Physicochemical Properties
| Molecular Formula | C12H14O4 |
| Molecular Weight | 222.2372 |
| Exact Mass | 222.089 |
| CAS # | 4046-02-0 |
| PubChem CID | 736681 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 382.3±0.0 °C at 760 mmHg |
| Melting Point | 63-65 °C(lit.) |
| Flash Point | 132.5±17.2 °C |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.566 |
| LogP | 1.94 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 16 |
| Complexity | 249 |
| Defined Atom Stereocenter Count | 0 |
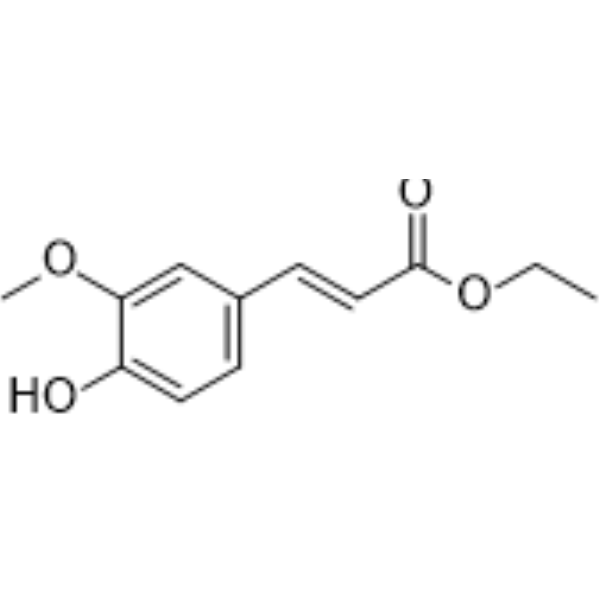
| SMILES | CCOC(=O)/C=C/C1=CC(=C(C=C1)O)OC |
| InChi Key | ATJVZXXHKSYELS-FNORWQNLSA-N |
| InChi Code | InChI=1S/C12H14O4/c1-3-16-12(14)7-5-9-4-6-10(13)11(8-9)15-2/h4-8,13H,3H2,1-2H3/b7-5+ |
| Chemical Name | ethyl (E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Ethyl ferulate targets heme oxygenase-1 (HO-1), AMP-activated protein kinase (AMPK), and nuclear factor erythroid 2-related factor 2 (Nrf2)[1,4] - Ethyl ferulate also modulates inflammatory mediators (e.g., TNF-α, IL-6, iNOS) and oxidative stress-related proteins (e.g., SOD, GSH-Px)[2,3,5] |
| ln Vitro |
In astrocytes and hippocampal neurons, ethyl ferulate (1-50 μM) increases HO activity, HO-1 mRNA, and protein expression [1]. The meta-scaffold GOX senses cell death, and ethyl ferulate (5 μM, 12 hours) can safeguard generation neural cells by ethyl ferulate (10-50 μM, 24). Aβ-peptide (1-42) senses HO-1[1] and protects hippocampus nerves. ROS build-up, cytotoxicity, 3-NT production, and possible peroxidation [2]. The RPE cell workstation is shielded from the CoCl2 (150 µM)-induced reduction in cell viability by ethyl ferulate (20-160 µM, 24 hours) [5]. 40 μM of ethanol ferulate activates Nrf-2 and decreases RPE cells in a 24-hour period. - Protection of rat cortical neurons against oxidative stress: Ethyl ferulate (10, 25, 50 μM) pretreatment increased cell viability (by 28%–65%) in H2O2-induced oxidative stress models (MTT assay). It induced HO-1 expression (2.5–4.0-fold increase via Western blot) and reduced reactive oxygen species (ROS) production (by 35%–58%) compared to the stress-only group[1] - Inhibition of Aβ1-42-induced neurotoxicity: In rat hippocampal neurons, Ethyl ferulate (5, 10, 20 μM) reversed Aβ1-42-induced cell death (viability increased by 22%–50%) and reduced ROS accumulation (by 30%–52%). It also restored SOD and GSH-Px activities (by 25%–48%) and decreased MDA levels (by 28%–50%)[2] - Suppression of inflammation in RAW264.7 macrophages: Ethyl ferulate (10, 20, 40 μM) inhibited LPS-induced TNF-α (by 40%–72%) and IL-6 (by 35%–68%) secretion (ELISA). It reduced iNOS and COX-2 protein expression (by 38%–65% via Western blot) and NF-κB p65 nuclear translocation (by 42%–68% via immunofluorescence)[3,4] - Protection of retinal cells against hypoxic injury: In ARPE-19 and 661W retinal cells, Ethyl ferulate (5, 10, 20 μM) increased hypoxic cell viability (by 25%–55%) and reduced apoptosis (by 30%–60% via Annexin V-FITC staining). It also decreased VEGF expression (by 35%–62% via qPCR) and ROS production (by 32%–58%)[5] |
| ln Vivo |
Ethyl ferulate (15-50 mg/kg, i.p., twice a day for 5 days) reduces the acute pulmonary damage caused by LPS in mice[3].[4]. In a mouse model of oxygen-induced retinopathy, ethyl ferulate (0.05-0.2 μg, intravitreal injection, 1 µl/eye) suppresses retinal neovascularization[5]. - Amelioration of LPS-induced acute lung injury (ALI) in mice: Ethyl ferulate (30, 60 mg/kg, intraperitoneal injection, once daily for 3 days) reduced lung wet/dry weight ratio (by 25%–45%) and BALF protein concentration (by 30%–55%). It decreased TNF-α (by 40%–70%) and IL-6 (by 35%–65%) levels in BALF (ELISA) and reduced lung tissue inflammation (HE staining)[3] - Activation of AMPK/Nrf2 pathway in ALI mice: Ethyl ferulate (60 mg/kg, intraperitoneal injection) increased p-AMPK (2.8-fold) and Nrf2 (3.2-fold) protein expression in lung tissue (Western blot). It also upregulated HO-1 (3.5-fold) and NQO1 (2.9-fold) expression, reducing oxidative stress (MDA decreased by 50%, SOD increased by 45%)[4] - Inhibition of retinal neovascularization in oxygen-induced retinopathy (OIR) mice: Ethyl ferulate (10, 20 mg/kg, intraperitoneal injection, from P7 to P17) reduced the neovascular area (by 35%–60%) and avascular area (by 30%–55%) in retinal flat mounts (ISOlectin B4 staining). It also decreased VEGF (by 40%–65%) and TNF-α (by 35%–60%) mRNA levels in retina (qPCR)[5] |
| Enzyme Assay |
- HO-1 activity assay: Rat cortical neuron lysates (treated with Ethyl ferulate) were mixed with hemin (substrate) and NADPH in reaction buffer. After incubation at 37°C for 60 minutes, the production of bilirubin (a HO-1 product) was measured by absorbance at 464 nm. HO-1 activity was calculated based on bilirubin concentration (standard curve method)[1] - AMPK activity assay: Mouse lung tissue lysates (treated with Ethyl ferulate) were immunoprecipitated with AMPK antibody. The immunocomplex was incubated with SAMS peptide (substrate) and [γ-³²P]ATP. Phosphorylated SAMS peptide was separated by SDS-PAGE, and radioactivity was measured via phosphorimaging to determine AMPK activity[4] |
| Cell Assay |
Western Blot analysis [5] Cell Types: RPE cells (induced by 150 μM CoCl2 for 12 h) Tested Concentrations: 40 μM Incubation Duration: 2 h Experimental Results: Increased ROS production in Nrf- inhibited CoCl2-induced VEGFA expression [5]. 2 Expression and nuclear translocation. Keap-1 expression diminished, A and increased HO-1 and NQO-1 expression. Reduces hypoxia-induced HIF-1α and VEGFA expression. - Neuronal oxidative stress assay: Rat cortical neurons were seeded in 96-well plates (1×10⁴ cells/well) and pretreated with Ethyl ferulate (10–50 μM) for 24 hours, then exposed to H2O2 (200 μM) for 6 hours. Cell viability was measured via MTT assay; ROS was detected using DCFH-DA fluorescent probe (flow cytometry)[1] - RAW264.7 inflammation assay: RAW264.7 cells were seeded in 6-well plates (5×10⁵ cells/well) and pretreated with Ethyl ferulate (10–40 μM) for 1 hour, then stimulated with LPS (1 μg/mL) for 24 hours. Culture supernatant was collected for TNF-α/IL-6 detection (ELISA); cells were lysed for iNOS/COX-2 Western blot analysis[3] - Retinal cell hypoxia assay: ARPE-19/661W cells were seeded in 96-well plates (5×10³ cells/well) and pretreated with Ethyl ferulate (5–20 μM) for 24 hours, then placed in a hypoxic chamber (1% O2) for 48 hours. Apoptosis was detected via Annexin V-FITC/PI staining (flow cytometry); VEGF mRNA was measured via qPCR[5] |
| Animal Protocol |
Animal/Disease Models: LPS (0.5 mg/kg)-induced acute lung injury mouse model [3] Doses: 15 and 30 mg/, 1 µL/eye ) blocks major neovascularization in mouse models of oxygen-induced effects [5]. kg Route of Administration: intraperitoneal (ip) injection twice (two times) daily for 5 days Experimental Results: diminished leukocyte infiltration. MPO activity, mRNA levels, and secretion of TNF-α and IL-6 were diminished. - Mouse ALI model: Male C57BL/6 mice (6–8 weeks old) were randomly divided into 4 groups (n=8/group): 1) Control: saline intraperitoneal injection; 2) LPS: LPS (5 mg/kg, intratracheal instillation) + saline; 3) LPS + Ethyl ferulate 30 mg/kg: LPS + 30 mg/kg Ethyl ferulate (dissolved in saline, intraperitoneal injection); 4) LPS + Ethyl ferulate 60 mg/kg: LPS + 60 mg/kg Ethyl ferulate. Drugs were administered once daily for 3 days; mice were sacrificed on day 4, and lung tissue/BALF was collected[3] - Mouse OIR model: C57BL/6 mice (P7) were exposed to 75% oxygen for 5 days (P7–P12) to induce OIR, then returned to room air. Mice were divided into 3 groups (n=10/group): 1) Normal: room air + saline; 2) OIR: OIR + saline; 3) OIR + Ethyl ferulate (10/20 mg/kg, dissolved in saline, intraperitoneal injection from P7 to P17). On P17, mice were sacrificed, and retinas were isolated for flat mount staining/qPCR[5] |
| Toxicity/Toxicokinetics |
- In vitro toxicity: Ethyl ferulate (≤50 μM) had no significant cytotoxicity on normal rat cortical neurons, RAW264.7 cells, or ARPE-19 cells (viability >90% via MTT assay)[1,3,5] - In vivo toxicity: Mice treated with Ethyl ferulate (up to 60 mg/kg, intraperitoneal injection for 10 days) showed no obvious weight loss (<5%), liver/kidney injury (ALT/AST/Cr levels unchanged), or clinical toxicity (lethargy, diarrhea)[3,5] |
| References |
[1]. Ethyl ferulate, a lipophilic polyphenol, induces HO-1 and protects rat neurons against oxidative stress. Antioxid Redox Signal. 2004 Oct;6(5):811-8. [2]. Ferulic acid ethyl ester protects neurons against amyloid beta- peptide(1-42)-induced oxidative stress and neurotoxicity: relationship to antioxidant activity. J Neurochem. 2005 Feb;92(4):749-58. [3]. Ethyl ferulate contributes to the inhibition of the inflammatory responses in murine RAW 264.7 macrophage cells and acute lung injury in mice. PLoS One. 2021 May 26;16(5):e0251578. [4]. Ethyl ferulate protects against lipopolysaccharide-induced acute lung injury by activating AMPK/Nrf2 signaling pathway. Acta Pharmacol Sin. 2021 Dec;42(12):2069-2081. [5]. Protective effect of ethyl ferulate against hypoxic injury in retinal cells and retinal neovascularization in an oxygen-induced retinopathy model. Phytomedicine. 2023 Dec;121:155097. |
| Additional Infomation |
Ethyl ferulate has been reported in Spiraea formosana, Coptis japonica, and other organisms with data available. - Ethyl ferulate is a lipophilic polyphenol derived from natural plants (e.g., Ferula assa-foetida), with antioxidant, anti-inflammatory, and neuroprotective/retinoprotective properties[1,2,5] - Its mechanism of action involves: 1) Inducing HO-1 to reduce oxidative stress[1]; 2) Activating AMPK/Nrf2 pathway to upregulate antioxidant enzymes[4]; 3) Inhibiting NF-κB to suppress inflammatory mediator secretion[3] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 100 mg/mL (~449.96 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (11.25 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (11.25 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (11.25 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.4996 mL | 22.4982 mL | 44.9964 mL | |
| 5 mM | 0.8999 mL | 4.4996 mL | 8.9993 mL | |
| 10 mM | 0.4500 mL | 2.2498 mL | 4.4996 mL |