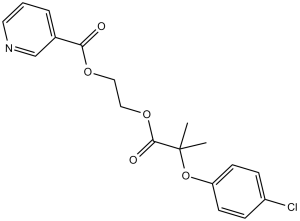
Etofibrate, the ethandiol-1,2 diester of the nicotinic and clofibric acid, is a combination of conjugated clofibrate and niacin that is used to treat hyperlipemia. Etofibrate treatment heightens the cytosolic glycerol-3-phosphate dehydrogenase activity and the total carnitine concentration in the liver of rats, whereas it reduces triacylglycerol and cholesterol concentrations. Etofibrate decreases plasma levels of cholesterol, triacylglycerols, free fatty acids (FFA) and glycerol, as well as the total and unesterified cholesterol concentrations, in liver microsomes of rats.
Physicochemical Properties
| Molecular Formula | C18H18CLNO5 | |
| Molecular Weight | 363.79 | |
| Exact Mass | 363.087 | |
| CAS # | 31637-97-5 | |
| Related CAS # |
|
|
| PubChem CID | 65777 | |
| Appearance | White to off-white solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Boiling Point | 486.8±35.0 °C at 760 mmHg | |
| Melting Point | 42-44ºC | |
| Flash Point | 248.2±25.9 °C | |
| Vapour Pressure | 0.0±1.2 mmHg at 25°C | |
| Index of Refraction | 1.555 | |
| LogP | 3.15 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 9 | |
| Heavy Atom Count | 25 | |
| Complexity | 449 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O=C(C1=CC=CN=C1)OCCOC(C(C)(OC2=CC=C(Cl)C=C2)C)=O |
|
| InChi Key | XXRVYAFBUDSLJX-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C18H18ClNO5/c1-18(2,25-15-7-5-14(19)6-8-15)17(22)24-11-10-23-16(21)13-4-3-9-20-12-13/h3-9,12H,10-11H2,1-2H3 | |
| Chemical Name | 2-[2-(4-Chlorophenoxy)-2-methylpropanoyl]oxyethyl pyridine-3-carboxylate | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Etofibrate targets peroxisome proliferator-activated receptor α (PPARα) [1] |
| ln Vitro | In vitro activity: Etofibrate treatment heightens the cytosolic glycerol-3-phosphate dehydrogenase activity and the total carnitine concentration in the liver of rats, whereas it reduces triacylglycerol and cholesterol concentrations. Etofibrate decreases plasma levels of cholesterol, triacylglycerols, free fatty acids (FFA) and glycerol, as well as the total and unesterified cholesterol concentrations, in liver microsomes of rats. Etofibrate increases the activity of liver cytosolic glycerol-3-P dehydrogenase in the rats, whereas it decreases the activity of both microsomal HMG-CoA reductase and cholesterol 7 alpha-hydroxylase and does not affect acyl-CoA: cholesterol acyltransferase (ACAT). Etofibrate decreases the availability of lipolytic products in the liver by acting on their release from adipose tissue and on their intrinsic hepatic metabolism. Etofibrate stimulates the rate of fatty acid re-esterification when incubations are done under basal conditions in epididymal fat pad pieces from fed rats. |
| ln Vivo |
The ethandiol-1,2 diester of nicotinic and clofibric acids is called etofibrate. Both in humans and animals, etofibrate has demonstrated to be a highly effective hypolipidemic medication. Following a 10-day Etofibrate treatment period, the rats' body weight is comparable to that of the control group, but their liver weight has dramatically increased while their levels of triacylglycerol and plasma cholesterol have dropped. When the enterohepatic circulation is interrupted, etofibrate administration causes the animals' bile flow to rise. This effect is most noticeable during the first two hours, and after 30 and 90 minutes, the difference with the controls is statistically significant. Rats treated with Etofibrate and control groups exhibit a highly significant linear association in the cumulative amount of bile secreted plotted against time, with the Etofibrate group's slope being much higher than the control group's[1]. In normolipidemic Wistar rats (200–250 g), intraperitoneal administration of Etofibrate (100 mg/kg and 200 mg/kg) significantly increased bile production. At 2 hours post-administration, bile flow rate was elevated by ~22% (100 mg/kg) and ~35% (200 mg/kg) compared to vehicle control. The effect persisted for up to 4 hours, with bile flow remaining ~18% (100 mg/kg) and ~28% (200 mg/kg) higher than control at 4 hours [1] - Etofibrate (200 mg/kg, i.p.) modified bile composition: cholesterol content in bile decreased by ~30% at 2 hours post-administration, while total bile acid concentration increased by ~25%. Phospholipid levels in bile showed no significant change compared to vehicle control [1] |
| Animal Protocol |
Male Sprague Dawley rats weighing 200 to 220 g, fed ad libitum a standard laboratory diet and subjected to a 12 hr on-off light cycle and 22 to 24°C are used. Food is removed from the cages at the onset of the light cycle (7.00 a.m.) and 3 hr later, Etofibrate is administered by stomach tube without anesthesia at a dose of 300 mg/kg body weight/day to one group of animals whereas another, control, group is treated with the medium. After treatments, rats are again allowed free access to food. On the 10th day, the animals are treated as above, but 2 hr after receiving the Etofibrate or the medium the rats are anesthetized with an intraperitoneal injection (0.30 mL/100 g body weight). Animals are maintained at 37°C in a thermostatically controlled cabinet Male Sprague Dawley rats Normolipidemic rat bile production assay: Male Wistar rats (200–250 g) were fasted for 12 hours but allowed free access to water. Rats were anesthetized with an appropriate anesthetic, and the common bile duct was cannulated with a polyethylene tube to collect bile. After a 30-minute stabilization period (basal bile collection), Etofibrate was dissolved in a suitable solvent (e.g., dimethyl sulfoxide diluted with saline) and administered intraperitoneally at doses of 100 mg/kg or 200 mg/kg. Vehicle control rats received the same volume of solvent. Bile was collected at 1-hour intervals for 4 hours, and bile volume was measured gravimetrically. Bile samples were analyzed for cholesterol, total bile acids, and phospholipid concentrations using standard biochemical assays [1] |
| Toxicity/Toxicokinetics |
In vivo toxicity: Intraperitoneal administration of Etofibrate (100–200 mg/kg) in normolipidemic rats did not cause obvious toxic symptoms (e.g., lethargy, respiratory distress, abnormal behavior) during the 4-hour observation period. No significant changes in body weight or gross organ appearance were observed at the end of the experiment [1] |
| References | [1]. Bocos C, et al. Effect of etofibrate on bile production in the normolipidemic rat. Gen Pharmacol. 1995 May;26(3):537-42 |
| Additional Infomation |
Etofibrate is a monocarboxylic acid. Etofibrate is a fibrate produced by the combination of clofibrate ester linked to niacin. These components separate in the body slowly allowing for pharmacokinetics similar to controlled-release formulations. Etofibrate is an orally-available combination of clofibrate and niacin linked together by an ester bond. In the body clofibrate and niacin separate, acting as a controlled-release formulation. Etofibrate is a fibrate class hypolipidemic agent that acts primarily through activation of PPARα [1] - Its effect on bile production in normolipidemic rats suggests a role in modulating hepatobiliary lipid metabolism, potentially via increased bile acid synthesis and secretion, and reduced biliary cholesterol excretion [1] - The dose-dependent increase in bile flow indicates that Etofibrate enhances hepatocellular bile secretion, which may contribute to its cholesterol-lowering effect by promoting fecal cholesterol excretion via bile [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.87 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (6.87 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (6.87 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7488 mL | 13.7442 mL | 27.4884 mL | |
| 5 mM | 0.5498 mL | 2.7488 mL | 5.4977 mL | |
| 10 mM | 0.2749 mL | 1.3744 mL | 2.7488 mL |