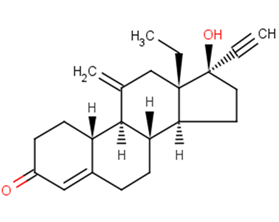
Etonogestrel (ORG3236; ORG-3236; Nexplanon; Implano; NuvaRing; Circlet) is a synthetic progestin derivative with long half-life that is an active metabolite of desogestrel and has been used in numerous hormonal contraceptive devices. It is a birth control pill that women can use as an implant under their skin under their upper arm or as a vaginal ring combined with ethinylestradiol.
Physicochemical Properties
| Molecular Formula | C22H28O2 |
| Molecular Weight | 324.4565 |
| Exact Mass | 324.208 |
| Elemental Analysis | C, 81.44; H, 8.70; O, 9.86 |
| CAS # | 54048-10-1 |
| Related CAS # | 54048-10-1 |
| PubChem CID | 6917715 |
| Appearance | White to off-white solid powder |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 473.1±45.0 °C at 760 mmHg |
| Melting Point | 182-184ºC |
| Flash Point | 201.1±21.3 °C |
| Vapour Pressure | 0.0±2.7 mmHg at 25°C |
| Index of Refraction | 1.575 |
| LogP | 4.22 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 24 |
| Complexity | 677 |
| Defined Atom Stereocenter Count | 6 |
| SMILES | O([H])[C@@]1(C#C[H])C([H])([H])C([H])([H])[C@@]2([H])[C@]3([H])C([H])([H])C([H])([H])C4=C([H])C(C([H])([H])C([H])([H])[C@]4([H])[C@@]3([H])C(=C([H])[H])C([H])([H])[C@@]21C([H])([H])C([H])([H])[H])=O |
| InChi Key | GCKFUYQCUCGESZ-BPIQYHPVSA-N |
| InChi Code | InChI=1S/C22H28O2/c1-4-21-13-14(3)20-17-9-7-16(23)12-15(17)6-8-18(20)19(21)10-11-22(21,24)5-2/h2,12,17-20,24H,3-4,6-11,13H2,1H3/t17-,18-,19-,20+,21-,22-/m0/s1 |
| Chemical Name | (8S,9S,10R,13S,14S,17R)-13-ethyl-17-ethynyl-17-hydroxy-11-methylidene-2,6,7,8,9,10,12,14,15,16-decahydro-1H-cyclopenta[a]phenanthren-3-one |
| Synonyms | ORG 3236; ORG3236; ORG-3236; Implanon; Etonogestrel; nexplanon; 3-keto-desogestrel, 3-ketodesogestrel, 3-oxo desogestrel, 3-oxodesogestrel |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Human Endogenous Metabolite |
| ln Vitro |
Etonogestrel reduces fertility through preventing the release of luteinizing hormone (LH), a reproductive hormone crucial to ovulation. Etonogestrel also makes cervical mucus more viscous, which prevents spermatozoa from passing through and modifies the uterine lining to stop a fertilized egg from implanting into the endometrium. Etonogestrel, a progestin, is an oxidative substrate of CYP 3A4. Etonogestrel induces the expression of FKBP51 mRNA and protein in cultured human endometrial stromal cells (HESCs)[1]. |
| ln Vivo | Etonogestrel (Implanon, Nexplanon, 3-Oxodesogestrel, 3-keto-Desogestrel) is a synthetic version of progesterone, a hormone that is naturally present in female sex. |
| Cell Assay | In basal medium (BM), which is a 1:1 v/v mixture of Dulbecco's MEM and Ham's F-12 without phenol red, thawed HESCs are grown to confluence with 100U/ml penicillin, 100μg/ml streptomycin, and 0.25μg/ml fungizone complex supplemented with 10% charcoal-stripped calf serum. Confluent HESCs are cultured in parallel in BM with 0.1% ethanol (vehicle control), 10 nM ethradiol or E2+100 nM progesterone, or 100 nM of either MPA (medroxyprogesterone acetate) or ETO (etonogestrel). The cultures are kept in a serum-free defined media (DM) that contains BM plus ITS+ (insulin, transferrin, selenium, and linoleic acid) premix, 5 μM of FeSO4, 50 μM of ZnSO4, 1 nM of CuSO4, 20 nM of Na2SeO3 trace elements, 50 μg/ml of ascorbic acid, and 50 ng/ml of recombinant epidermal growth factor. The cultures are kept in this medium for seven days after the serum is removed twice using 1X phosphate-buffered saline (PBS) to remove any remaining serum. After twice washing the cultures in 1X PBS to get rid of any remaining steroids, DM containing the appropriate vehicle control or steroids is added to the cultures for either six or twenty-four hours. Following their incubation periods, HESCs are stored at −70°C after being rinsed with ice-cold 1X PBS and prepared for total RNA and protein extraction. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Vaginal administration of etonogestrel is known to be significantly absorbed through the vaginal epithelium but it does not increase the levels of etonogestrel in the urine. On the other hand, oral administration is absorbed in the GI tract and it goes through the first-pass metabolism. When etonogestrel is administered subdermally it is absorbed rapidly into the bloodstream and it presents a bioavailability of 82%. It is reported that the implant releases around 60 mcg per day in the first 3 months and then decreases steady reaching a concentration of 30 mcg at the end of year 2. The elimination of etonogestrel and its metabolites is mainly done renally. The apparent volume of distribution of etonogestrel is of around 201 L. The clearance rate of etonogestrel is reported to be of 7.5 L/h. Metabolism / Metabolites Etonogestrel is highly metabolized in the liver by the action of the cytochrome isoenzyme 3A4 mainly by the presence of hydroxylation, sulfate conjugation and glucuronide conjugation reactions. Biological Half-Life The elimination half-life of etonogestrel is reported to be of 25 hours which indicates a reversible contraceptive effect. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Etonogestrel is available in the United States in a combination contraceptive vaginal ring (NuvaRing) that releases 120 mcg of etonogestrel and 15 mcg of ethinyl estradiol daily, and subcutaneous implants (Implanon, Nexplanon) that release etonogestrel at a decreasing rate over a 3-year period. Based on the available evidence, expert opinion in the United States holds that postpartum women who are breastfeeding should not use combined hormonal contraceptives (e.g., NuvaRing) during the first 3 weeks after delivery because of concerns about increased risk for venous thromboembolism and generally should not use combined hormonal contraceptives during the fourth week postpartum because of concerns about potential effects on breastfeeding performance. Postpartum breastfeeding women with other risk factors for venous thromboembolism generally should not use combined hormonal contraceptives until 6 weeks after delivery. World Health Organization guidelines are more restrictive, stating that combined oral contraceptives should not be used in nursing mothers before 42 days postpartum and the disadvantages of using the method generally outweigh the advantages between 6 weeks and 6 months postpartum. A decrease in milk supply can happen over the first few days of estrogen exposure. Expert opinion holds that the risks of progestin-only contraceptive products usually are acceptable for nursing mothers at any time postpartum. A review of published data concluded that the etonogestrel implant appears to have a risk of lactation suppression of about 0.9%. Low quality evidence indicates that there may be no difference in breastfeeding rates at 6 months between immediate and delayed insertion of progestin-releasing IUDs. Some preliminary evidence indicates that secretory activation (lactogenesis II) might be delayed by the etonogestrel implant. However, other studies found no difference between immediate and delayed insertion of an etonogestrel implant. ◉ Effects in Breastfed Infants Forty-two women had an etonogestrel subcutaneous implant (Implanon) inserted between 28 and 56 days postpartum. Compared with the infants of 38 similar mothers who had a nonhormonal intrauterine device, no statistically significant difference was found in infant illnesses or growth rates between the groups, except for a statistically nonsignificant higher weight gain in the male infants, and respiratory conditions and skin disorders in the infants of women who received the implants. Infants were followed up at 3 years of age; no differences in growth or psychomotor development were found. A non-blinded, randomized study of exclusively breastfeeding women compared those who received an etonogestrel implant 24-48 hours after delivery (n = 20) to those who received a 150 mg depot medroxyprogesterone acetate injection at 6 weeks postpartum (n = 20). No difference in infant weight gain was noted between the two groups. A randomized, prospective study compared the growth of infants of mothers who received an etonogestrel implant either within 48 hours (n = 50) of delivery or 6 weeks (n = 50) postpartum. Breastfeeding rates and growth of the infants were not significantly different between the groups over the first year of life. A prospective, nonrandomized trial compared 4 contraceptives in 10 women each to assess their effect on milk production. One of the following were begun on day 42 postpartum as chosen by the mother: combined ethinyl estradiol 30 mcg plus levonorgestrel 150 mcg (Microvlar), etonogestrel implant (Implanon), levonorgestrel intrauterine system (Myrena), or a copper IUD (Optima). Milk intake was measured using deuterium oxide given to the mother and measured in the infants' saliva as well as numbers of wet diapers per day. Infants were also weighed and measured to assess growth. No differences in milk intake or infant growth were observed between the methods from days 42 through 63. A small study randomized postpartum women to either an etonogestrel implant (Implanon) within 48 hours postpartum (n = 12) or no contraception for the first 6 weeks postpartum (n = 12). Breastmilk intake was measured in the first 48 hours postpartum and on day 29 postpartum. No difference in milk intake was found between the two groups and no difference was seen in the weight gain of newborns during the follow-up period. ◉ Effects on Lactation and Breastmilk Forty-two women had an etonogestrel subcutaneous implant (Implanon) inserted between 28 and 56 days postpartum. Compared with 38 similar mothers who had a nonhormonal intrauterine device, no difference was found in milk volume, or in milk lactose, protein or fat content. No difference was seen between the two groups in duration of lactation, averaging 421 days in the Implanon group and 423 days in the IUD group. A non-blinded, randomized study of exclusively breastfeeding women compared those who received an etonogestrel implant 24 to 48 hours after delivery (n = 20) to those who received a 150 mg depot medroxyprogesterone acetate injection at 6 weeks postpartum (n = 20). The rates of exclusive breastfeeding were similar between the groups at 6 and 12 weeks postpartum. A randomized, controlled study compared etonogestrel implant insertion at 1 to 3 days postpartum (n = 35) to insertion at 4 to 8 weeks postpartum (n = 34). Several outcome parameters were measured: time to lactogenesis II, prevalence of lactation failure, use of formula supplementation and milk composition at 6 weeks postpartum. No differences were found in any of the outcomes between the two groups. A woman was breastfeeding a newborn successfully with good infant growth. On day 39 postpartum, an etonogestrel implant (Nexplanon) was inserted. By day 70 postpartum, the mother reported a decrease in milk supply and infant had lost weight, going from the 44th percentile to the 6th percentile for growth. Five weeks later, the mother had transitioned completely to formula feeding. The authors requested reports on etonogestrel from the FDA and found a similar case of loss of milk supply. From a review of 4 published studies, the authors estimated that there is about a 0.9% (range 0.2 to 5.4%) chance of lactation suppression with the etonogestrel implant. A randomized, nonblinded trial compared the insertion of an etonogestrel contraceptive implant (brand not specified) in postpartum women 14 to 24 years old either before discharge or at 6 weeks postpartum. Breastfeeding rates at 3 and 6 months postpartum were slightly higher in the women who had immediate placement of the insert, but the differences were not statistically significant. A randomized, prospective study compared the growth of infants of mothers who received an etonogestrel implant either within 48 hours (n = 50) of delivery or 6 weeks (n = 50) postpartum. Breastfeeding rates were not significantly different between the groups over the first year of life. A study in Malawi compared the breastfeeding rates between women who received an etonogestrel (n = 28) or levonorgestrel (n = 112) implant immediately postpartum. Mothers chose the method and were followed for 2 years postpartum. Most women breastfed for 2 years. No difference was seen in the exclusive breastfeeding rate at 6 months between the groups nor in the continuation of breastfeeding to 2 years. A study of women who expressed a desire to breastfeed randomized them to receive a progestin implant (presumably etonogestrel) either in the delivery room or at 24 to 48 hours after delivery. The time to lactogenesis II was not significantly different between the two groups. A small, nonrandomized, prospective pilot study compared the time to secretory activation in women who received an etonogestrel implant (Nexplanon; n = 8) to those receiving no contraception (n = 24). Women who received the etonogestrel implant had a delay in secretory activation compared to those who did not, as measured by biochemical markers. A noninferiority study randomized postpartum women to insertion of an etonogestrel implant at either 0 to 2 hours (n = 35) or 24 to 48 hours (n = 34) post-delivery. No significant difference was found between the two groups in the time to lactogenesis II. A study randomized postpartum women to receive the etonogestrel implant either within 30 minutes of placental delivery, 24 to 72 hours postpartum, or 6 or more weeks postpartum. There was no significant difference in time to lactogenesis II with an average time of 62 hours. Duration and exclusivity of breastfeeding, satisfaction with the implant, and reports of heavy or irregular bleeding were similar among all groups. A woman reported persistent galactorrhea after implantation of Nexplanon 2 months after delivery and breastfeeding her infant. The galactorrhea persisted for 2 years until the Nexplanon implant was removed. Her galactorrhea was improved 1 and 7 months after removal. Protein Binding Etonogestrel is highly bound to plasma proteins being mainly albumin followed by sex-hormone binding globulin. The protein bound form of the etonogestrel represents around 96-99% of the administered dose. |
| References |
[1]. Dionne D. Maddox, et al. Etonogestrel (Implanon), Another Treatment Option for Contraception. P T. 2008 Jun; 33(6): 337–347. [2]. Ozlem Guzeloglu Kayisli, et al. Progestins Upregulate FKBP51 Expression in Human Endometrial Stromal Cells to Induce Functional Progesterone and Glucocorticoid Withdrawal: Implications for Contraceptive- Associated Abnormal Uterine Bleeding. PLoS One. 201 . |
| Additional Infomation |
Etonogestrel is a 17beta-hydroxy steroid, a 3-oxo-Delta(4) steroid and a terminal acetylenic compound. It has a role as a contraceptive drug, a progestin and a female contraceptive drug. Etonogestrel molecule is a 3-ketodesogestrel or 19-nortestosterone which is a synthetic biologically active metabolite of progestin desogestrel. The first product including etonogestrel was developed by the Merck subsidiary Organon and FDA approved in 2001. Etonogestrel is a Progestin. Etonogestrel is a synthetic form of the naturally occurring female sex hormone progesterone. Etonogestrel binds to the cytoplasmic progesterone receptors in the reproductive system and subsequently activates progesterone receptor mediated gene expression. As a result of the negative feedback mechanism, luteinizing hormone (LH) release is inhibited, which leads to an inhibition of ovulation and an alteration in the cervical mucus and endometrium. See also: Ethinyl estradiol; etonogestrel (component of). Drug Indication Etonogestrel is administered in subdermal implants as long-acting reversible contraception. It is known to be effective in postpartum insertion including breastfeeding women. Etonogestrel is part of the long-acting contraceptive implants that prevent pregnancy. The implant's effect can remain for 5 years. FDA Label Mechanism of Action Etonogestrel binds with high affinity to the progesterone and estrogen receptors in the target organs. From the target organs, they include the female reproductive tract, mammary gland, hypothalamus, and pituitary. Once bound, this drug changes the synthesis of different proteins which in order decreases the level of gonadotropin-releasing hormone and the luteinizing hormone. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 50~65 mg/mL (154.1~200.3 mM) Ethanol: 65 mg/mL (~200.3 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.71 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0820 mL | 15.4102 mL | 30.8204 mL | |
| 5 mM | 0.6164 mL | 3.0820 mL | 6.1641 mL | |
| 10 mM | 0.3082 mL | 1.5410 mL | 3.0820 mL |