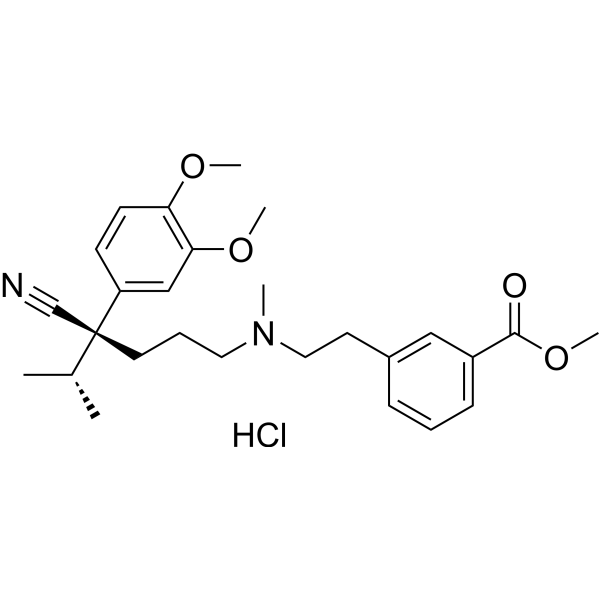
| SMILES | CC(C)[C@](CCCN(C)CCC1=CC(=CC=C1)C(=O)OC)(C#N)C2=CC(=C(C=C2)OC)OC.Cl |
| InChi Key | JHOAIMAMBNLIMD-YCBFMBTMSA-N |
| InChi Code | InChI=1S/C27H36N2O4.ClH/c1-20(2)27(19-28,23-11-12-24(31-4)25(18-23)32-5)14-8-15-29(3)16-13-21-9-7-10-22(17-21)26(30)33-6;/h7,9-12,17-18,20H,8,13-16H2,1-6H3;1H/t27-;/m0./s1 |
| Chemical Name | methyl 3-[2-[[(4S)-4-cyano-4-(3,4-dimethoxyphenyl)-5-methylhexyl]-methylamino]ethyl]benzoate;hydrochloride |
| Synonyms | MSP-2017 hydrochloride; 2560549-35-9; Etripamil hydrochloride; Etripamil (hydrochloride); 9TA9AC3H8Z; orb2945709; (-)-MSP-2017 hydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | L-type calcium channel |
| ln Vitro | Etripamil is a novel, short-acting, nondihydropyridine L-type calcium channel blocker formulated for intranasal administration. It exerts its anti-arrhythmic effect by inhibiting voltage-dependent L-type calcium channels (Cav1.2 and Cav1.3), which are critical for mediating calcium entry into cardiac myocytes and regulating excitation-contraction coupling and nodal conduction. Specifically, etripamil targets the L-type calcium channels expressed on atrioventricular (AV) nodal cells, arterial smooth muscles, and contractile myocardial cells. By blocking calcium influx, the drug prolongs the refractory period and slows electrical conduction through the AV node, thereby interrupting the re-entrant circuits responsible for AV-nodal dependent paroxysmal supraventricular tachycardia (PSVT) and facilitating the restoration of sinus rhythm. |
| ln Vivo |
Etripamil is a fast-acting agent with a short duration of action. Etripamil prolonged the baseline PR interval by 8% to 10% approximately five minutes following an intranasal dose of 70 mg. Clinical data shows significant efficacy in terminating arrhythmias within 15 to 60 minutes post-administration, but its effects diminish rapidly, with no significant difference in conversion rates observed by 300 minutes compared to placebo. Etripamil is a nondihydropyridine, L-type calcium channel blocker. It is a fast-acting drug with a short duration of action. The nasal formulation of etripamil was first approved by the FDA on December 12, 2025 for the conversion of acute symptomatic episodes of paroxysmal supraventricular tachycardia (PSVT) to sinus rhythm in adults. ETRIPAMIL is a small molecule drug with a maximum clinical trial phase of III (across all indications) and has 4 investigational indications. |
| Animal Protocol | Objectives: The purpose of this study was to assess the efficacy and safety of etripamil nasal spray, a short-acting calcium-channel blocker, for the rapid termination of paroxysmal supraventricular tachycardia (SVT). Methods: This phase 2 study was performed during electrophysiological testing in patients with previously documented SVT who were induced into SVT prior to undergoing a catheter ablation. Patients in sustained SVT for 5 min received either placebo or 1 of 4 doses of active compound. The primary endpoint was the SVT conversion rate within 15 min of study drug administration. Secondary endpoints included time to conversion and adverse events. |
| ADME/Pharmacokinetics |
Absorption After one 70 mg dose of etripamil, mean (%CV) area under the concentration-time curve (AUC) is approximately 5461 (51.6%) ng x min/mL and the Cmax is approximately 99 (64.6%) ng/mL. After a second 70 mg dose of etripamil administered 10 minutes after the first dose, mean (%CV) AUC is approximately 7721 (50.3%) ng x min/mL and the Cmax is approximately 132 (59.1%) ng/mL. The median (range) Tmax is 7 minutes (3 to 20 minutes) following a single intranasal administration of 70 mg. Median Tmax is 13 minutes (3 to 35 minutes) following a second intranasal administration of 70 mg. Route of Elimination After a single dose of radiolabeled intranasal etripamil 70 mg to healthy subjects, approximately 29% of the dose was recovered in urine (<0.05% unchanged), 26% was recovered in feces (<0.05% unchanged), and the remainder was recovered on nose and face tissues. Approximately 71% of the total administered dose was recovered in 7-10 days. Volume of Distribution Etripamil mean apparent volume of distribution ranges from approximately 2200 to 3500 L. Protein Binding Etripamil plasma protein binding is approximately 50%. Metabolism / Metabolites Etripamil metabolic pathways include hydrolysis, demethylation, N-dealkylation, and secondary oxidation, glucuronidation, and taurine conjugation. Etripamil is primarily metabolized by blood esterases and hepatic metabolism, primarily via CYP3A4, and CYP3A5. Etripamil contains a methyl ester which renders it metabolically sensitive to blood-borne esterases. Biological Half-Life Average etripamil concentration fell by approximately 60% of its peak value (Cmax) at 25 minutes and 80% of the Cmax by 60 minutes after dosing. Subsequently, concentrations decrease at a slower rate, and this decline is associated with a half-life of approximately 2.5 hours. |
| References |
[1]. CO143 Impact of Investigational, at-Home, Self-Administered, Intranasal Etripamil on the Need for Additional Medical Intervention in Patients with Supraventricular TachycardiaJ. Value in Health, 2023, 26(6): S41. |
| Additional Infomation | Results: One hundred four patients were dosed. Conversion rates from SVT to sinus rhythm were between 65% and 95% in the etripamil nasal spray groups and 35% in the placebo group; the differences were statistically significant (Pearson chi-square test) in the 3 highest active compound dose groups versus placebo. In patients who converted, the median time to conversion with etripamil was <3 min. Adverse events were mostly related to the intranasal route of administration or local irritation. Reductions in blood pressure occurred predominantly in the highest etripamil dose. Conclusions: Etripamil nasal spray rapidly terminated induced SVT with a high conversion rate. The safety and efficacy results of this study provide guidance for etripamil dose selection for future studies involving self-administration of this new intranasal calcium-channel blocker in a real-world setting for the termination of SVT. (Efficacy and Safety of Intranasal MSP-2017 [Etripamil] for the Conversion of PSVT to Sinus Rhythm [NODE-1]; NCT02296190). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0448 mL | 10.2239 mL | 20.4478 mL | |
| 5 mM | 0.4090 mL | 2.0448 mL | 4.0896 mL | |
| 10 mM | 0.2045 mL | 1.0224 mL | 2.0448 mL |